![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
135 Cards in this Set
- Front
- Back
|
Benzene What are arenes? |
Aromatic hydrocarbons containing one or more benzene ring |
|
|
Benzene Describe the properties of Benzene |
- Colourless liquid - C6H6 - Has a sweet odour - Is flammable |
|
|
Benzene What was Kekulé's original model for Benzene? |
Alternating double bonds in a ring of six carbons. |
|
|
Benzene Why was this model not accepted? |
* If C=C double bonds were present, benzene would act as an alkene and decolourise bromine water, BUT it does not react at all - the approaching bromine molecule is not attracted to the C=C bond. * All the C-C bonds were found to have the same length * All internal bond angles were found to be 120° * When the enthalpy of hydrogenation of a C=C bond is measured it = -120kJmol-1, so benzene should have an enthalpy of hydrogenation of 3x(-120kJmol-1) but it does not.
|
|
|
Benzene Describe the real model for the structure of benzene |
6 carbon atoms arranged in a planar hexagonal ring Bond angles are trigonal planar - 120° around each carbon The electrons in one p-orbital of one carbon atom overlaps with electrons in other p-orbitals. This overlap produces a system of pi-bonds, in which electrons are delocalised in a ring of electron density |
|
|
Benzene Why is Benzene less reactive than alkenes? |
(It does not react with bromine water to decolourise it or react with strong acids such as HCl or react with halogens such as Cl, Br or I.) Benzene is less reactive because a) the delocalised system of benzene of benzene is very stable b) to take part in addition reactions an electron from the already stable ring would have to bond to a different atom- forming something that is less stable. This is not energetically favourable. c) It would disrupt the delocalisation of the ring structure. |
|
|
Benzene What kind of reactions does Benzene take part in?
(Be specific pls) |
Substitution - one of the hydrogens in the benzene ring is replaced by another atom/group of atoms. This does not disrupt the stability of the delocalisation of the ring. Specifically ;) the region of high electron density above and below the carbon plane attracts electrophiles - and so takes part in electrophilic substitution. |
|
|
Benzene Draw the nitration of benzene and the conditions required
|
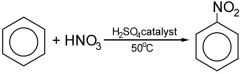
|
|
|
Benzene What is the method for the nitration of benzene? |
1) Prepare the nitrating mixture in a round-bottom flask 2) Mix concentrated HNO3 and concentrated H2SO4 carefully while cooling in a beaker of cold water 3) Add benzene carefully to the nitrating mixture - keep below 50°C 4) Once all the benzene is added, fit a reflux condenser and heat to 50°C in a water bath |
|
|
Benzene What can happen if the reaction of nitration of benzene gets hotter than 50°C? |
More than one nitro group is substituted |
|
|
Benzene Outline the nitration of methylbenzene/Toluene (HINT- it's dynamite) |
Methylbenzene can be nitrated with a mixture of concentrated nitric and sulfuric acids. The reaction is faster than the nitration of benzene, and can lead to the formation of 2,4,5,-trinitromethylbenzene (trinitrotoluene - TNT). |
|
|
Benzene What are the properties of Nitrobenzene? |
* Pale-yellow * Liquid * Important starting material for dyes, pesticides and pharmaceuticals |
|
|
Benzene Draw the mechanism for the Nitration of benzene, show the catalysts |
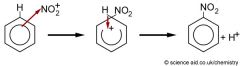
1) HNO3 + H2SO4 -> NO2 + HSO4- + H2O 3) H+ + HSO4 -> H2SO4 (= catalyst) |
|
|
Benzene How does Benzene react with halogens? |
Benzene doesn't react with halogens on their own, but requires a halogen carrier catalyst |
|
|
Benzene Give 5 examples of halogen carriers |
1) AlCl3 2) FeBr3 3) AlBr3 4) FeCl3 5) Iron metal can also be used as it can react with any halogen present to form the required iron(III) halide |
|
|
Benzene Draw the reaction between Bromine and Benzene (with a halogen carrier) |
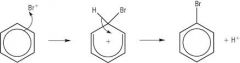
1) Br2 + FeBr3 -> Br+ + FeBr4- 3) FeBr4 + H+ -> FeBr3 + HBr (= catalyst) |
|
|
Benzene Explain the mechanism of the halogenation of benzene |
1) The electrophile accepts a pair of electrons from the delocalised ring to form a covalent bond 2) An intermediate forms where the delocalised pi-electron cloud is disrupted with the electrophile attached to the ring - this is less stable 3) As a result, Hydrogen is lost as a H+ ion. The delocalised ring reforms and stability is restored as the hobbits return to the Shire. |
|
|
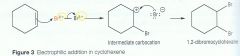
Benzene Draw the mechanism of the reaction of cyclohexene and bromine |
|
|
|
Benzene When aqueous bromine is added dropwise to an alkene, what is the colour change? |
Orange to colourless
|
|
|
Benzene What type of reaction occurs between an alkene and a halogne? |
Electrophilic addition |
|
|
Benzene Explain the mechanism for the electrophilic addition of alkenes |
1) The pi-bond in the cyclohexene contains localised electrons above and below the two carbon atoms in the double bond. This creates a region of high electron density. 2) When bromine approaches the alkene, the electrons in the pi-bond repel the electrons in the Br-Br bonds, inducing a dipole, creating a polar molecule. 3) The pi-electron pair from the double bond is attracted to the partially positive Br causing the double bond to break. 4) A new bond forms between one of the carbon atoms and a bromine atom, forming a positive carbocation. The bond between the two Bromines break by Heterolytic fission. This forms a bromide ion Br-. 5) Finally, the Br- is attracted to the intermediate carbocation forming a covalent bond. |
|
|
Benzene What happens when Bromine is added to Benzene? |
No reaction takes place - the bromine mixture remains orange. |
|
|
Benzene What happens when iron filings or iorn(III) bromide is added to a bromine/benzene mixture? |
Bromine is decolourised and white fumes of hydrogen bromide gas are observed. |
|
|
Benzene Compare the properties of Benzene and alkenes |
Alkenes * Localised pi-electrons above and below two carbon atoms * High electron density * Sufficient pi-electron densty above and below the double bond to depolarise approaching halogen molecules * Doesn't require a halogen carrier
Benzene * Delocalised pi-electrons spread overthe 6 carbon ring * Low electron density * Insufficient pi-electron density to depolarise an approaching halogen molecule * Requires a halogen carrier to create a powerful electrophile able to attract the pi-electrons from the ring |
|
|
Phenols What are phenols?
|
Organic compounds where an -OH group is directly attached to a benzene ring
|
|
|
Phenols At room temperature what state is phenol in? |
Solid state. |
|
|
Phenols Describe the solubility of phenols |
Phenols are slightly soluble in water due to the presence of the OH group, which forms hydrogen bonds with H2O, but the benzene ring makes it less soluble. |
|
|
Phenols What reactions of phenols should you know about? |
1) Sodium hydroxide 2) Sodium 3) Bromine |
|
|
Phenols Write the word equation for the reaction between phenols and Sodium Hydroxide. |
Phenol + NaOH (aq) -> Salt + H2O
Phenols are neutralised by sodium hyrdroxide to form sodium phenoxide. |
|
|
Phenols What happens when phenols are dissolved in water? |
When dissolved in water, phenols form a weak acidic solution by losing H+ ions from the OH group. |
|
|
Phenols Draw the reaction between Sodium Hyrdroxide and Phenol. |
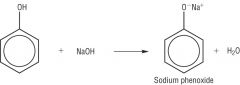
|
|
|
Phenols Write the word equation for the reaction between phenols and Sodium |
Reactive metal (sodium) + Phenol -> H2 Gas |
|
|
Phenols Draw the reaction between Sodium Hyrdroxide and Sodium |
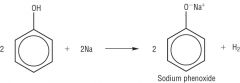
|
|
|
Phenols Draw the reaction between a halogen and phenol. |
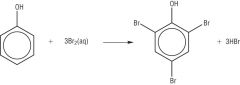
|
|
|
Phenols Why do phenols have increaed reactivity? |
Phenols have an increased reactivity because of the OH group. The p orbital lone electron pair on the oxygen atom is drawn into the ring which creates a high electron density. The ring is activated. This increased electron density is able to polarise the Bromine molecule which is then attracted more strongly to the ring than in benzene. |
|
|
Phenols What are the uses of Alkyl Phenols? |
Surfactants and detergents
|
|
|
Phenols What are the uses of Chlorophenols? |
Antiseptics and disenfectants
|
|
|
Phenols What are the uses of Salycylic Acid? |
Used in the preperation of pharmaceuticals (aspirin) |
|
|
Phenols What are the uses of Bisphenol? |
Used in the production of epoxy resins for paints. |
|
|
Carbonyls What is the functional group of a carbonyl? |
C=O |
|
|
Carbonyls What is the functional group of an aldehyde? |
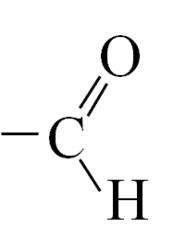
|
|
|
Carbonyls What is the functional group of a ketone? |
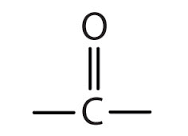
|
|
|
Carbonyls Draw the formation of the C=O bond in a carbonyl |
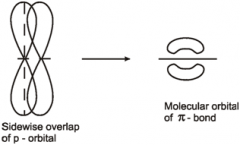
|
|
|
Carbonyls Why does the carbonyl bond not react in the same way as a double bond in an alkene? |
Because the oxygen atom is more electronegative which creates a dipole. |
|
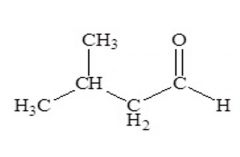
Carbonyls Name this aldehyde:
|
3-methylbutanal |
|
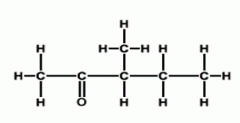
Carbonyls Name this ketone: |
3-methylpentanone |
|
|
Carbonyls
What are the uses and properties of Benzylaldehyde? |
* Colourless liquid
* Almond-like odour * Used to make almond essence - an ingredient in bakewell tarts * Gives the flavour to marzipan under the icing in Christmas cakes |
|
|
Carbonyls What are the uses and properties of Phenylethanone? |
* Used to create fragrances: cherry, honeysuckle, jasmine and strawberry |
|
|
Carbonyls What are the uses and properties of Cinnamaldehyde? |
* Makes up 80-90% of the essential oils in cinnamon bark which can treat coughing, sore throats and hoarseness. |
|
|
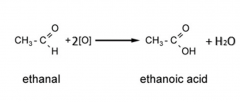
Carbonyls
Write the equation for the oxidation of a primary alcohol, including the conditions, when it is distilled immediately. |
![Primary alcohol + [O] -> Aldehyde + H2O
Conditions:
- K2Cr2O2/H2SO4
- Distil immediately](https://images.cram.com/images/upload-flashcards/22/86/51/8228651_m.jpg)
Primary alcohol + [O] -> Aldehyde + H2O
Conditions: - K2Cr2O7/H2SO4 - Distil immediately |
|
|
Carbonyls
Write the equation for the oxidation of a primary alcohol, including the conditions, when it is heated under reflux. |
Conditions:
- K2Cr2O7/H2SO4 - Heat under reflux -Observations Orange->Green |
|
|
Carbonyls Write the equation for the oxidation of a secondary alcohol. |
|
|
|
Carbonyls Write the equation for the oxidation of an aldehyde including the conditions. |

Conditions - Reflux - H+ / K2Cr2O7 |
|
|
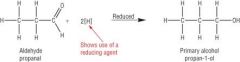
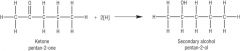
Carbonyls What are carbonyl compounds reduced to? |
Alcohols |
|
|
Carbonyls What does a carbonyl require to be reduced? |
- NaBH4 - Reducing agent - H2O as the solvent
|
|
|
Carbonyls Write the equation for the reduction of an aldehyde. |
|
|
|
Carbonyls Write the equation for the reduction of a ketone. |
|
|
|
Carbonyls What is the type of reaction that reduces aldehydes or ketones? |
Nucleophilic addition reactions. |
|
|
Carbonyls Draw the mechanism for the nucleophilic addition of the reduction of an aldehyde. |
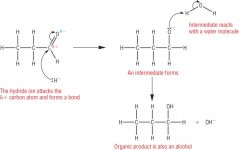
|
|
|
Carbonyls Explain the mechanism for the nucleophilic addition of carbonyls |
1) The partially positive (electron deficient) carbon atom is attacked by the Hyrdide ion (the nucleophile) which donates a lone pair of electrons, forming a covalent bond 2) At the same time the C=O bond breaks forming an intermediate negatively charged oxygen atom 3) The negatively charged intermediate donates a pair of electrons to a hydrogen on a molecule of water, forming a dative covalent bond and a hydroxide ion, OH-. |
|
|
Carbonyls How can you identify a carbonyl? |
2,4-Dinitrophenylhydrozine (2,4-DNP) in a mixture with methanol and sulfuric acid - also known as Brady's reagent. When Brady's reagent is added to a carbonyl a orange/yellow precipitate is formed. There is no precipitation with a carboxylic acid or ester. |
|
|
Carbonyls How can you differentiate between an aldehyde or a ketone? |
Tollen's reagent (ammonical silver nitrate)- a weak oxidising agent - oxidises aldehydes to carboxylic acids. Ketones are not oxidised. Upon oxidation, a silver-grey solid or 'silver mirror is formed'. |
|
|
Carbonyls How is Tollen's reagent produced? |
Aqueous sodium hydroxide is added to aqueous silver nitrate until a brown precipitate of silver oxide is formed. Dilute aqueous ammonia is then added until the precipitate dissolves. |
|
|
Carbonyls Draw the equation for the oxidation of an aldehyde using Tollen's reagent. Include the equation for silver ions. |

Ag+ (aq) + e- -> Ag (s) |
|
|
Carbonyls How can you determine what aldehyde or ketone it is? |
1) Filter the impure product fromed from the 2,4-DNP derivative and recrystallise it to produce a purified sample of yellow or orange crystals - this is filtered and allowed to dry. 3) Measure the melting point of the the purified derivative. 4) Compare the melting point to a database to identify the original aldehyde or ketone. |
|
|
Carboxylic Acids What is the functional group of a carboxylic acid? |
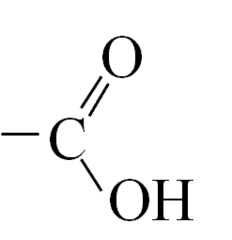
|
|
|
Carboxylic Acids Name the 7 examples of Carboxylic Acids you should know. |
1) Methanoic (Formic) Acid 2) Butanoic Acid 3) Hexanoic acid 4) Ethanoic (Acetic) Acid 5) Malic Acid 6) Oxalic Acid 7) Citric Acid |
|
|
Carboxylic Acids What is the formula for a) Methanoic (Formic) Acid b) Butanoic Acid c) Hexanoic acid d) Ethanoic (Acetic) Acid e) Malic Acid f) Oxalic Acid g) Citric Acid |
a) HCOOH b) CH3CH2CH2COOH c) CH3(CH2)6COOH d) CH3COOH e) HOOCCH2CH2COOH f) HOOCCOOH g) ----------------- |
|
|
Carboxylic Acids Where are the following found in nature? a) Methanoic (Formic) Acid b) Butanoic Acid c) Hexanoic acid d) Ethanoic (Acetic) Acid e) Malic Acid f) Oxalic Acid g) Citric Acid |
a) Ant Stings b) Rancid Butter c) Smell of goats and other farmyard animals d) Vinegar e) Apples f) Rhubarb leaves g) Citrus Fruits |
|
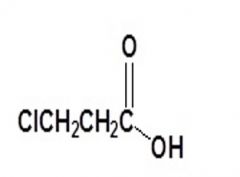
Carboxylic Acids Name this compound: |
3-chloropropanoic acid |
|
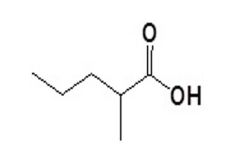
Carboxylic Acids Name this compound: |
2-methylpentanoic acid |
|
|
Carboxylic Acids Explain the solubility of Carboxylic Acids |
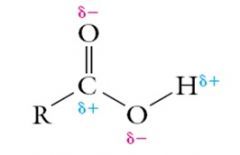
Carboxylic acids are very soluble in water due to the difference in electronegativities of Carbon and Oxygen. Hydrogen bonds form between water and the polar groups on a carboxylic acid.
|
|
|
Carboxylic Acids What reactions of Carboxylic acids should you know? |
1) Reaction with metals 2) Reaction with metal carbonates 3) Reaction with bases |
|
|
Carboxylic Acids Write the word equation for the reaction between a carboxylic acid and a metal. Include an example in your answer. |
Acid + Metal -> Salt + H2
CH3COOH + Na -> Ch3COO-Na+ + 1/2 H2 |
|
|
Carboxylic Acids Write the word equation for the reaction between a carboxylic acid and a metal carbonate. Include an example in your answer. |
Acid + Metal Carbonate -> Salt + CO2 + H2O
2 CH3COOH + MgCO3 -> (CH3COO-)2Mg2+ + CO2 + H2O |
|
|
Carboxylic Acids Write the word equation for the reaction between a carboxylic acid and a base. Include an example in your answer. |
Acid + Base -> Salt + Water
CH3COOH + NaOH -> CH3COO-Na+ + H2O |
|
|
Esters What is the functional group of an ester? |
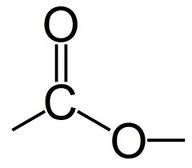
|
|
|
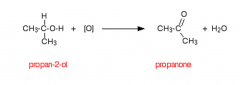
Esters How is an ester formed? |
From the reaction between a carboxylic acid and an alcohol in the presence of a metal catalyst.
Example - Propanoic acid + Ethanol -> Ethyl Propanoate + H2O |
|
|
Esters How is an ester formed from acid anhydrides? |
Esters can be prepared by gently heating an acid anhydride with an alcohol. This gives a better yeild.
Example - Ethanoic Anhydride + methanol -> Methyl Ethanoate + Ethanoic acid |
|
|
Esters What is Esterification? |
The reaction of an alcohol with a carboxylic acid to form an ester and water.
|
|
|
Esters What is an acid anhydride? |
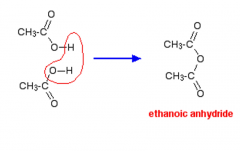
A molecule formed by the removal of a molecule of water from two carboxylic acids. |
|
|
Esters What is hydrolysis?
|
A reaction with water or hydroxide ions that breaks a chemical compound into two compounds. |
|
|
Esters Under what conditions can the hydrolysis of esters take place? |
With an aqueous acid or an aqueous alkali. |
|
|
Esters What are the conditions for the acid hydrolysis of esters? |
The ester is heated under reflux with dilute sulfuric acid or dilute hydrochloric acid |
|
|
Esters Draw the equation of the acid hydrolysis of propyl ethanoate. |

|
|
|
Esters What are the conditions for an alkaline hydrolysis of an ester? |
Aqueous sodim or potassium hydroxide is refluxed with the ester. |
|
|
Esters Write the equation for the alkaline hydrolysis of ethyl propanoate. |
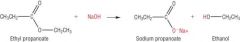
|
|
|
Esters What is saponification? |
The reaction between an ester and aqueous sodium or potassium hydroxide, that is non-reversible, to produce the sodium salt of the carboxylic acid. It is the basis of soap-making. |
|
|
Esters What are the uses of esters? |
* Many esters are found in essential oils, obtained by steam distillation of organic plant matter. Example - Benzyl ethanoate, CH3COOCH3C6H5, is found in many flowers and is the main component of essential oils from jasmine flowers. It is used for perfumery and cosmetics and apple and pear flavourings. It is found in shampoo, perfumes, fabric softner, soap, hairspray and deoderants. Example - Oil of Wintergreen: the essential oil obtained from the Wintergreen plant. Can be massaged into muscles and joints to relieve pain as 'deep heat'. The ester involved is methyl salicylate. |
|
|
Fats and Oils What are the functions of fats and oils in the body? |
- Insulation - Long term energy store |
|
|
Fats and Oils How do fats and Oils differ? |
They have different melting points.
m.t.p above room temp = fat (ex. butter) m.t.p below room temp = oil (ex. olive oil)
|
|
|
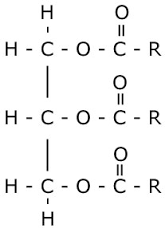
Fats and Oils What are triglycerides? |
They are triesters of an alcohol - propane-1,2,3-triol (glycerol) and three fatty acid molecules. |
|
|
Fats and Oils Draw the structure of a triglyceride |
|
|
|
Fats and Oils What are fatty acids? |
Long chain carboxylic acids. |
|
|
Fats and Oils What are unsaturated fatty acids? |
Fatty acids without any double bonds. |
|
|
Fats and Oils What are saturated fatty acids? |
Fatty acids with one or more double bond(s). |
|
|
Fats and Oils Explain how to use shorthand for the structure of fatty acids. |
1) The first number indicates the number of Carbon atoms 2) The second number indicates the number of double bonds 3) The number in the brackets indicates the position of the double bond. |
|
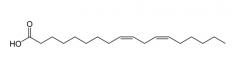
Fats and Oils What is the shorthand for the following fatty acid? |
18:2 (9,12) Octadeca-9,12-dienoic acid |
|
|
Fats and Oils Describe the structure of a triglyceride |
Triglycerides are triesters with three fatty acids joined to each of the alcohol groups on glyceral through an ester linkage. |
|
|
Triglycerides, diet and Health Why does E/Z isomerisation occur?
|
Due to the restricted rotation around the double bond. |
|
|
Triglycerides, diet and Health What is cis-trans isomerism? |
A special type of E/Z isomerism where each carbon of the C=C bond carries one atom or group that is the same. Ie. H H \ / c=c / \ R R |
|
|
Triglycerides, diet and Health Draw E/Z and Cis/Trans |
Z/Cis = Ze zame zide, Same side E/Trans = Enemies, different sides |
|
|
Triglycerides, diet and Health In nature, what form do unsaturated fatty acids usually exist in? |
The cis form. |
|
|
Triglycerides, diet and Health How does the cis form affect the melting point of unsaturated fatty acids? |
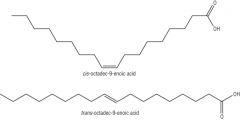
In cis form, the fatty acids cannot pack closley together, so they exist at liquids at room temperature. In trans form they can pack closely: it has a linear sructure. Therefore, it has a higher m.t.p.
|
|
|
Triglycerides, diet and Health Why are double bonds removed in the food industry? |
To make the fats and oils more solid. |
|
|
Triglycerides, diet and Health How are double bonds removed in the food industry? |
By partial hydrogenation. |
|
|
Triglycerides, diet and Health What are the side effects of removing double bonds? |
The remaining double bonds may structurally be changed, so are in the more unhealthy trans form. |
|
|
Triglycerides, diet and Health What are lipoproteins? |
Particles that carry lipids, such as cholesterols and fats, through the blood. |
|
|
Triglycerides, diet and Health What are LDLs? |
Low-density Lipoproteins Responsible for carrying cholesterol and triglycerides from the liver to the tisses. They can deposit lipids onto artery walls, building up fatty acid deposits that restrict blood flow |
|
|
Triglycerides, diet and Health What are HDLs? |
High-density Lipoproteins Believed to remove cholesterol from the arterioles and transport it back to the liver for excretion. |
|
|
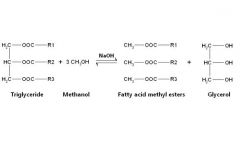
Triglycerides, diet and Health What is the chemical structure of biodiesel? |
An ethyl or methyl ester of a fatty acid. |
|
|
Triglycerides, diet and Health By what process is biodiesel made? |
Transesterification. Triglycerides in fats and oils are reacted with methanol or ethanol in the presence of a sodium or potassium hydroxide catalyst. Glycerol is formed also, which can be sold to pharmaceutical companies (=yay! atom economy) |
|
|
Triglycerides, diet and Health Write the equation of transesterification for the production of biodiesel. |
|
|
|
Amines What are amines? |
Ammonia derivatives. |
|
|
Amines What is a primary amine, a secondary and tertiary amine? |
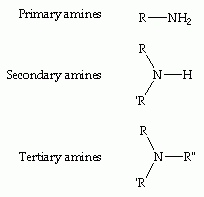
Primary - One H atom replaced Secondary - Two H atoms replaced Tertiary - Three H atoms replaced |
|
|
Amines What are the functions of a) Amphetamine b) Phenylephrine c) Adrenaline |
a) A complex amine used to treat daytime drowsiness and chronic fatigue syndrome b) Commonly userd as a decongestant. Found in many cold and flu remedies with paracetemol c) The 'fight or flight' amine that helps the body to deal with sudden stress. |
|
|
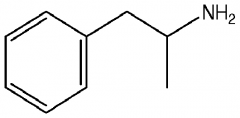
Amines What is the structure of Amphetamine? |
|
|
|
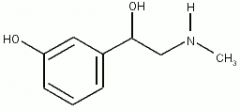
Amines What is the structure of Phenylephrine? |
|
|
|
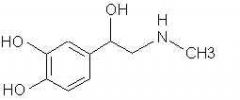
Amines What is the structure of Adrenaline? |
|
|
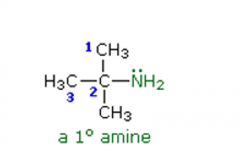
Amines Name the following amine: |
2-methyl-2-propanamine |
|
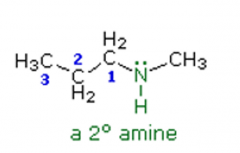
Amines Name the following amine: |
N-methylpropanamine |
|
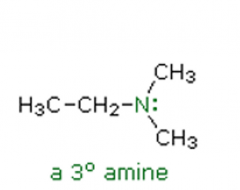
Amines Name the following amine: |
N,N-dimethylethanamine |
|
|
Amines What is a base? |
A proton acceptor. |
|
|
Amines What makes amines weak bases? |
The lone pair of electrons on the Nitrogen atom. The fact that it can accept a H+ |
|
|
Amines What happens when an amine accepts a proton? |
A dative covalent bond forms between the lone pair of electrons of the Nitrogen atom and the proton. |
|
|
Amines Draw the reaction between methylamine and a proton. |
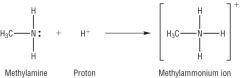
|
|
|
Amines Base + Acid = ? |
Salt |
|
|
Amines Draw the reaction of phenylamine and nitric acid |
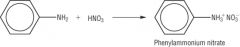
|
|
|
Amines How are aliphatic amines prepared? |
By warming halogenoalkanes gently with an excess of ammonia, with ethanol as a solvent. |
|
|
Amines Write the reaction for the formation of propylamine and the subsequent reactions that follows. Describe what happens in each reaction. |
1) CH3CH2CH2Cl + NH3 -> CH3CH2CH2NH2 + HCl Ammonia reacts with 1-chloropropane by nucleophilic substitution. Ammonia has a lone pair of electrons that attacks the partially positive carbon atom in the C-Cl bond.
2) NH3 + HCl -> NH4+Cl- Further ammonia then reacts with the HCl formed.
3) CH3CH2CH2Cl + CH3CH2CH2NH2 -> (CH3CH2CH2)2NH + HCl The product of the first reaction - propylamine, also has a lone pair of electrons which attacks another molecule of 1-chloropropane, causing further stubstitution.
THE EXCESS AMMONIA IS USED TO MINIMISE THE FURTHER SUBSTITUTION.
|
|
|
Amines How do you prepare an aromatic amine? |
By reducing nitrobenzen or other nitroarenes using a mixture of tin and concentrated hydrochloric acd, heated under reflux, followed by neutralisation of the excess HCl. |
|
|
Amines Draw the reduction of nitrobenzene. |
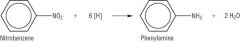
|
|
|
Amines What are the two steps in the industrial preperation of dyestuffs? |
1) Diazotisation 2) Coupling reactions |
|
|
Amines Describe the first step in the preperation of azo dyes. |

Diazotisation - forms the diazoniun ion When a mixture of phenylamine and nitrous acid is kept below 10°C, a diazonium salt is formed.
1) HNO2 (nitrous acid) is generated by the following reaction: NaNO2 + HCl -> HNO2 + NaCl
2) The cold nitrous acid then reacts with an aromatic amine to form a diazonium salt.
|
|
|
Amines Describe the second step in the preperation of azo dyes. |
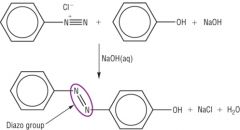
Coupling A coupling reaction occurs when the diazonium salt, benzenediazonium chloride, is reacted with a phenol/aromatic compound under alkaline conditions. In the reaction, two benzene rings are linked through an azo functional group -N=N- The prodct is a brightly coloured compound used as an azo dye.
|

