![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
68 Cards in this Set
- Front
- Back
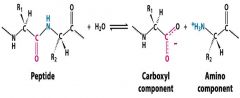
what kind of catalyses does proteinase catalyse in this reaction?
|
Proteinase, catalyses the hydrolysis of peptide bonds into a carboxyl and an amino group
|
|
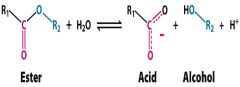
what kind of catalysis does proteinase hydrolyse in this reaction?
|
proteinase hydrolyses an ester bond into an acid and an alcahole
|
|
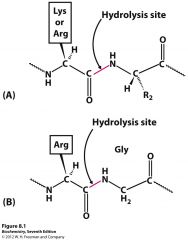
which reaction does the enzymes Trypsin and Thrombin cleave?
|
Trypsin reactions with the carboxy peptide bond next to LYs and ARg
Thrombin reacts with the Peptide bond on the carboxy side of Arg in a unique peptide sequence |
|
|
what are the 6 classes of enzymes?
Key: OTHLIL (Old truths have lived innocent love) |
1) Oxidoreductase
2) Transferases 3) Hydrolases 4) Lyases 5) Isomerases 6) Ligases |
|

what kind of enzyme is responsible for this reaction?
|
Oxidoreductases
Catalyse oxidation-reduction reactions • Eg. Lactate dehydrogenase (EC 1.1.1.27) |
|

what kind of enzyme is responsible for this reaction?
|
Transferases
Catalyse group transfer reactions • May require co-enzymes • Eg. Alanine transaminase (EC 2.6.1.2) |
|
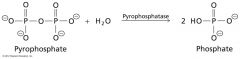
what kind of enzyme is responsible?
|
Hydrolases
• Catalyse hydrolysis • Eg. Pyrophosphatase (EC 3.6.1.1) |
|
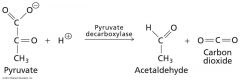
what kind of enzyme is responsible?
|
Lysases
Lysis of a substrate, generates a double bond • Not→ hydrolytic, oxidative • Reverse direction→ + 1 substrate to double bond of 2nd substrate • Synthase- lyase that catalyses an addition reaction • Eg. Pyruvate decarboxylase (EC 4.1.1.1) |
|

what kind of enzyme is responsible?
|
Isomerase
• Catalyse structural change within a molecule • Eg. Alanine racemase (EC 5.1.1.1) |
|
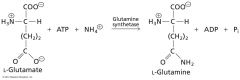
what kind of enzyme is responsible?
|
Ligases
• Catalyse ligation • Requires energy input (ATP) • Also called synthethetases • Eg. Glutamine synthetase (EC 6.3.1.2) |
|
|
what is an EC number?
|
an enzyme class number made of 4 numbers that classify a class of enzyme and its type of reaction that it catalyses
|
|
|
what are enzymes?
|
catalysts that effect only the rate (speed) of a reaction
|
|
|
how does an enzyme speed up a reaction?
|
by stabilising the transition state by lowering the amount of free energy of activation
|
|
|
the free energy of activation is higher during a catalysed or an uncatalysed reaction? why?
|
free energy of activation is higher during a catalysed reaction rather than an uncatalysed reaction,
hence a catalysed reaction uses more free energy to stabilise the transition state |
|
|
how does free energy of activation activate the reaction to go forward on its own?
|
the free energy of activation once added to the system is able to do this because it becomes activated and this free fall of energy forces reaction to move on its own, this it is called free energy
|
|
|
in the activation of free energy, the rate of reaction is proportional to what parameter?
|
reaction rate is proportional to concentration of the transitional state
|
|
|
what is activation energy?
|
the minimal energy needed to start a reaction
|
|
|
what is free enregy
|
the amount of work that a system can perform, it is free energy once it has free fallen past the transition state, after free energy of activation has been added to the system
|
|
|
how does the concentration of the transition state effect the rate of reaction
|
the higher the C of transitional state, the faster the reaction goes, hence C is proportional to Pressure
|
|
|
9s the free energy of the transitional state regarded as free energy that is released when the transitional state is converted into products?
|
no, it is a separate entity
|
|
|
describe classical competitive inhibition, what does it bind and what is the effect on Km and Vmax?
Keyword: race |
Competitive inhibitor competes with the substrate, it binds to the Enzyme, increasing Km and leaving Vmax unchanged
can be overcome by increasing [S] |
|
|
describe Uncompetitive inhibition, what does it bind and what is the effect on Km and Vmax?
Keyword: different |
Binds to the ES complex, lowering both Vmax and Km proportionally, the ratio of Vmax/Km (slop) stays the same
it Cannot be overcome by increasing [S] |
|
|
describe Noncompetitive inhibition, what does it bind and what is the effect on Km and Vmax?
Keyword: friends |
binds to the E or the ES complex, lowering Vmax only and leaving Km unchanged
It cannot be overcome by increasing [S] |
|
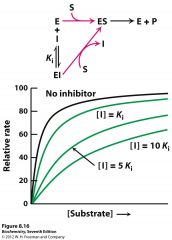
to which inhibitor type does this belong? how can you tell?
|
Competitive inhibition
since Km is increased, but Vmax is unchanged and only the Enzyme has been bound |
|
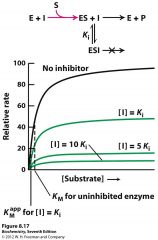
to which inhibitor type does this belong? how can you tell?
|
Uncompetitive inhibition,
since it lowers both Vmax and Km it binds to the ES complex only, forming ESI complex which results in an unproductive reaction since it is a dud substrate |
|
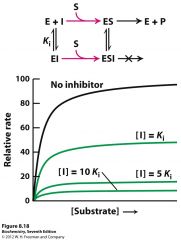
to which inhibitor type does this belong? how can you tell?
|
Noncompetitive inhibition
since it decreases Vmax and leaves Km untouched, the inhibitor binds to the E and ES complex |
|
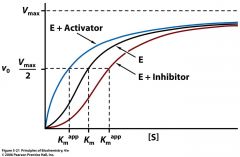
what does allosteric activation do to Km? what does allosteric inhibition do to Km?
|
activator shifts the sigmoidal curve towards hyperbola, lowering the Km and raising the activity of [S]
the inhibitor raises the Km and lower the activity of [S] |
|
|
what forces are involve in a substrate binding to an active site?
|
weak forces:
H bonds, Van der waals, and hydrophobic effect |
|
|
describe the arrangement of amino acids in a primary structure (sequence of aa's) Vs a tertiary arrangement
|
amino acids are far apart in a primary structure but close together in a tertiary structure, where a small portion of amino acids form the substrate binding site, the other aa's form the structural building material of the active site
|
|
|
why is the activation energy lower in a catalysed reaction compared to an uncatalysed reaction
|
because the catalyst enzyme stabilizes the Transitional state
|
|
|
if given a Lineweaver burk plot, with the value, y=0.174 x + 0.435, what is the Km and the Vmax?
how would you plot this? |
Vmax = 1/0.435 = 2.30
Km = 0.174 x 2.30 = 0.4 |
|
|
what is the site on an enzyme where a reaction occurs?
|
active site
|
|
|
what is the substance that the enzyme binds and converts to products?
|
the substrates
|
|
|
what are enzymes that dont have the required cofactor bound?
|
apoenzymes
|
|
|
the Kcat is referred to as the
|
turnover number
|
|
|
what is a frequent action which catalysis occurs?
|
stabilisation of the transition state
|
|
|
the steady state assumption as applied to enzyme kinetics, implies
|
the ES complex is formed and broken down at equivalent rates
|
|
|
true of false
at saturating levels of substrate, the rate of an enzyme catalyzed reaction is proportional to the enzyme concentration |
true
|
|
|
true or false
if enough substrate is added, the normal Vmax of a reaction can be attained even in the presence of a competitive inhibitor |
true
|
|
|
true or false
the rate of a reaction decreases steadily with time as substrate is depleted |
true
|
|
|
true or false, the activation energy for the catalyzed reaction is the same as for the uncatalyzed reaction, bu the equilibrium constant is more favourable in the enzyme catalyzed reaction
|
false
|
|
|
true or false
the michaelis menten constant Km equals the [S] at which V= half of Vmax |
true
|
|
|
enzyme accelerates the rate of a chemical reaction by doing what to the free energy of activation of the reaction
|
decreasing it
|
|
|
what kind of inhibitor has a structure similar to the substrate and reversibly binds to the active site of the enzyme
|
competitive inhibitor
|
|
|
the straigtht line kinetic plot of 1/Vo versus 1/S is called a
|
Lineweaver Burk Plot
|
|
|
give definition for initial rate
|
Vo
|
|
|
give definition for Vmax
|
maximal rate when all active sites are full
|
|
|
give definition for [S]
|
Substrate concentration
|
|
|
give definition for Km
|
michealis constant
|
|
|
Which of the following statements is FALSE?
Enzymes speed up the attainment of a reaction equilibrium. Enzymes lower the amount of energy needed for a reaction. Enzymes make reactions 103 to 1020 times faster. Enzymes are proteins. Enzymes are chemically unchanged during the actual catalytic process. |
Enzymes are chemically unchanged during the actual catalytic process.
|
|
|
Unlike typical catalyzed reactions in organic chemistry enzyme catalyzed reactions are
modulated to change activity levels. reaction specific. essentially 100% efficient. usually stereospecific. All of the above |
All of the above
|
|
|
The hydrophobic cleft in globular proteins which binds substrate molecules is called the ________.
substrate pocket active site modulator site oligomeric site activity site |
active site
|
|
|
An enzyme that catalyzes conversions of L-sugars to D-sugars is called a/an ________.
hydrolase lyase isomerase synthetase synthase |
isomerase
|
|
|
In a first order chemical reaction, the velocity of the reaction is proportional to the ________, while in a zero order reaction, the velocity of the reaction is proportional to the ________.
concentration of substrate; amount of enzyme concentration of substrate; speed of the reaction amount of enzyme; concentration of substrate speed of the reaction; concentration of substrate |
concentration of substrate; amount of enzyme
|
|
|
The main difference between chemical and enzyme kinetics is that
enzyme reactions depend on the concentration of the substrate. the rate constant for the formation of products is k2. enzyme reactions are altered by pH. enzyme reactions depend on the concentration of the enzyme and its recycling. |
enzyme reactions depend on the concentration of the enzyme and its recycling.
|
|
|
At the beginning of an enzyme-catalyzed reaction the ________ is negligible.
formation of E + P disappearance of ES formation of ES conversion of ES to E + S |
formation of E + P
|
|
|
The initial velocity of an enzyme reaction (v0) describes
the concentration of both at the start of the reaction. the concentration of the enzyme at maximal velocity. the concentration of substrate at maximal velocity. the rate of the reaction at when the substrate and enzyme are first mixed. |
the rate of the reaction at when the substrate and enzyme are first mixed.
|
|
|
Which enzyme below is fastest?
papain, kcat = 10 kinase, kcat = 10^3 carboxypeptidase, kcat = 10^2 catalase, kcat = 10^7 |
catalase, kcat = 10^7
|
|
|
The Michaelis constant, Km, is equal to the ________.
substrate concentration which gives the best enzyme assay for an enzyme reaction substrate concentration when the rate is equal to half its maximal value maximum velocity divided by two maximum velocity that any given enzyme reaction can achieve |
substrate concentration when the rate is equal to half its maximal value
|
|
|
The assumptions made in calculating the Michaelis-Menten Equation include
that the concentration of the substrate is much greater than the concentration of E. that the value of k-2 can be ignored. that the formation and decomposition of ES is the same for a period of time. A, B and C A and B only |
A, B and C
|
|
|
The (lower, higher) the value of Km, the (less, more) tightly the enzyme is bound to the substrate.
lower, less lower, more higher, less higher, more B and C |
B and C
|
|
|
It is difficult to determine either Km or Vmax from a graph of velocity vs. substrate concentration because
an asymptotic value must be determined from the graph. the graph is sigmoidal. too much substrate is required to determine them. the points on the graph are often not spread out on the hyperbola. |
an asymptotic value must be determined from the graph.
|
|
|
What distinguishes reversible inhibitors from irreversible inhibitors?
There is an equilibrium between bound and unbound reversible inhibitor. There usually is little back reaction for the binding of an irreversible inhibitor. Reversible inhibitors are not covalently bound to enzymes but irreversible inhibitors are. All of the above None of the above |
All of the above
|
|
|
An inhibitor binds to a site other than the active site of the enzyme. Which statement below correlates with this observation?
The inhibition must be irreversible. It must be a competitive inhibitor. It could be noncompetitive or uncompetitive inhibition. It could be irreversible, competitive, noncompetitive or uncompetitive. The data do not relate to the type of inhibition. |
It could be noncompetitive or uncompetitive inhibition.
|
|
|
Which statement is false about allosteric regulation?
Allosteric modulators bind non-covalently at sites other than the active site and induce conformational changes in the enzyme. The regulation usually is important to the conservation of energy and materials in cells. It is usually the mode of regulation for the last step in reaction pathways since this step produces the final product. Cellular response is faster with allosteric control than by controlling enzyme concentration in the cell. |
It is usually the mode of regulation for the last step in reaction pathways since this step produces the final product.
|
|
|
Allosteric modulators seldom resemble the substrate or product of the enzyme. What does this observation show?
Modulators always act as activators. The enzyme catalyzes more than one reaction. Modulators bind non-covalently to the enzyme. Modulators likely bind at a site other than the active site. |
Modulators likely bind at a site other than the active site.
|
|
|
At sufficiently high substrate concentration all types of reversible inhibition can be overwhelmed. At high substrate concentration the enzyme will be saturated and the reaction will proceed at the same maximum velocity as in the absence of inhibitor.
True False |
False
|
|
|
out of L-glyceradlahyde and D-Glyceraldehyde, which one occurs in vivo, which isomer is present in nature?
|
the D isomer occurs in nature and D isomer occurs in vivo
|

