![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
76 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Crystalline Solid |
Solid with atoms/ions/molecules in an ordered arrangment |
|
|
|
Unit Cell |
Smallest unit of crystalline solid that can be repeated |
|
|
|
Coordination Number |
Number of "things" connected to central ion/atom |
|
|
|
Simple Cubic Unit Cell |
Particles' centres are at corners of cube |
|
|
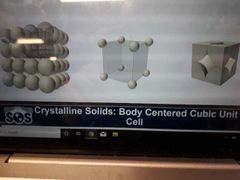
Cordination number, number of atoms per unit cell, and packing |
Cordination number: 6 Number of atoms per unit cell: 1/8 x 8= 1 Packing: Inneficient (52.4%) |
|
|
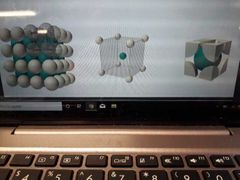
Body centred Cube Cordination number, number of atoms per unit cell, packing |

|
|
|
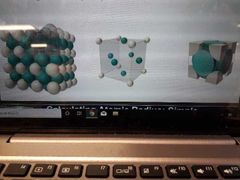
Face centred cube Coordination number, number of atoms per unit cell, packing |

|
|
|
|
Increasing energy of intermolecular forces |
London dispersion forces < dipole dipole < h-bonding < ion dipole < ionic bonding |
|
|
|
Ion dipole |
An ion attracting a polar molecule A cation (+) attracts a partially negative part of polar molecule An anion (-) attracts partially posotive part of polar molecule |
|
|
|
Dipple dipole |
Polar molecules attracting one another due to dipole moment |
|
|
|
How to know if a molecule is polar |
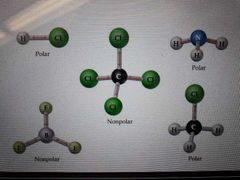
If a molecule is symmetric = non polar = no dipole dipole If molecole is asymetric = polar = dipole dipole |
|
|
|
Hydrogen bonding |
Polar molecules with H-X bonds attracting one another where X is N, F, or O |
|
|
|
What goes on in hydrogen bonding? |
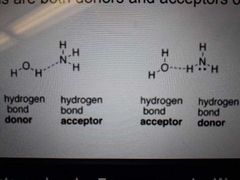
H is attracted to lone pair on X of another molecule H is donated while lone pair X of another molecule accepts H H-X compounds are both donors and acceptors of hydrogen bond |
|
|
|
Van der Waals/London Dispersion Forces |
Any molecule attracting another molecule due to induced dipole movment |
|
|
|
Which molecules have the highest LDF? |

Higher total e- and atomic size |
|
|
|
Vapour pressure |

Pressure of gas (VP) over a liquid at a specific temperature (T) when evapouration rate equals condensation rate |
|
|
|
Boiling point |
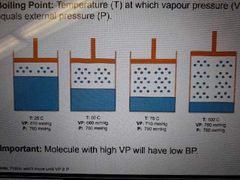
Temperature (T) at which vapour pressure (VP) equals external pressure (P) |
|
|

|

|
|
|
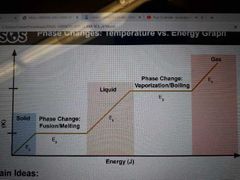
What are the main ideas from this graph (3) |
Energy is used to beak down intermolecular forces, not increased temperature Temperature does not change during phase change (Ep) Temperature only changes between phase changes (Ek) |
|
|
|
|
|
|
|
Phase diagram |
Pressure vs Temperature graph that shows phases as well as boundaries at which phases exist at equillibrium |
|
|
|
Triple point |
Pressure and temperature where all 3 phases are in equillibrium |
|
|
|
Critical point |
Pressure and temperature above which liquid and gas dissapear and a supercritical fluid exists |
|
|
|
Slope |
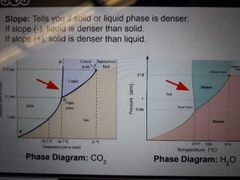
Tells you if solid or liquid phase is denser |
|
|
|
|
|
|
|
Boyle's Law |

Volume is inversely proportional to pressure |
|
|
|
Charles' Law |
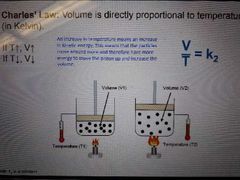
Volume is directly proportional to pressure (in Kelvin) |
|
|
|
Avogadro's Law |
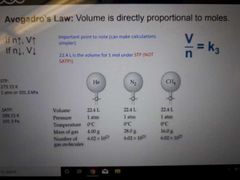
Volume is directly proportional to moles |
|
|
|
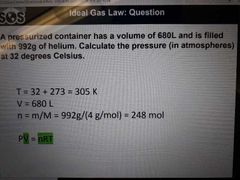
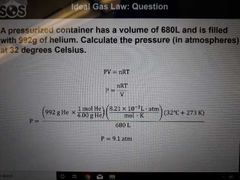
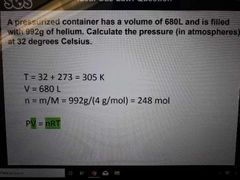
Ideal gas law |
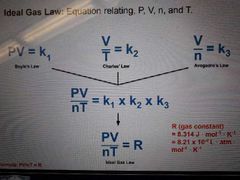
Equation relating, PV=nRT |
|
|
|
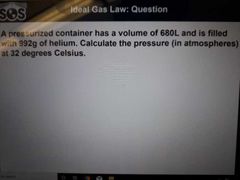
What is an ideal gas (5) |
1. The particles are negligible in size vs container volume (no space) 2. The particles are in constant motion 3. The average kinetic energy is proportional to the absolute temperature 4. No intermolecular forces occur between gas molecules 5. Gas particles undergo elastic collisions with container walls therefore no net energy is lost |
|
|
|
|
|
|
|
|
|
|
|
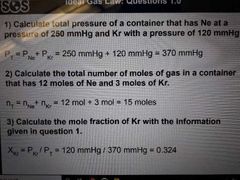
Dalton's Law of Partial Pressures (2) |

1) Total pressure of a mixture of gasses is equal to the sum of individual gas pressures 2) total number of moles of a mixture of gases is equal to sum of individual gas moles |
|
|
|
Mole fraction (2) |

1) moles of specific gas divided by total moles of gases in mixture 2) pressure of specific gas divided by total pressure of gases in mixture |
|
|

|
|
|
|

|

|

|
|
|
Gases are MOST ideal at (high/low) temoerature and (high/low) pressure |
Gases are MOST ideal at HIGH temperature and LOW pressure |
|
|
|
What is Z |
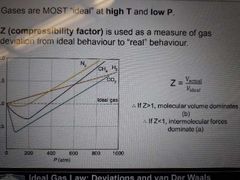
Used as a measure of gas deviation from ideal behaviour to 'real' behaviour |
Compressibility factor |
|
|
If Z>1 If Z<1 |
When Z>1 molecular volume dominates When Z<1 intermolecular forces dominate |
|
|
|
Deviation from Van der Waals equation |

|
|
|
|
Effusion |
Gas molecules moving from one space to another through small hole |
|
|
|
Lighter and higher temperature = (greater/lesser) average speeds = effuse (slower/faster) |
Lighter and higher temperatures = greater average speeds = effuse faster |
|
|
|
Graham's Law of Effusion |

|
|
|
|
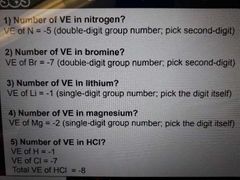
Six bonding rules in Lewis structures |

|
|
|

|
|
|
|

|

|
|
|
|
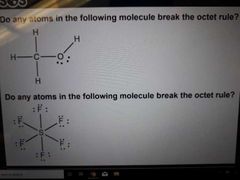
Octet rule Exceotions? |

Each atom in compound must be surrounded by 8e- Exceptions: H(2e-) B(6e-) and atoms in period 3 or greater (10e-, 12e-, 14e-) |
|
|
|

|
|
|
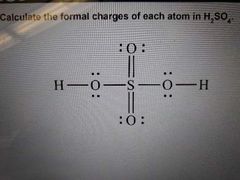
|

|
|
|

|

|
|
|
|
Resonance structures |

Two or more Lewis structures that describe what a molecules looks like These Lewis structures differ only in the placement of the electrons. Double headed arrows are used to differentiate each Lewis structure |
|
|
|
When drawing resonance structures remember... (3) |

1) draw the initial Lewis structure normally 2) move electrons without breaking the octet rule. If you can't, structure doesn't have resonance 3) want FC to be as close to 0. If you can't get it to 0, put (-) FC on more electronegative atom and (+) FC on more electroposotive atom - molecule will be more stable this way |
|
|
|
VESPR |
Provides molecular geometry/shape and hybridization |
|
|
|
AXE method |
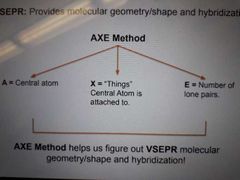
Helps us figure out VSEPR molecular geometry/shape and hybridization |

|
|
|
Using AXE, what is AX3 |

|
|
|
|
Using AXE, what is AX4 |

|
|
|
|
Using AXE, what is AX5 |

|
|
|
|
Using AXE, what is AX6 |

|
|
|
|
What does AXE look like in terms of hybridization? |

|
|
|
|
How do you calculate hybridization? |

|
|
|
|
Bond energy |
Energy required to break an intramolecular bond |
|
|
|
Energy absorbed (+) during product breaking = (reactant/product) side Vice versa? |
Energy absorbed during bond breaking is reactant side |
|
|
|
Enthalpy of reaction DeltaH = (-) vs (+) means... |
A measure of whether the overall chemical reaction is endothermic or exothermic DeltaH = (-) when products release more energy than reactants absorb energy DeltaH = (+) when reactants absord more energy than products releasing energy |

|
|

|

|
|
|
|
Molecular orbital (MO) |
MOs are formed from mixing of atomic orbitals |
|
|
|
Types of MOs (4) |

|
|
|
|
Sigma bond for H2 State electron configuration, valence electrons, and atomic orbitals of H and draw diagram Shows and state bond order |

|
|
|
|
Pi bond for C2State electron configuration, valence electrons, and atomic orbitals of C and draw diagramShows and state bond order |
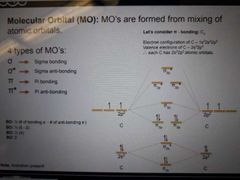
|
|
|
|
2 arrangments of Pi bonding. Which molecules belong to each? |
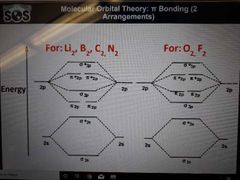
|
|
|
|
What is LUMO |
Lowest Unoccupied Molecular Orbital |
|
|
|
What is HOMO |
Highest Occupied Molecular Orbital |
|
|
|
What is bond order |
1/2 (# of bonding e- substract # of antibonding e-) |
|
|

Where and what is the atomic number, mass number, and explain isotopes |

|
|
|
|
Draw and label a simple light spectrumn Where is f, E, and wave length highest? Lowest? |
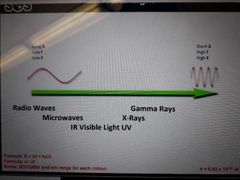
|
|
|
|
What is the nm range of visible light? |
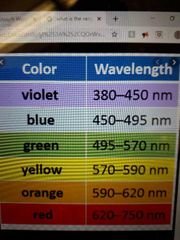
|
|
|

|

|
|

