![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
264 Cards in this Set
- Front
- Back
|
What are TAGs and why are they good at their job?
|
Long term storage of lipid in the body - anhydrous so efficient
|
|
|
How are Fatty Acids synthesised and what stimulates the process?
|
Insulin in fed state stimulates fatty acid synthesis:
Oxaloacetate and Acetyl CoA form citrate in mitochondria Citrate passes into cytosol and dissociates Acetyl CoA and bicarbonate form Malonyl CoA (rate limiting) Malonyl CoA then binds Acetyl CoA giving off CO2 NADPH required for fatty acid synthesis Fatty acid synthetase enzyme also involved |
|
|
Do newly formed fatty acids pass back into mitochondria for oxidation?
|
No. Malonyl CoA inhibits Carnitine Transferase, which transports FAs to mitochondria
|
|
|
Why is the Hexose Monophosphate shunt important?
|
It induces Glucose 6-phosphate dehydrogenase raising supply of NADP
|
|
|
What is fatty acid synthetase?
|
Complex enzyme with mutiple active sites
|
|
|
How many carbons in a typical fatty acid chain?
|
16 carbons - palmitate
|
|
|
How are TAGs formed from fatty acids?
|
3 FAs bind with a glycerol phosphate in liver
|
|
|
How are TAGs transported to tissues?
|
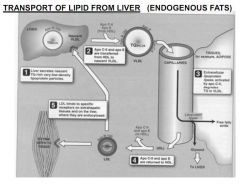
In VLDLs, which TAG/cholesterol esters in core, and apoproteins, cholesterol and phospholipids on surface
|
|
|
What are the different types of lipoproteins?
|
Chylomicrons, biggest - lots of dietary TAGs
VLDLs - 60% endogenous TAG IDLs - 30% endogenous TAG LDLs - 5% endogenous TAG (liver to tissues) HDLs - 5% endogenous TAG (tissue to liver) |
|
|
How is exogenous (dietary) fat transported to liver?
|
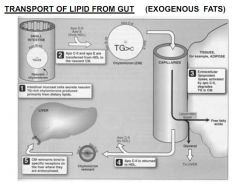
Intestinal chylomicrons express APO-B48
Gain APO-CII and E from HDLs Insulin acts on APO-CII to activate Lipoprotein Lipase Lipoprotein Lipase breaks down TAGs and FFAs go to adipose sites APO-CIIgoes back to HDL Remnant expressing APO-B48 and E back to liver for endocytosis |
|
|
How is endogenous fat transported from liver to tissues?
|
Liver secretes VLDL expressing APO-B100
HDL donates APO-CII and E Insulin acts on APO-CII to activate Lipoprotein Lipase Lipoprotein Lipase breaks down TAGs and FFAs go to adipose sites IDLs exist until APO-CII and E returned to HDL Remaining LDL expresses APO-B100, 50% go to tissues, 50% back to liver |
|
|
How do HDLs transport cholesterol back to liver?
|
HDL from diet and liver express APO-A1, E and C
APO-A1 activates PCAT, Phosphatidylcholine Acyltrasferase PCAT esterises free cholesterol by transferring an acyl chain CholesterolEster passes into HDL Swollen HDL expressing APO-A back to liver |
|
|
How is cholesterol brought in the cell?
|
Cell expresses LDL receptor on apical surface (APO-B100)
LDL expressing APO-B100 binds and contents released into cell by endocytosis Cholesterol passes to nucleus which generates negative feedback Cell stops expressing LDL receptors, and stops synthesising enzyme HMG-CoA |
|
|
How is cholesterol synthesised?
|
Acetyl CoA and Acetoacyl CoA become HMG CoA
HMG CoA becomes Mevalonate due to HMG-CoA reductase Mevalonate becomes cholesterol |
|
|
How do statins work?
|
Inhibit HMG-CoA reductase
|
|
|
What consequences might you expect from a defective LDL receptor?
|
High circulating LDLs in blood, hypercholesterolaemia
|
|
|
What consequences might you expect is Lipoprotein Lipase is deficient?
|
High chylomicrons and VLDL
|
|
|
What consequences might you expect from an APO-CII deficiency?
|
High chylomicrons and VLDL
|
|
|
What consequences might you expect from an apoprotein deficiency?
|
High chylomicrons and VLDL
|
|
|
What might cause secondary hyperlipoproteinaemias?
|
Obesity, diabetes type 2, high cholesterol, alcoholism
|
|
|
What is lipoprotein A?
|
LDL with apoprotein A
Associated with CHD risk increase Levels of Lipoprotein A are genetic Oestrogen brings levels down Trans fats bring levels up Competes with plasmiogen, slowing breakdown of clots, risk of thrombus formation |
|
|
Why might high circulating LDL affect atherosclerosis?
|
Macrophages fill with cholesterol from LDLs, form a fatty streak
This causes endothelial damage, formation of plaques, inflammation etc |
|
|
Scenario 11 Lecture 8
|
Minerals
|
|
|
What are the dietary requirements for Zinc, Iron and Copper?
|
Zinc: 10mg/day, Iron: Men and post-menopause women 1mg/day, young women 2mg/day, pregnant women 3mg/day, Copper 1-2mg/day
|
|
|
Why are minerals important?
|
Copper and Iron metalloproteins carry O2, and protect against oxidation
|
|
|
What are superoxide dismutases and what do they do?
|
Break down free radicals like hydrogen peroxide into water and O2. Example is Glutathione Peroxidase
|
|
|
How much zinc do we have and where is it found?
|
2-3g stored in all tissues but high in liver, kidney, bone, retina, muscles and prostate
|
|
|
What does zinc do?
|
Vital component in 600+ enzymes
|
|
|
What does a zinc deficiency cause?
|
Growth retardation, hypogonadism, slow wound healing - Acrodermatitis Enteropathica (cured by Zinc therapy)
|
|
|
List five functions of iron in the body?
|
Oxidative energy production, oxygen transport (haemoglobin/myoglobin), mitochondrial respiration, inactivating O2 radicals, DNA synthesis
|
|
|
How much iron do we have and where is it stored?
|
Store 3-5mg in body, 67% in haemoglobin, 27% in ferritin and haemosidirin, 3.5% myoglobin
|
|
|
How does the body lose iron?
|
1mg per day, lost through urine, sweat, sloughing of mucosal cells in GI tract, bile, GI bleeding
|
|
|
Can the body remove excess iron?
|
No, there is no physiological mechanism to remove iron in the body
|
|
|
How can excess iron be absorbed?
|
Not usually through cooking - but often in drinks. Drinks brewed in iron casks cause iron excess as they usually contain vitamin C/fructose which aid iron uptake
|
|
|
What is the Hydroxyl Radical?
|
Very damaging free radical OH
Created when reduced ferrous Fe2+ iron is oxidised to ferric Fe3+ in the presence of H2O2 |
|
|
What is ferritin and how is iron taken up by it?
|
Hollow shell which can store 4300 units of FE3+
Soluble Iron taken up as Fe2+, then oxidised to Fe3+ |
|
|
Why is ferritin formed?
|
Presence of iron stimulates synthesis of ferritin
|
|
|
What happens to ferritin in iron excess?
|
Mucosal cells gain ferritin deposits.
The plasma levels of ferritin increase. |
|
|
What is haemosidirin?
|
Water-insoluble
Derived from ferritin Found in spleen, bone marrow and liver |
|
|
What is trasferrin?
|
Bilobal protein which carries 2 Fe3+ molecules
Transports iron in body from absorption sites (GI) and sites of breakdown (reticuloendothelial system) to sites of iron storage (liver) or utilisation (bone marrow) |
|
|
Does [transferrin] increase in iron deficiency?
|
Yes, iron deficiency leads to increased [transferrin]
|
|
|
Does [transferrin] increase in iron overload?
|
No, iron overload causes a decrease in [transferrin]
|
|
|
How is iron taken up into cells?
|
Iron-loaded transferrin binds to transferrin receptor on cell
Whole lot (transferrin & receptor) are endocytosed and coated in clathrin Loss of clathrin coat lowers intra-vesicular pH to 5.5 At low pH, iron dissociates from transferrin Transported into cell as Fe2+ for usage or storage (in ferritin) Vesicle, with receptor-bound transferrin return to membrane |
|
|
What are disorders of iron deficiency?
|
Anaemia
|
|
|
What causes anaemia?
|
Bleeding, mensturation, pregnancy, lack of dietary iron
|
|
|
What is haemochromatosis?
|
Genetic defect in iron uptake, transport or metabolism causing excess iron
|
|
|
Other than haemochromatosis, what other causes are there of iron overload?
|
Transfusion, dietary overload, atransferrinaemia (absence of transferrin), aceruloplasminaemia (absence of enzyme which oxidises Fe2+ to Fe3+)
|
|
|
Classify genetic haemochromatosis
|
Type 1 mutation in HFE
Very common in Northern Europe Causes uptake of 3-4mg/day Massive build up over years Causes liver failure, diabetes, cardiac failure, ash grey skin |
|
|
How would you test for type 1 haemochromatosis?
|
Measure transferring sats (>50% indicator for condition)
Measure serum ferritin (>300ug/l indicator) Genetic test for mutation Liver biopsy |
|
|
How would you treat type 1 haemochromatosis?
|
Phlebotomy - weekly bleed of 500ml shown to reduce iron by 250mg each time
|
|
|
What other types of haemochromatosis are there?
|
Type 2a, 2b, 3 and 4 all very rare, type 1 is main
|
|
|
How might someone acquire secondary haemochromatosis?
|
Transfusions - can cause deposit of excess iron in heart - CHD, liver failure
|
|
|
How might you treat secondary haemochromatosis?
|
Chelation therapy - drugs which bind iron to excrete from the body
|
|
|
What makes a good chelation agent?
|
High affinity for iron
Non-toxic Efficient at iron removal Active orally Most popular is Deferixamine, but this is not orally actiuve, given in injection |
|
|
How is iron absorbed in the gut?
|
Haem-bound iron taken up by HCP-1
Non-haem bound iron converted from Fe3+ to Fe2+ by Duodenal Cytochrome B (DCytB) Non-haem bound Fe2+ taken up by DMT-1 In the cell IRP (Iron Regulatory Protein) controlls synthesis of ferrtin/transferrin receptors Ferroprotein-1 transports excess iron out of cell via Hephaestin Hephaestin is a membrane bound ferridoxase which oxidises Fe2+ to Fe3+ Haem-Oxygenase breaks down haem bound iron Ferritin provides intra-cellular iron pool |
|
|
How much copper do we store and where?
|
We sotre 75-100mg of copper in liver, brain, heart and kidneys
|
|
|
How much copper do we need each day?
|
1-2mg
|
|
|
What is copper required for?
|
Key enzymes include ferroxidase for iron oxidation, collagen cross-linking enzymes, and production of melanin and catecholamines
|
|
|
Copper deficiency causes what disorder?
|
Menke's disease - depigmentation of hair, X-linked, dead in 2 years
Copper treatment ineffective |
|
|
Copper overload causes what disorder?
|
Wilson's disease - autosomal recessive, impaired Cu excretion
Causes build up in liver and brain - liver failure, mental retardation Key sign = Kayser-Fleischer (copper ring round cornea) Treat with copper chelation therapy |
|
|
Scenario 11 Lectures 9 and 10
|
Metabolism in the fed and fasting state
|
|
|
When would the body need to store/mobilise energy?
|
When it is available and is not needed immediately it would store, and when unavailable it can be mobilised
|
|
|
How does the body change metabolic pattern?
|
Vary the amount of substrate available e.g. FA use in starvation
Allosteric effects on enzymes e.g. AMP appearing when ATP is low Covalent modifications e.g. phosphorylation of glycogen Changes in enzyme synthesis e.g. Glucokinase in dietary CHO |
|
|
Name the hormones controlling metabolism
|
Insulin - hypoglycaemic, Glucagon - hyperglycaemic, Adrenalin (short term), Cortisol (long term) and Growth Hormone - counter insulin
|
|
|
Where does insulin and glucagon come from, and which cells exactly?
|
Islets of Langerhan in the pancreas
B-cells secrete insulin Alpha-cells secrete glucagon |
|
|
What stimulates and inhibits insulin release?
|
Rise in blood glucose
Rise in [amino acid] in blood Secretin released after food intake Glucagon also stimulates insulin for fine tuning Adrenalin inhibits insulin release |
|
|
How is insulin secreted?
|
Glucose and amino acids metabolised in B-cells to ATP
ATP closes K+ channel Closed K+ channel changes membrane polarity Ca2+ channel opens Raised intracellular Ca2+ causes proinsulin release |
|
|
How is proinsulin processed into insulin?
|
Proinsulin is cleaved to insulin and C-peptide
|
|
|
How would you measure self-produced insulin in a diabetic on insulin therapy?
|
C-peptide is a good measure of self-produced insulin
|
|
|
What stimulates and inhibits glucagon release?
|
Low blood glucose
Rise in [amino acid] in blood - prevents hypoglycaemia after protein meal Adrenalin stimulates glucagon release to raise blood glucose |
|
|
What metabolic effects does insulin have?
|
Storage of glucose after eating
Promotes growth Stimulates glycogen synthesis and storage Stimulate fatty acid synthesis Stimulates AA uptake and protein synthesis |
|
|
Which receptors mediate the metabolic effects of insulin?
|
Via binding to tyrosine kinase receptor, insulin stimulation of Glut 4 receptors allows muscles to take up glucose when levels are high. Brain, RBCs have Glut 1 glucose receptors which are expressed independently of insulin
|
|
|
How is the glucose supply to the brain, blood cells and pancreas protected?
|
Receptors are not insulin dependent
|
|
|
What 6 effects does glucagon have?
|
Mobilises fuel
Maintains blood glucose when fasting Activates glyconeogenesis (creation of glycogen) Activates gluconeogenesis (creation of glucose) Activates FA release from adipose tissues Activates FA oxidation and ketone body formation in liver |
|
|
How does adrenalin act?
|
Mobilises fuel during stress, stimulating glyconeogenesis and fatty acid release
|
|
|
How does cortisol act?
|
Provides long term requirements
|
|
|
What happens to levels of blood glucose, insulin and glucagon after a meal?
|
Insulin and glucose go up, glucagon goes down
|
|
|
Summarise carbohydrate metabolism in the fed state
|
Liver always engaged in gluconeogenesis except when fed
In fed state, glycogenesis activated by Glucokinase Glycogen Synthase activated and Phosphorylase inhibited Glycolysis activated by PFK and pyruvate kinase Gluconeogenesis is inhibited |
|
|
Summarise metabolism of fat by the liver in the fed state
|
Acetyl CoA Carboxylase activated to convert Acetyl CoA to Malonyl CoA
Malonyl CoA inhibits Carnitine Transferase |
|
|
How does glucose feed brain and red blood cells?
|
Transported in via Glut-1 - independent of insulin
|
|
|
Summarise glucose transport into muscle in the fed state
|
Raised blood glucose stimulates increased expression of Glut-4 transporters
Glycogen Synthetase activated, phophorylase inhibited Amino acid uptake increased |
|
|
Summarise glucose uptake in adipose tissue in the fed state
|
Lipoprotein lipase activated by insulin
Allows uptake of FFA for esterification and storage of TAGs Hormone sensitive lipase inhibited so TAG is not degraded |
|
|
Where is fuel stored in man?
|
85% TAG. 14% protein, <1% glycogen and glucose
|
|
|
What happens in the fasting state to insulin and glucagon?
|
Insulin and glucose falls, glucagon rises
|
|
|
What is a typical blood glucose concentration maintained by the liver?
|
4mM
|
|
|
Hormone sensitive lipase is activated by what?
|
Adrenalin and glucagon
|
|
|
When is gluconeogenesis used for glucose?
|
After 24 hours fasting, gluconeogenesis supplies all body energy
|
|
|
Is the break down of pyruvate to acetyl CoA reversible?
|
No, the reaction catalysed by Pyruvate Dehydrogenase which breaks down pyruvate to Acetyl CoA is irreversible
|
|
|
What stimulates pyruvate dehydrogenase PDH?
|
Insulin activates PDH, glucagon inhibits it - this means that in fasting, all substrate is channelled to glucose production and not FA production
|
|
|
How are ketone bodies formed in fasting state?
|
FA oxidation leads to high [Acetyl CoA]
This exceeds TCA cycle capacity, leading to Ketone body formation |
|
|
What substrate can the erythrocyte and brain oxidise?
|
The brain oxidises both glucose and KBs, erythrocytes can only oxidise glucose
|
|
|
How much body protein can be lost before serious consequences are incurred?
|
1/3rd
|
|
|
How long does it take for ketone bodies to rise?
|
After 2 days they exceed fatty acid concentration
|
|
|
How does urea excretion differ in fed and fasting state?
|
Initially rises, then falls as ketone body production slows proteolysis
|
|
|
How do ketone bodies work in fasting state?
|
Act on pancreas B-cells to stimulate insulin release, limiting proteolysis, limiting breakdown of adipose tissue and conserving muscle
|
|
|
How long can you survive without food, and how would you die?
|
Depends on adipose stores, but typically 40-45 days, death usually by infection due to impaired immunity
|
|
|
How prevalent is diabetes?
|
2-3% of UK population, 90% of all endocrine disorders
|
|
|
What is the difference between type 1 and type 2?
|
Type 1 is insulin dependent, the body makes no insulin
Type 2 is non-insulin dependent, the body makes insulin but is resistant to it's effects |
|
|
Summarise type 1 diabetes cause and symptoms
|
Autoimmune destruction of B-cells
Early onset Polyuria, polydipsia, polyphagia, fatigue, muscle wasting, weakness |
|
|
What are the hallmarks of type 1 and how is it treated
|
Hyperglycaemia and ketoacidosis. Treatment with insulin.
|
|
|
Summarise type 2 diabetes cause and symptoms
|
Insulin resistance
Later onset Associated with diet and lifestyle |
|
|
What are the hallmarks of type 2 and how is it treated
|
Hyperglycaemia but no ketoacidosis. Treatment with diet modifications and oral hypoglycaemic agents
|
|
|
What is the metabolic pattern in diabetes?
|
Resembles starvation, but more exaggerated effects:
In starvation insulin low, in diabetes absent In diabetes glucagon acts unopposed KB's normally stimulate insulin release to protect protein, not in diabetes |
|
|
What are the chronic complications of diabetes?
|
Microangiopathy, retinopathy, neuropathy, nephropathy
|
|
|
How do type 2 diabetes treatments help condition?
|
Biguanides increase number of Glut-4 receptors, to lower circulating insulin
Sulphonylureas stimulate B-cells to increase insulin secretion |
|
|
What is the WHO classification of a metabolic disorder?
|
High fasting glucose/Insulin Resistance/Type 2 Diabetes plus 2 from: Hypertension, Dysplipidaemia (high TAG low HDL), Obesity, Microalbuminuria
|
|
|
What is the WHO classification of a metabolic disorder?
|
High fasting glucose/Insulin Resistance/Type 2 Diabetes plus 2 from: Hypertension, Dysplipidaemia (high TAG low HDL), Obesity, Microalbuminuria
|
|
|
Typical new born fat content
|
14%
|
|
|
Typical adult male fat content
|
15%
|
|
|
Typical adult female fat content
|
28%
|
|
|
Typical 10 year old girl fat content
|
19%
|
|
|
How can lean body mass be calculated?
|
Methyl histidine:creatinine ratio
Skin fold measurements Air Displacement Plethysmography (Bod Pod) |
|
|
How much energy in a gram of each food type?
|
Carbohydrate 4kcal/g
Fat 9.2kcal/g Protein 5.4kcal/g Alcohol 7kcal/g |
|
|
How much ingested energy is available for work?
|
Less than 50% (remainder is either not
absorbed or used for heat generation |
|
|
What are the basal metabolic demands for a one year old, a pregnant mother, and in lactation?
one year old compared to an adult? |
One year old: Double
Lactation: Extra 500kcal/day Pregnancy: Only 3rd trimester (extra 200kcal/day) |
|
|
How is energy requirement estimated?
|
Energy requirement proportional to
oxygen consumption 1L = 5kcal |
|
|
Which area of the brain controls appetite?
|
Hypothalamus
|
|
|
Where is the satiety centre?
|
Ventromedial nucleus
|
|
|
Where is the hunger centre?
|
Lateral hypothalamic area
|
|
|
How is appetite stimulated in the short term?
|
Low blood glucose stimulates hunger centre
Ghrelin produced in stomach |
|
|
How is satiety stimulated?
|
Distension of the GI tract stimulates vagal afferents
CCK, somatostatin, glucagon-like peptide stimulate satiety centre POMC released when stomach is full |
|
|
How is satiety stimulated long term?
|
Raised Leptin signals high state of fat stores
Raised Insulin signals high state of COH stores |
|
|
How is hunger stimulated long term?
|
Main hunger stimulator is Neuropeptide-Y
When insulin and leptin is low, hunger is stimulated |
|
|
Summarise hunger hormones
|
Neuropeptide-Y
Ghrelin |
|
|
Summarise satiety hormones
|
CCK
POMC Somatostatin Glucagon-like peptide |
|
|
What signals state of fat stores?
|
Leptin
|
|
|
What signals state of carbohydrate stores?
|
Insulin
|
|
|
What are the BMI ranges?
|
Normal 18.5 24.9
Overweight 25 - 29.9 Obese 30+ |
|
|
How big is the obesity epidemic?
|
2/3rds of UK adults overweight
1/4 of those are obese Rate has tripled in 20 years |
|
|
What are the main causes?
|
Energy intake has remained constant when energy
expenditure has gone down by 65% Socioeconomic |
|
|
Name 2 hormonal causes of obesity
|
Hypothyroidism (reduced BMR)
Hyperadrenalism (raised cortisol - antagonises insulin and stimulates release of Neuropeptide-Y |
|
|
Is type 2 diabetes a cause or a symptom of obesity
|
Symptom, type 2 is caused by obesity
|
|
|
Name 5 risk factors for obesity
|
Low level education
Chronic disease (lack of exercise) Alcohol consumption Marriage Giving up smoking |
|
|
Obesity raises the risk of what conditions?
|
CVD - MI (x2), Angina (x2.5), Stroke (x3)
Venous Thrombosis (x1.5), Type 2 (x3) Hypertension (x3), Gall bladder (x2), Arthritis (x12) |
|
|
Why do dieters regain weight?
|
Starvation reduces BMR, so after dieting you should eat less
|
|
|
Best diet?
|
Protein high (as induces satiety quicker), include
fruit, vegetables and wholegrain cereals |
|
|
Medicine for obesity?
|
Orlistat - inhibits pancreatic enzymes which reduce
fat, result is higher fat excretion |
|
|
Why might leptin therapy not work?
|
Obese patients tend to demonstrate leptin resistance
|
|
|
What surgical interventions might be considered?
|
Liposuction to remove adipose tissue
Stomach banding/stapling but only with >BMI |
|
|
What complications to gastric banding?
|
Vomiting/nausea after meals
High risk in anaesthetic for obese patients Higher risk of blood clots |
|
|
Why might a band be unsuccessful?
|
Possible to drink high calories (alcohol/milkshakes)
which the band would not affect |
|
|
Scenario 11 Lecture 3
|
Macronutrients and malnutrition
|
|
|
What is the average fat intake in UK per day?
|
88g, 40% of dietary energy intake
|
|
|
What are essential fatty acids?
|
Not synthesised in body, precursors of eicosanoids
(prostglandins, prostacyclins and thromboxanes) |
|
|
What is linoleic acid?
|
Omega-6
|
|
|
What is linolenic acid?
|
Omega-3
|
|
|
What is the RDA for essential fatty acid?
|
2-5g (UK consumption 8-15g)
|
|
|
Why is CVD important?
|
250,000 deaths per year, 1.5m have CVD
40% of all deaths are from CVD |
|
|
What are the risk factors for CVD?
|
Smoking, alcohol, sedentary lifestyle, hypertension,
high serum cholesterol, obesity, diabetes, trans fats |
|
|
How do different fats affect CVD risk?
|
Saturated fat increases serum LDL and cholesterol
Trans fats reduce HDL PUFA can reduce cholesterol |
|
|
How much energy do we derive from carbohydrates?
|
40% in affluent society, 80%+ in poor communities
|
|
|
What are cardohydrates?
|
Starch - rice, potatoes, grains
Non-starch Carbohydrates - fibre (inositol) Sugars - Sucrose, Glucose |
|
|
Is carbohydrate essential in diet?
|
No, energy could come from protein but CHO useful
|
|
|
What effect might a low carb diet have?
|
Cause metabolism of fat/protein - ketosis (pear drop urine)
|
|
|
Name 4 monosaccharides
|
Glucose, fructose, inositol (fibre) and sorbitol (commercial)
|
|
|
Inositol or phytic acid affects the absorption of which minerals?
|
Iron and calcium
|
|
|
Name 2 disaccharides
|
Sucrose (one glucose, one fructose)
Lactose (one glucose, one galactose) |
|
|
Name 2 polysaccharides
|
Starch, NSP (fibre)
|
|
|
What is the RDI for protein
|
0.75g/kg body weight for adults
Maximum 1.5g/kg of body weight |
|
|
Why is protein important in the diet?
|
Essential amino acids
|
|
|
What makes quality protein?
|
Animal better than plant
Better quality means more used, less wasted Low quality proteins lack some amino acids |
|
|
What is the protein requirement for a newborn?
|
2.4g/kg body weight (3x adult)
|
|
|
What is the protein requirement for a one year old?
|
1.4g/kg of body weight (around 2x adult)
|
|
|
Is high protein intake bad for you?
|
Yes, excessive intake can affect bone mineralisation and kidney function
|
|
|
What is marasmus?
|
Energy efficiency of all nutrients (starvation)
|
|
|
What is kwashiorkor?
|
Protein wasting, lack of nutrients, with oedema
|
|
|
Classify marasmus
|
<60% of expected weight for age, without oedema
|
|
|
Classify kwashiorkor
|
60-80% of expected weight for age, with oedema
|
|
|
Classify marasmic kwashiorkor
|
<60% of expected weight for age, with oedema
|
|
|
What symptoms might you find in marasmus
|
Extreme emaciation, impaired immunity, loss of mucosa in intestine, diarrhoea, apathy
|
|
|
What symptoms might you find in kwashiorkor
|
As with marasmus, but severe oedema, liver enlargement - distended abdomen, dermatitis, mental retardation
|
|
|
What BMI would you expect in protein energy malnutrition?
|
<16 = severe PEM
18.5 - 25 + Desirable |
|
|
How do you treat PEM?
|
Oral rehydration first - no foods!
Build up nutrients and allow abdominal recovery |
|
|
Scenario 11 Lecture 5 and 6
|
Vitamins
|
|
|
What is a vitamin?
|
Complex organic substance required in small amounts, and deficiency induces disease
|
|
|
Which vitamins are water soluble?
|
Vitamins B and C
|
|
|
Which vitamins are fat soluble?
|
Vitamins A, D, E, K
|
|
|
What features are found in water soluble vitamins?
|
Not stored, so required daily in diet, not toxic in excess
|
|
|
What features are found in fat soluble vitamins?
|
Stored, not required daily, toxic in excess
|
|
|
B-vitamins do what?
|
Co-enzymes in metabolic pathways
|
|
|
What is B1 and what does deficiency cause?
|
Thiamin - Beri beri
|
|
|
What does B1 do?
|
As Thiamin Pyrophosphate (TPP) coenzyme in Pyruvate to Acetyl CoA conversion
|
|
|
What are good sources of B1?
|
Grain, pork, poultry, fish, vegetables, dairy
|
|
|
What are bad sources of B1?
|
Polished rice, sugar, fat, processed foods
|
|
|
What reduces absorption of B1?
|
Carbohydrates (alcohol), coffee, tea, raw fish
|
|
|
What does deficiency of B1 cause?
|
Build up of lactate in muscles (not breaking down pyruvate)
|
|
|
What is wet beri beri?
|
Acute onset, cardiac failure, enlarged heart and liver
|
|
|
What is dry beri beri?
|
Chronic onset, symmetrical ascending peripheral neuropathy, weak, numb, ataxic gait
|
|
|
What is Wernicke's encephalopathy?
|
Reversible neuropathy induced by B1 deficiency
Causes confusion, ataxia, disorientation |
|
|
What is Korsakoff's psychosis?
|
Irreversible dementia common in alcoholics, alcohol inhibits uptake of B1 and inhibits enzyme which converts B1 to its active form TPP
|
|
|
What is vitamin B2?
|
Riboflavin, as FAD and FMN in redox reactions
|
|
|
Is vitamin B2 sensitive to light?
|
Yes, B2 is UV sensitive
|
|
|
What is a good source of B2?
|
Protein
|
|
|
What does a B2 deficiency cause?
|
Cracked lips (cheilosis), angular stomatitis, cataracts
|
|
|
What is vitamin B3?
|
Niacin: Nicotinic Acid and Nicotinamide are vitamers
|
|
|
What does vitamin B3 do?
|
As NAD and NADP in redox reactions
|
|
|
What are good sources of B3?
|
Cereals - protein (body can synthesise B3 from protein)
|
|
|
What does B3 deficiency cause?
|
Pellagra - 4 D's: Dermatitis (Casal's necklace), Dementia, Diarrhoea, Death
|
|
|
How does Casal's necklace develop?
|
NAD required in DNA synthesis, B3 deficiency causes poor DNA repair in UV exposed skin
|
|
|
What is vitamin B6?
|
Pyridoxine: Pyridoxamine or pyridoxal
|
|
|
What does B6 do?
|
Active form is Pyridoxal Phosphate (PP) essential in transaminations and deaminations of amino acids and haem synthesis
|
|
|
What causes B6 deficiency and what are symptoms?
|
Commonly secondary, TB drug Isonazid combines antagonises PP
|
|
|
What are the therapeutic uses of B6?
|
Treatment of seizures, Down's syndrome, autism
|
|
|
Is B6 toxic?
|
Women taking B6 for PMS show neuropathy after 1-3 years
|
|
|
What is vitamin B9?
|
Folate, active form Tetrahydrofolate (THF)
|
|
|
What does B9 do?
|
Carrier of free carbon units
|
|
|
What is vitamin B12?
|
Cobalamin - carries methyl groups
|
|
|
What are dietary sources of B12?
|
Animal tissues only, so can be deficient in vegan diet
|
|
|
How is B12 absorbed?
|
Binds to intrinsic factor secreted by gastric G-cells, absorbed in terminal ileum
|
|
|
What is the commonest cause of pernicious anaemia?
|
Lack of intrinsic factor
|
|
|
What are dietary sources of B9?
|
Green vegetables and liver
|
|
|
What are the DRV and serum values for folate?
|
DRV 50ug, serum levels 15ng/ml
|
|
|
What does folate do?
|
Carries free carbons, essential in purine and pyrimidine synthesis
Important cofactor in metabolism of homocysteine to methionine |
|
|
What does cobalamin do?
|
Important cofactor in metabolism of homocysteine to methionine
|
|
|
Do the signs of deficiency in folate and cobalamin differ?
|
No. Folate is required to supply the free carbons for MeTHF production. B12 essential for conversion of MeTHF back to folate pool, so in one will have the same effect
|
|
|
How does methotrexate work?
|
It inhibits the conversion of dihydrofolate (FH2) to Tetrahydrofolate (THF), the first step in the conversion of homocysteine to methionine
|
|
|
Why is methotrexate a useful chemotherapeutic drug?
|
By inhibiting dihydrofolate to THF reaction, methotrexate inhibits production of puridines and pyrimidines, preventing cells from multiplying
|
|
|
What is megaloblastosis?
|
Symptom of B9 and B12 deficiency - haemopoietic red cells cannot divide so die in bone marrow. Megaloblastosis = giant germ cells
|
|
|
What other symptoms of B9/B12 deficiency?
|
Inadequate myelin synthesis: causing numbness in extremities, tingling, ataxia, confusion, moodiness, depression, spinal cord and peripheral lesions
|
|
|
Why is folate important in pregnancy?
|
Prevent neural tube defects - v common in UK before pregnant mum's were advised to take B9 supplements
|
|
|
Name some neural tube defects
|
Anencephaly, spina bifida, encephalocele
|
|
|
What is pernicious anaemia?
|
Autoimmune gastritis, affects parietal cells causing high stomach pH and low release of intrinsic factor
|
|
|
What is B5?
|
Pantothenic acid
|
|
|
Where is B5 found?
|
All foods, so deficiency is rare
|
|
|
What does B5 do?
|
Component of enzyme CoASH important in fatty acid oxidation
|
|
|
What is biotin and what does it do?
|
Vitamin B7. Used in carboxylations such as pyruvate to oxaloacetate and Acetyl CoA to Malonyl CoA
|
|
|
What causes biotin deficiency?
|
Long term antibiotic therapy
|
|
|
What is ascorbic acid?
|
Vitamin C
|
|
|
What does vitamin C do?
|
Anti-oxidant, important in collagen formation, maintains Fe2 (ferric) so low vitamin C can cause iron deficiency
|
|
|
What is a vitamin C deficiency?
|
Scurvy - humans can store enough for ~6 months. Causes anaemia (not reducing dietary iron), haemorrhage (poor tissue formation), B9 deficiency
|
|
|
In the UK, who is most at risk from vitamin C deficiency?
|
Elderly, alcoholics, adolescents with poor diets
|
|
|
Should a smoker worry about vitamin C intake?
|
Yes, smokers need double normal intake
|
|
|
Does vitamin C in high doses have any benefits?
|
Unsure - thought to reduce length of cold symptoms. may improve immune function
|
|
|
Is vitamin C toxic in high doses?
|
Possible cause of kidney stones, diarrhoea, and systemic conditioning - pregnant women on high doses might cause dependence on high doses in new born
|
|
|
What are the fat soluble vitamins?
|
A, E, D, K
|
|
|
Deficiency of fat soluble vitamins caused by?
|
Primary deficiency a low fat diet, or fat malabsorption
|
|
|
What is vitamin A?
|
Retinol (retinal - active vision), retinoic acid (hormone)
|
|
|
Where is vitamin A found?
|
Animal liver, fish oils, whole milk, egg yolk
As B-carotnoids in yellow/orange veg and fruit |
|
|
How much retinol in a B-carotene and how is potency expressed?
|
Actually 2:1, but to get one useful retinol you need 6 carotenes - potency is expressed as retinol equivalents
|
|
|
What does vitamin A do?
|
Retinoic acid important in cell growth and differentiation
In vision retinal participates in conversion of light in rod cells, aiding low light vision (scotopic) |
|
|
How is vitamin A transported from diet to liver, and from liver to tissues?
|
In chylomicrons to liver. Then bound to pre-albumin to tissues.
|
|
|
What is vitamin A deficiency?
|
Night blindness, keratinisation of cornea leading eventually to irreversible blindness
|
|
|
What is vitamin A toxicity?
|
Dermatitis, hair loss, liver dysfunction, thinning bones
|
|
|
Should pregnant women eat liver?
|
No - high vitamin A content
|
|
|
What is vitamin E?
|
Tocopherols, especially alpha-tocopherol
|
|
|
What does vitamin E do?
|
Antioxidant. Prevents PUFA oxidation and subsequent oxidation chain reaction. This protects cell membranes and prostaglandins. Vitamin E becomes a free radical itself, but is stable.
|
|
|
Is human milk a good source of vitamin E?
|
No
|
|
|
Can vitamin E protect against atherosclerosis?
|
Possibly. Thought to protect APO-B and FAs in LDLs preventing breakdown
|
|
|
What is vitamin D?
|
Ergocalciferol (D2), Cholecalciferol (D3)
|
|
|
What are good sources of vitamin D?
|
Milk, dairy and eggs plus UV!!!
|
|
|
What does vitamin D do?
|
Preserves correct levels of calcium in the blood
|
|
|
What is vitamin D deficiency?
|
Rickets in children, and osteomalacia in adults
Rickets causes decrease in bone:bone matrix ratio - bendy bones Osteomalacia is decalcification of long bones causing weakness |
|
|
Is vitamin D toxic in excess?
|
Yes. Hypercalcaemia can cause calcification of viscera, plus GI disturbances and can be fatal
|
|
|
What is vitamin K?
|
Quinone and deriratives
|
|
|
What does vitamin K do?
|
Essential in blood clotting, vitamin K antagonist (Warfarin) used as an anti-clotting agent
|
|
|
What are good sources of vitamin K?
|
Green vegetables and milk, eggs, cereals
|
|
|
Does vitamin K cross the placenta?
|
No, so infants can be deficient and this can be fatal. As a result babies given vitamin K at birth since 1950s
|
|
|
What is a vitamin K deficiency?
|
Blood clot formation deficiency - caused by antibiotic therapy
|
|
|
What is the DRV for vitamin C?
|
75mg
|
|
|
What is present in capillaries, activated by insulin and is responsible for the hydrolysis of triacylglycerol (TAG) present in chylomicrons prior to its storage in adipose tissue?
|
Lipoprotein Lipase
|
|
|
What catalyses the hydrolysis of most of the dietary TAG in the small intestine?
|
Pancreatic Lipase
|
|
|
What is bound to the plasma membrane. It catalyses the release of Inositol trisphosphate which results in increased calcium ion concentration in the cell?
|
Phospholipase C
|
|
|
What is activated by glucagon in the fasting state, catalyses the hydrolysis of TAG stored in the adipocyte which results in the release of fatty acids in the circulation?
|
Hormone sensitive Lipase
|
|
|
What catalyses the hydrolysis of TAG present in Very Low
Density Lipoprotein (VLDL) prior to its storage in adipose tissue? |
Lipoprotein Lipase
|

