![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
15 Cards in this Set
- Front
- Back
|
stability of resonance structures |
^ stability = ^ delocalization energy |
|
|
resonance contributor rules |
1. ^ stability = ^ contribution 2. ^ number contributors = ^ delocalization energy 3. ^ equivalency = ^ delocalization energy |
|
|
Isolated vs. conjugated dienes |
isolated = separated by more than 1 single bond conjugated = separated by 1 single bond more stable bc ^ # resonance structures |
|
|
Benzylic cation
5 resonance structures |
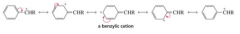
|
|
|
1,2 addition reaction |
occurs in conjugated dienes forms faster kinetic product
|
|
|
1,4 addition reaction |
adds to 1 and 4 position moves double bond over thermodynamic product |
|
|
kinetic product |
irreversible forms most rapidly occurs at low temps 1,2 addition |
|
|
thermodynamic product |
reversible most stable occurs at high temps 1,4 addition |
|
|
Diels-Alder reaction |
conjugated diene (C=C-C=C) + dienophile (C=C)
= cyclic molecule with R substituent
(1,4 addition) |
|
|
pericyclic reaction |
reaction taking place in one step by a cyclic shift in electrons |
|
|
cycloaddition reaction |
reaction in which two reactants form a cyclic product |
|
|
concerted reaction |
things occurring in the same step |
|
|
cyclic dienes |
form bridged bicyclic compounds |
|
|
exo configuration |
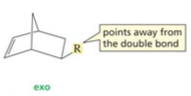
|
|
|
endo configuration
(diels alder rxns are endo-selective) |
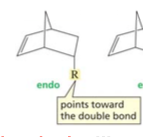
|

