![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
78 Cards in this Set
- Front
- Back
|
Aldehyde: Formula |
-CHO |
|
|
Ketone: formula |
C-C(=O)-C |
|
|
Carboxylic acid: Formula |
-COOH |
|
|
Alkane |
homologous series with formula C(n)H(2n+2) |
|
|
Displayed formula |
a formula showing the relative positioning of all the atoms in a molecule and the bonds between them |
|
|
Functional group |
The part of an organic molecule responsible for its chemical reactions |
|
|
Homologous series |
A series of organic compounds with the same functional group but with each successive member differing by -CH2 |
|
|
Skeletal formula |
A simplified organic formula with H atoms removed leaving just a carbon skeleton and associated functional group |
|
|
Structural isomerism |
Molecules with the same molecular formula but with a different structural arrangement of atoms |
|
|
Stereo isomerism |
Molecule with the same structural formula but a different arrangement of atoms in space |
|
|
2 things need for E/Z isomerism |
1 - C=C with no free rotation 2 - Each carbon of C=C must have two different groups |
|
|
Cis-trans isomerism |
special type of E/Z isomerism in which there is a non-hydrogen group & a hydrogen group on each carbon of C=C |
|
|
Shape of ethene |
- all bond angles are 120 - molecule is planar - no free rotation |
|
|
Formation of pie bond |
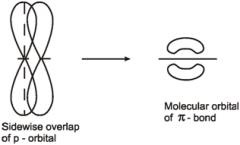
|
|
|
Reactions of alkenes: Addition of Bromine |
- Br2 - room T&P |
|
|
Reactions of alkenes: Addition of hydrogen halides |
- e.g HBr - room T & P |
|
|
Reactions of alkenes: Hydration |
- H20 - High T & P - uses H3PO4 catalyst - forms alcohols |
|
|
Reactions of alkenes: Hydrogenation |
- H2 - High T & P - Nickel catalyst - makes alkanes |
|
|
Electrophilic addition mechanism |
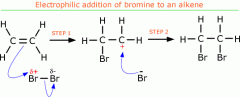
|
|
|
Mechanism of addition of hydrogen halides |
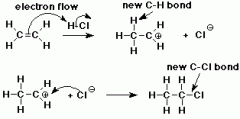
|
|
|
why do alkenes attract electrophiles? |
The double bond of C=C is a region of high electron density |
|
|
Electrophile |
A species capable of accepting a pair of electrons |
|
|
Atom economy |
Mr of desired product / Sum of Mr of all products (x100) |
|
|
Percentage yield |
actual yield / theoretical yield (x100) |
|
|
Why is 100% yield rarely achieved? |
- reaction may be at equilibrium - side reactions may occur - reactants may not be pure - some reactants and products may be left behind in the apparatus |
|
|
Polymer |
A long molecular chain built up of monomer units |
|
|
Pi bond |
Reactive part of a C=C formed above and below the plane of the bonded atoms by sideways overlap of porbitals |
|
|
polymer repeat unit |
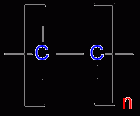
|
|
|
problems with polymers |
- they are not biodegradable - on combustion they produce toxic or acidic gases - none renewable feedstock |
|
|
Solutions to polymers |
- uses plastics made from starches - high temperature combustion with scrubbers - sort and recycle - crack polymers to short alkane chains and alkenes to use as feedstocks |
|
|
Activation energy |
The minimum energy required to start a reaction by the breaking of bonds |
|
|
Enthalpy |
The heat content that is stored in a chemical system |
|
|
Q= |
mc/\T |
|
|
/\H= |
-Q (in kJ) / mole |
|
|
Standard conditions |
T: 25oC / 298K P: 100 kPa / 1 atmosphere C: 1.0dm-3 |
|
|
Standard Enthalpy change of combustion |
is the enthalpy change that takes place when one mole of substance reacts completely with oxygen under standard conditions with all reactants and products in their standard states |
|
|
Standard Enthalpy change of Formation |
of a compound is the enthalpy change that takes place when one mole of a compound is formed form its constituent elements in their standard states under standard conditions |
|
|
Standard Enthalpy change of Reaction |
is the enthalpy change that accompanies a reaction in the molar quantities expressed in a chemical equation under standard conditions and states |
|
|
Why does the composition of crude oil mean it is not suitable as an energy source? |
- varies from different sources - doesn't ignite easily as contains mostly straight chained alkanes - contains nitrogen and sulfur compounds (acid rain) |
|
|
Fractional distillation |
The separation of the different components of oil according to their boiling points |
|
|
Catalytic cracking |
- The conversion of a long chain alkene into a shorter alkane and alkene - requires heat and Zeolite catalyst - shorter chains are in higher demand (fuels) |
|
|
2 refining crude oil techniques |
- Isomerism (unbranched to branched) - Reforming (unbranched to cyclic) both burn more efficiently |
|
|
Hess' Law |
If a reaction can take place by more than one route and the initial and final conditions are the same, the total enthalpy change is the same for each route |
|
|
Hess cycle for enthalpy change of combustion |
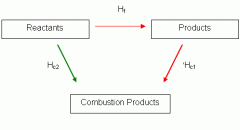
|
|
|
Hess cycle for enthalpy change of formation |
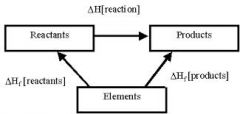
|
|
|
Average bond enthalpy |
The average energy change that takes place when one mole of a given type of bond in molecules of a gaseous state is broken by homolytic fission |
|
|
Define: Radical |
A species with an unpaired electron |
|
|
What type of bond fission produces free radicals? |
Homolytic fission |
|
|
Substitution reaction |
A reaction in which an atom or group of atoms is replaced with a different atom or group (2 reactants -> 2 products) |
|
|
3 stages of a substitution reaction involving free radicals |
Initiation, propagation and termination |
|
|
Why do free radical substitution reactions produce a mixture of products? |
Termination is a random process and many different radicals and halogenoalkanes can form in the mixture so many different termination reactions can occur |
|
|
Define: Nucleophile |
A species capable of donating a pair of electrons to form a new covalent bond |
|
|
What is reflux? |
Continuos boiling and condensing of a reaction mixture |
|
|
Nucleophilic substitution mechanism |
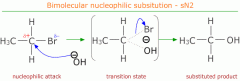
|
|
|
What are CFC's? |
Chlorofluorocarbons are any molecules which contain C, F, and Cl only |
|
|
Overall reaction of O2 with UV in atmosphere |
302 <=> 203 |
|
|
Reaction of free radicals and ozone |
R* + O3 -> RO* + O2 RO* + O3 -> R* + 2O2 |
|
|
Alternatives to CFC's |
- Hydrocarbons - CO2 - perfluorocarbons - HCFC's |
|
|
Examples of Nucleophiles |
NH3 H2O X- OH- |
|
|
Making ethanol by fermentation of sugars |
C6H12O6 ->2CH3CH2OH+ 2CO2 Requires no oxygen, 37oC, sugar in aq and presence of yeast |
|
|
Making ethanol by hydration of ethene |
H2C=CH2 + H20 -> CH3CH2OH Requires high t (300oC) & p (60atm) H3PO4 |
|
|
Uses of ethanol and methanol |
- alcoholic drinks, fuel, methylated spirits (solvents) - fuel, feedstock for chemical industry |
|
|
Dehydration of an alcohol |
high temperatures and H2SO4 |
|
|
Oxidation of alcohols |
- potassium dichromate (VI) solution - colour change from orange to green/blue |
|
|
Oxidation of primary alcohol |
Mild - aldehyde Stronger - carboxylic acid |
|
|
Oxidation of secondary alcohol |
Ketones |
|
|
Oxidation of Tertiary alcohol |
resistant to oxidation |
|
|
Esterification |
Carboxylic acid + alcohol -> ester + water heat under reflux and conc H2SO4 catalyst yl-oate yl - from alcohol and oate - from acid |
|
|
Define: rate of reaction |
The change in the concentration of a reactant at a given time |
|
|
Botlzmann distribution |
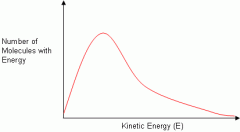
|
|
|
Effect of temperature on Botlzmann distribution |
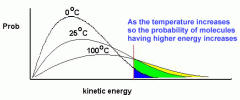
|
|
|
Define: Catalyst |
A substance that increases the rate of reaction without being used up in the process |
|
|
Heterogeneous and homogenous catalysts |
hetero - has different physical state from reactants (e.g Ni in hydration of ethene) homo - same physical state as reactants (e.g H2SO4 in esterification) |
|
|
Overall reaction of catalytic converter in cars |
2CO + 2NO -> 2CO2 + N2 |
|
|
Define: Dynamic equilibrium |
The equilibrium that exists in a closed system when the rate of the forward and reverse reactions are equal |
|
|
Le Chatelier’s Principle |
Le Chatelier’s Principle states that when a system in dynamic equilibrium is subjected to a change, the position of equilibrium will shift to minimise (oppose) the change. |
|
|
Greenhouse effect |
is the process in which the absorption and subsequent emission of infrared radiation by atmospheric gases warms the lower atmosphere and the planets surface |
|
|
Greenhouse gases |
3 most important - water vapour, CO2, methane 2 factors influencing contribution to global warming - lifetime in atmos, and ability to absorb radiation |

