![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
74 Cards in this Set
- Front
- Back
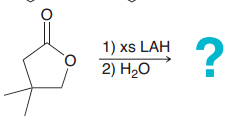
|
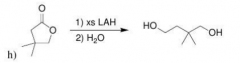
LAH will reduce Ester as far as possible (1. a. acyl substitution after Hydride attack => open up the ring / b. Hydride attack to open up the C=O / 2. protonation)
|
|
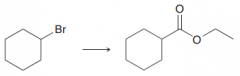
Propose a way to perform the following transformation?
|
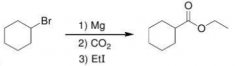
1). Make grignard 2)attack of gringnard on CO2 to give carboxylic acid 3)Esterification
|
|
|
Smell of many fruits are results by what type of compound?
|
Esters
|
|
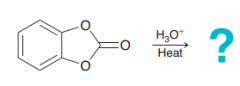
|
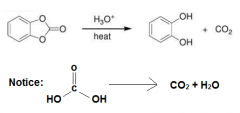
The molecule has two esteric bond which cleaves to give two alcholos and carboxylic acid with two OH on it which is unstable and decomposes to CO2
|
|
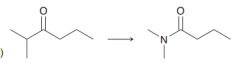
Propose reagents to preform the following transformation?
|
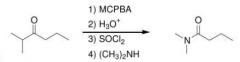
First the compound should be converted to an carboxylic acid and then to an amide (1-Byerviliger Oxidation -> insert O on the left side of C=O 2-acidic hydrolysis of Esters => carboxylic acids 3). Fromation of acid chlorides 4)Amonolysis of acid chlorides)
|
|
|
Acetaminophen
|
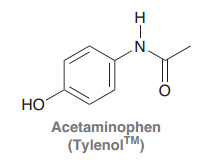
Benzene ring with hydroxyl and acetamide at para position
|
|
|
describe different position in regard to distance with C=O group?
|
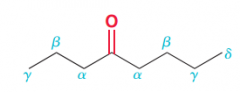
|
|
|
Enolate
|
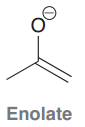
|
|
|
generally speaking in Keto-enol tautomerization, the position of equilibrium will significantly favor the ketone. In what circumstances the enol formation is favored? Example?
|
1.The enol has a conjugated P system (2) the enol can form an intramolecular H-bonding interactions between the hydroxyl proton and the nearby carbonyl group (e.g. beta-diketones and Phenols)
|
|
|
Why It is difficult to prevent tautomerization?
|
Because keto-enol tautomerization can still be catalyzed by the trace amounts of acid or base that are adsorbed to the surface of the glassware
|
|
|
Enolates are called ambient nucleophiles. Why?
|
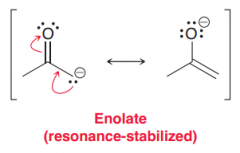
because they possess two nucleophilic sites, each of which can attack an electrophile (When the oxygen atom attacks an electrophile, it is called O-attack; and when the A carbon attacks an electrophile, it is called C-attack)
|
|
|
Enolates can show two types of nucleophilic attack. What are they? Which one is more common?
|
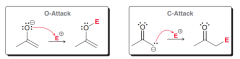
O-attack and C-attack . C-attack is more common (Although the oxygen atom of an enolate bears the majority of the negative charge)
|
|
|
Presence of C=O in compound, makes hydrogen in …..position acidic.
|
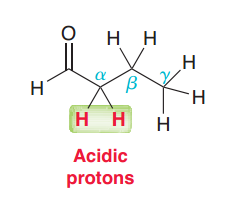
Alpha
|
|
|
Aldehydes and ketones typically have pKa values in the range of …..
|
Aldehydes and ketones typically have pKavalues in the range of 16–20
|
|
|
Judge the quality of removal of alpha hydrogen of aldehydes or ketons by a.EtO- b.NaH c.LDA
|
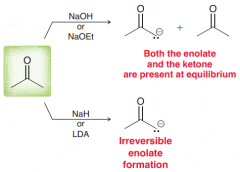
a. Equilibrium (but it does not favors enolate formation) / b.completely convert it to enolate / c.Equilibrium (but almost all of them are enlalte so equilibrium strongly favores formation of enolates)
|
|
|
What is LDA? What is used for?
|
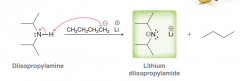
Lithium diisopropylamide. Used to deporotonate alpha position of C=O
|
|
|
With what kind of bases we can deprotonate the alpha position of beta-diketones?
|
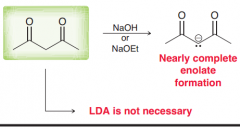
EtO- would be enough . LDA and NaH is not necessary (Since the H is in alpha position of two C=O is more acidic than usual alpha hydrogens)
|
|
|
Define Alpha Halogenation of ketones (in Acidic condition)?
|
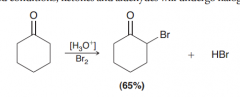
Under acid-catalyzed conditions and existence of Cl2, Br2 or I2, the alpha position (of C=O) with more substitution undreroes halogenation(solvent could be acetic acid, water, chloroform, and diethyl ether)
|
|
|
Define Alpha halogenation of ketones? (in basic condition)
|
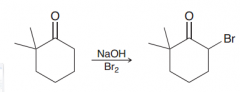
With bases like OH- , deprotonating at alpha position occurs then SN2 reaction
|
|
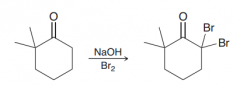
What is the main problem of halognation in basic condition? Why this happens?
|
Polyhalogenation because presence of the first halide makes the alpha position more acidic.
|
|
|
Explain mechanism of alpha halognation of C=O (in acidic condition) ? Which one is the rate determining step?
|
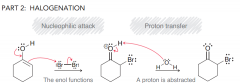
1.formation of enol (keto-enol tautormerizaiton) 2.Halogenation via nucleophilic attack (enol serves as a nucleophile to attack Br2). The rate determining step is the first one.
|
|
|
Is the alpha halogenation of C=O group autocatalytic? Why?
|
Yes. It produces HX which catalyzes the first step of the reaction
|
|
|
What is the meaning of autocatalytic
|
the reagent necessary to catalyze the reaction is produced by the reaction itself
|
|
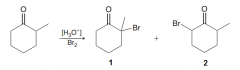
Which one the following products would be the major product of the alpha halogenation of the C=O group (1 or 2)? Why?
|
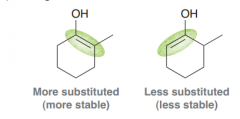
1. Because the first step of the reaction goes via the more substituted enol (the more stable alkene)
|
|
|
lithium carbonate? What is used for?
|
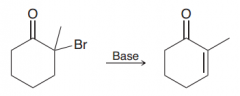
Li2CO3. It is a moderate base to perform elimination of the halides at the alpha position of C=O
|
|
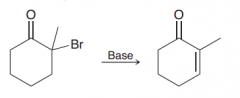
What kind of bases can perform the following E2 reaction?
|
Since the resulting alkene is conjugated even weak bases like tBuO- or pyridine can perform this reaction
|
|

What is the strategy introducing alpha,beta-unsaturation in a ketone? (e.g the following example)
|
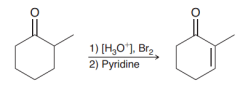
1. Alpha halogenation (acidic condition) 2.Elimination
|
|
|
What kind of compounds containing C=O group can not undergo alpha halogens (under acidic condition)? Why?
|
Carboxylic acids, esters, or amides because these functional groups are not readily converted to their corresponding enols.
|
|
|
Unlike ketones, Carboxylic acids can not undergo halogenation under acidic condition . How we can do alpha halogenation?
|

By Hell-Volhard-Zelinski reaction which is a 2 step reaction as described below
|
|
|
Define Hell-Volhard-Zelinski reaction?
|
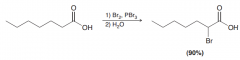
Halogenation of Carboxylic acids on alpha position by 1.Br2, PBr3 / 2. H2O [ Memorzie: vasate janhanam (hell) Ye bareye pokhte shode (Br) acid pashi mikone ru suratam (carboxylic acid) , vaghti dare suratam misuze par (PBr3) mikone tu chasham o bad shlinga ab ro mikone tu gusham (H2O) ]
|
|

Explain mechanism of Hell-Volhard-Zelinski reaction?
|
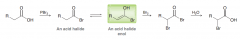
1.PBr3 gives acid halide 2.halogenation of C=O at alpha position 3.hydrolysis of acid halide to a carboxylic acid
|
|
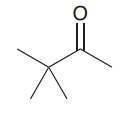
How a methyl ketone can be converted to the acetic acid?
|
By haloform reaction: 1.excess base and excess halogen ( a. three alpha halogenation in basic condition / b.Nuchelophilic attack of OH- to kick trihalomethyl out) / 2.Protonation
|
|
|
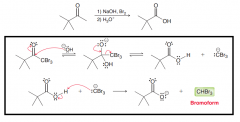
Define haloform reaction?
|
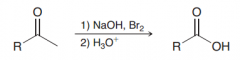
Converting methyl ketones (when the other side of the ketone has no Alpha-protons.) to carboxyilic acids by 1.excess base and excess halogen ( a. three alpha halogenation in basic condition / b.Nuchelophilic attack of OH- to kick trihalomethyl out) / 2.Protonation
|
|
|
In what condition halform reaction works the best?
|
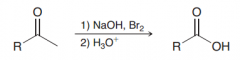
The haloform reaction is most efficient when the other side of the ketone has no Alpha-protons.
|
|
|
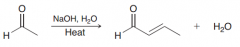
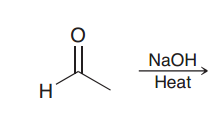
Define Aldol addition?
|
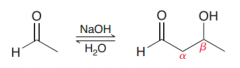
Addition of one aldehyde to itself via a nucleophile attack (to give a compound poses aldehyde and OH group at beta position)
|
|
|
What is the mechanism of aldol addition reaction?
|
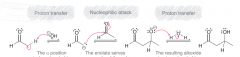
1.deporotonation of alpha position to give enolate / 2. Nuclelophilic attack on C=O / 3. protonation
|
|
|
Discuss the position of equilibrium in an aldol addition reaction for a. aldehydes b. ketones
|
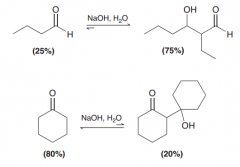
a. Equilibrium favors aldol product / b. Equilibrium disfavors aldol product
|
|
|
What happnes to the Beta-hydroxy ketones in the presence of NaOH, H2O (basic condition)?
|
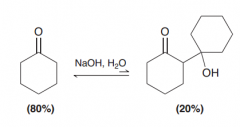
retro-aldol reaction to give two ketones
|
|
Despite the fact that aldol addition reaciton results in a poor yield for ketones, what would be the possible aldol product of the reaction? Why?
|
Carbonanion intermediate forms at the less substituted position
|
|
|
Define aldol condensation?
|
A two-step process (aldol addition plus dehydration) of an aldehye or ketone is called an aldol condensation. The final product is and alpha,beta-unsaturated compound
|
|
|
What condensation means? (e.g in aldol condesation)
|
any reaction in which two molecules undergo addition accompanied by the loss of a small molecule such as water, carbon dioxide, or nitrogen gas
|
|
|
In Aldol condesation, what kind of dehydration happens (E1, E2)?
|
Neither. It is called E1cb mechanism which unlike the other ones, the leaving group only leaves after deprotonation occurs
|
|
Predict the major product of aldol condensation?
|
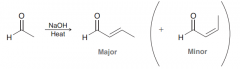
In aldol condensation cases where two stereoisomeric P-bonds can be formed, the product with the least steric hindrance is generally the major product.
|
|
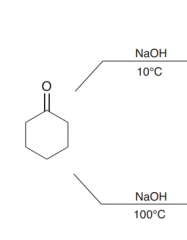
|
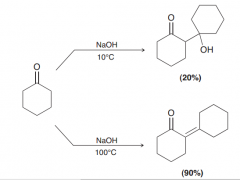
At low temperature = aldol addition / at higher temperature aldol condensation (notice: the yield of the top reaction is very poor. As explained earlier, the starting material is a ketone, and the equilibrium does not favor formation of the aldol addition product )
|
|
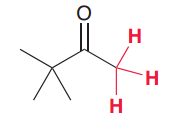
How we can easily draw the product of aldol condensation of the following compound?
|
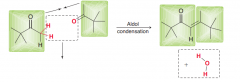
(1.Draw two molecules of ketones wehre O of C=O and two alpha Hydrogens are colse 2. remove the water molecule tconnect the carbons with a double bond as follows:
|
|
|
What is crossed aldol (mixed aldol) reaction?
|
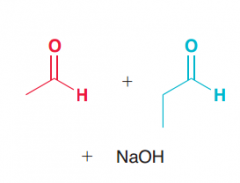
Aldol reaction with between two different aldehyde or ketone (wich result in a mixture of product of the possibility of aldol reaction between each compound with itself and the other compound
|
|
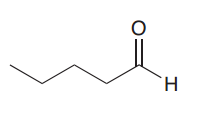
What would be the product of aldol condensation of the following compound?
|
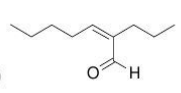
|
|
|
In what condition crossed aldol condensation would be practically useful?
|
1.when one of the starting material is formaldehyde and benzaldehyde which do not undergo self-aldol reaction / 2. directed aldol addition: Use LDA on of the starting materail to convert it to its enolate ion then mix it with second C=O containing comound
|
|
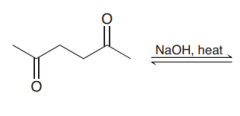
|
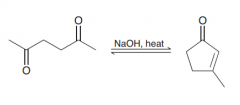
Intramolecular Aldol condensation
|
|
|
Define Claisen Condensation?
|
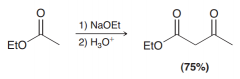
Reaction of an ester with itself in a basic condition via deprotonating and nucleophile attack of alpha position
|
|
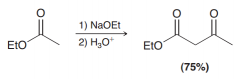
Discuss mecahnism of the Claisen condensation?
|
1. (a. deprotonation of alpha carbon / b. Acyl nucelphilic substitution) 2. protonation
|
|
|
can Hydroxide be used as the base for a Claisen condensation? Why?
|
No. because it can cause hydrolysis of the starting ester
|
|
|
What kind of base is usually used for Claisen condensation? Why?
|
RO- same as OR of the ester (because OH- cause hydrolysis of the ester and other OR- can force transesterification
|
|
|
In what condition crossed Claisen condensation is practically useful?
|
(1. one of esters has no alpha hydrogen) or (2.Using a base like LDA to deprotonate alpha hydrogen of one the esters prior to mixing with the other one)
|
|
|
Define Dieckmann cyclization?
|
It is just a Intramolecular Claisen Condensations [Memorize: Dick gir karde tu Closet e khune)
|
|
|
How to perform Alkylation of alpha position (of ketones) ?
|
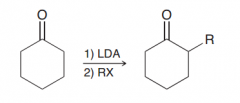
1)strong base like LDA to convert ketone completely to enolate ion (OH- and OR- cannot be used) 2) a simple SN2 reaction with an alkyl halides
|
|
|
Define thermodynamic enolate and Kinetic enolate?
|
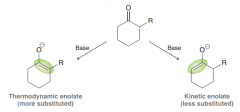
When forming enolates, The more-substituted enolate is more stable and is called the thermodynamic enolate . The less-substituted enolate is less stable, but it is formed more rapidly and is therefore called the
|
|
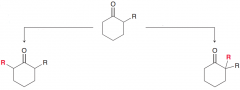
Write condition to perform the two following reactions?
|
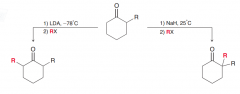
1.Alkylation via Kinetic enolate : low temperature and LDA // 2. Alkylation via thermodynamic enolate: Room temperature and a nonsterically hindered base (such as NaH)
|
|
|
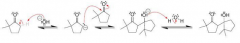
Define malonic ester synthesis?
|
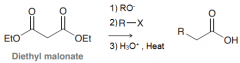
To synthesize carboxylic acid bearing an alkyl substitution on alpha poistion by 1)deprotonation of malonic ester at alpha position 2)SN2 attack on RX 3)Acidic hydrolysis to give a dicarboxylic acid compound 4)decarboxylation by heat to give monosubstituted acetic acid
|
|
|
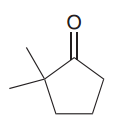
Malonic ester
|
|
|
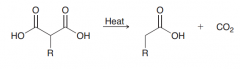
Explain mechanism of decarboxylation?
|
![1. [1,5] Pericyclic reaction 2.keto-enol tautomerization](https://images.cram.com/images/upload-flashcards/47/34/20/2473420_m.png)
1. [1,5] Pericyclic reaction 2.keto-enol tautomerization
|
|
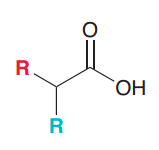
Synthesize the following compound by malonic ester synthesis?
|
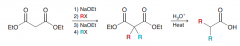
Two successive malonic ester synthesis
|
|
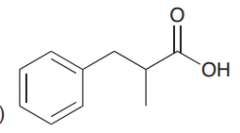
Synthesize by malonic ester synthesis?
|
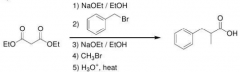
|
|
|
Define acetoacetic ester synthesis?
|
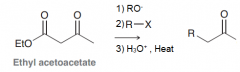
Synthesize ketones with substitution at alpha poistion (very similar to malonic ester synthesis) : 1)deprotonation of acetoacetic ester at alpha position 2)SN2 attack on RX 3)Acidic hydrolysis to give a compound with two C=O at one carbon 4)decarboxylation by heat to give monosubstituted acetic acid 5)keto-enol tautomerization
|
|
|
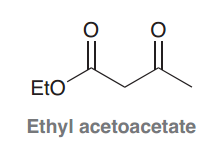
acetoacetic ester ?
|
|
|
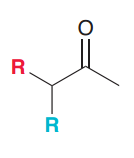
Synthesize the following compound by acetoacetic ester synthesis?
|
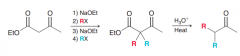
|
|
|
Define michael donor and acceptor?
|
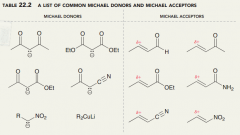
the doubly stabilized enolate is called a Michael donor, while the Alpha,Beta-unsaturated aldehyde is called a Michael acceptor
|
|
|
Define michael reaction?
|
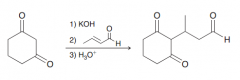
A 1,4-conjugate addition of a Micheal donor to the beta position of a Micheal acceptor to give a 1,4-conjugate addition (which after keto-enol tautomerization seems like a 1,2-addition)
|
|
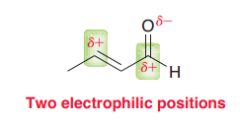
There are two electrophiles in an alpha,beta-unsaturated compoud. Explain what kind of carbon nucleophile attacks which one of these elecctophilic sites? (a. Grignard reagent RMgX / b.Gilman reagent R2CuLi)
|
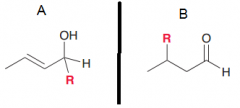
a. Attacks C=O b. attacks double bond to gaive 1,4-addition (which after keto-enol tautomerization seems like a 1,2 addition)
|
|
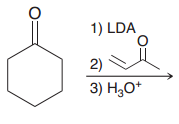
|
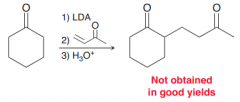
Michael reaction does not occur because michael donor should be doubly stabilized enolates
|
|
|
Define Stork enamine synthesis? What is used for?
|
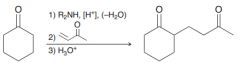
To perform a michael reaction on a simple ketone by (1) formation of an enamine, (2) a Michael addition, and (3) hydrolysis.
|
|
|
What is Robinson annulation reaction?
|
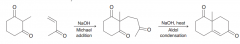
A two step process to make a ring by 1). Michael addition 2) aldol condensation
|
|
|
How to do Alkylation of the Alpha and Beta Positions for ketones?
|
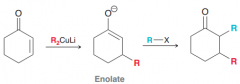
1). Michael addition (=> beta substution) 2). Treatment with RX (SN2 reaction)
|
|
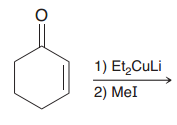
|
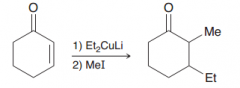
1). Michael addition (=>1,4-conjugate addition and formation of enolate) 2). SN2
|

