![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
33 Cards in this Set
- Front
- Back
|
Kinetic energy is the energy of ______: |
Motion. |
|
|
Chemical energy is a form of _____ energy: |
Potential. |
|
|
In your body, what process converts the chemical energy found in glucose into the chemical energy found in ATP? |
Cellular respiration. |
|
|
What are the by-products of cellular respiration? |
Heat, carbon dioxide, and water. |
|
|
The process of cellular respiration, which converts simple sugars such as glucose into CO2 and water, is an example of _____: |
A catabolic pathway. Cellular respiration is a catabolic pathway. |
|
|
Bonds are a form of _____ energy: |
Potential energy. |
|
|
A chemical reaction in which both the reactants and products are not being produced or used in any active metabolic pathway is an example of: |
Chemical equilibrium. |
|
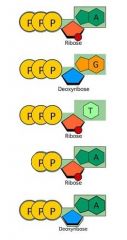
Which one of these molecules is ATP? |
The first one: Three phosphates attached to a ribose attached to an adenine. |
|
|
What type of reaction breaks the bonds that join the phosphate groups in an ATP molecule? |
Hydrolysis. |
|
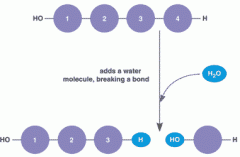
What type of reaction is this? |
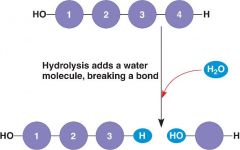
Hydrolysis. |
|
|
The following reaction A --> B + C + heatis a(n) _____ reaction: |
Exergonic. Energy has been released. |
|
|
What is a metabolic or chemical process accompanied by the release of heat/energy: |
Exergonic. |
|
|
What is a metabolic or chemical process accompanied by or requiring the absorption of energy, the products being of greater free energy than the reactants. |
Endergonic. |
|
|
A(n) _____ reaction occurs spontaneously: |
Exergonic. In exergonic reactions the products have less potential energy than the reactants. |
|
|
What type of reaction requires a net input of energy from its surroundings? |
Endergonic. The products of endergonic reactions have more potential energy than the reactants. |
|
|
The reaction ADP + P --> ATP is a(n) _____ reaction: |
Endergonic. Energy has been acquired from the surroundings |
|
|
The energy for an endergonic reaction comes from a(n) _____ reaction: |
Exergonic. The energy released by an exergonic reaction can be used to drive an endergonic reaction. |
|
|
What is the fate of the phosphate group that is removed when ATP is converted to ADP? |
It is acquired by a reactant in an endergonic reaction. By acquiring the phosphate group the reactant acquires energy. |
|
|
The products contain more potential energy than the reactants. This a(n) ________ reaction: |
Endergonic. |
|
|
This type of reaction releases heat/energy. |
Exergonic. |
|
|
What is energy coupling? |
The use of energy released from an exergonic reaction to drive an endergonic reaction |
|
|
Enzymes work by _____: |
Reducing EA. Energy of activation. |
|
|
What name is given to the reactants in an enzymatically catalyzed reaction? |
Substrate. |
|
|
What is a catabolic pathway? |
Release of energy. Breaking down complex molecules into simpler ones. |
|
|
What is an anabolic pathway? |
Consumption of energy. Building complex molecules from simpler ones. |
|
|
What is a closed system? |
Isolated from surroundings. Does not interact with other systems. |
|
|
What is an open system? |
Energy and matter can be transferred between systems and its surroundings. |
|
|
What is the second law of thermodynamics? |
The Second Law of Thermodynamics Every every transfer or transformation INCREASES entropy (disorder) of the universe. -Energy transformations proceed spontaneously to convert matter from a more ordered, less stable form to less ordered. -No process is 100% efficient - energy is lost. |
|
|
What is phosphorylation? |
The addition of a phosphoryl group (PO3^2-) |
|
|
What is dephosphorylation? |
The removal of a phosphoryl group. |
|
|
What are the three properties of enzymes? |
1. An enzyme lowers the activation energy for a given reaction 2. An enzyme is not changed or altered. 3. Names often end with "-ase" |
|
|
What is activation energy? |
energy input required to destabilize exisiting chemical bonds and initiate a chemical reaction for both endergonic and exogonic reactions. |
|
|
Higher activation energy = _____ reaction. |
Slower. |

