![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
37 Cards in this Set
- Front
- Back

What is the pH of a solution that tested pink, orange, yellow and clear in each of the indicators.
|
pH = 5
Make sure that you can interpret this chart for indicator effective range and for the specific pH of a solution that test a combination of colors. Reference the Acid-base Indicators Worksheet. |
|
|
What ions do an Arrhenius acid give in solution?
|
H+
and/or H3O+ |
|
|
What ions do Arrhenius bases give in solution?
|
OH-
|
|
|
What acid is in your stomach?
What acid is in vinegar? What acid is in some fruits? |
HCl - Hydrochloric acid
HC2H3O2 - Acetic acid Citric Acid |
|
|
What is the term for the separation of ions that occurs when an ionic compound is dissolved?
|
dissociation
|
|
|
What is the term for any process that results in the formation of ions?
|
ionization
|
|
|
What are some properties of acids?
|
Have a pH below 7.0.
Are corrosive and can burn your skin. Acids have a sour taste. Acids turn litmus paper red. Acids react with metals to produce hydrogen gas. Acids react with bases in a neutralization reaction to produce salt and water. |
|
|
What are some properties of bases?
|
Have a pH above 7.0. Bases are caustic and can burn your skin. Bases have a bitter taste. Bases turn litmus paper blue. Bases react with acids in a neutralization reaction to produce salt and water. |
|
|
What is the term for an acid or base that CAN conduct electricity in solution?
|
An electrolyte.
A strong electrolyte conducts well. A weak electrolyte barely conducts. A non-electrolyte does not conduct. |
|
|
Be able to classify each part of the pH scale as an acid or base.
|
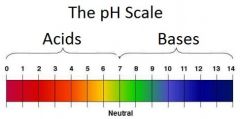
|
|
|
Describe the pH of some common household chemicals.
|

|
|
|
What is the difference between a strong acid and a weak acid?
|
The difference is the amount of ions that dissociate. A strong acid has almost 100% dissociation and a weak acid only has a partial dissociation.
|
|
|
What is the difference between a strong base and a weak base?
|
The difference is the amount of ions that dissociate. A strong base has almost 100% dissociation and a weak base only has a partial dissociation.
|
|
|
What is the difference between monoprotic, diprotic, and polyprotic acids.
|
The number of H+ ions that can dissociate from the acid.
Mono = HCl Di = H2SO4 Poly = H3PO4 |
|
|
What is the definition of an Arrhenius acid?
|
A compound that increases the concentration of hydrogen ions (H+) in solution.
|
|
|
What is the definition of an Arrhenius base?
|
A compound that increases the concentration of hydroxide ions (OH-) in solution.
|
|
|
What is the definition of a Bronsted-Lowry a
|
A molecule or ion that is a proton (H+) donor.
|
|
|
What is the definition of a Bronsted-Lowry base?
|
A molecule or ion that is a proton (H+) acceptor.
|
|
|
What is the term for a substance that can act as both an acid or a base?
|
amphoteric
|
|
|
In the Bronsted-Lowry theory, what is the conjugate base?
|
It is the new compound formed after the B-L acid donated the proton (H+).
|
|
|
In the Bronsted-Lowry theory, what is the conjugate acid?
|
It is the new compound formed after the B-L base accepted the proton (H+).
|
|
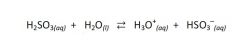
In the reaction, be able to identify:
The B-L acid, B-L base, conj. acid, & conj. base. |
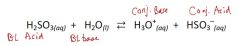
|
|
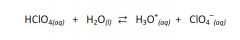
In the reaction, be able to identify:The B-L acid, B-L base, conj. acid, & conj. base.
|
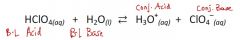
|
|
|
What is the definition of a Lewis Acid?
|
An electron pair acceptor.
|
|
|
What is the definition of a Lewis base?
|
An electron pair donor.
|
|
|
What is the formula to calculate the pH of a solution if you know the [H+]?
|
pH = -log[H+]
|
|
|
What is the formula to calculate the pOH of a solution if you know the [OH-]
|
pOH = -log[OH-]
|
|
|
What is the formula to calculate the hydrogen ion concentration if the pH is known?
|
[H+] = antilog(-pH)
|
|
|
What is the formula to calculate the hydroxide ion concentration if the pH is known?
|
[OH-] = antilog(-pOH)
|
|
|
Calculate the pH of a 0.0035 M HBr solution.
|
2.45
pH = -log(0.0035) = 2.45 |
|
|
Calculate the pOH of a 0.02 M NaOH solution.
|
1.7
pOH = -log(0.02) = 1.7 |
|
|
Calculate the hydrogen ion concentration of a acidic solution with a pH of 4.5.
|
0.000032
[H+] = antilog(-4.5) = 0.000032 |
|
|
Calculate the hydroxide ion concentration if the pOH of the solution is 3.8.
|
0.00016
[OH-] = antilog(-3.8) = 0.00016 |
|
|
If the pH is 4, what is the pOH?
|
10
pH + pOH = 14 |
|
|
If the pOH is 6, what is the pH?
|
8
pH + pOH = 14 |
|
|
What is the pH of a 0.0038 M NaOH solution.
|
11.6 This is the concentration that makes it a base... pOH = -log(0.0038) = 2.4 pH + pOH = 14 pH + 2.4 = 14 pH = 11.6 |
|
|
Calculate the hydrogen ion concentration of a 0.00064 M NaOH solution.
|
0.0000000000158 or 1.58 x 10 -11
*CAUTION* Use the base to calculate the pOH. (3.2) 14 - 3.2 = 10.8 (pH) [H+] = antilog (-10.8) = 0.0000000000158 |

