![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
25 Cards in this Set
- Front
- Back
|
What is the pallium and how does it differ between lower and higher verts |
All verts have dorsal pallium → develops into cortex (and palial nuclei) Chicks → 3 layers Rodents → 5 layers (because 2/3 combined) Humans → 6 layers + upper layers exapanded |
|
|
What's the canonical circuit in the cortex? |
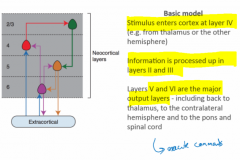
Entry at 4 →
Processed in 2/3→ Output from 5/6 → either corticocortical or corticofugal (i.e. to thalamus, tectum, bulb, spine) Although obviously specific cortical areas have specific functionality |
|
|
Name some developmental and adult cortex diseases |
Developmental Cerebral palsy, autism, epilepsy, microcephaly, downs
Adult Dementia, stroke, tumour |
|
|
What molecules define the AP/DV axes in vert CNS? |
Anterior - Cerberus, Dickopff, RA → protects Anterior Posterior - Wnt, FGF → takes over Anterior Dorsal - Wnt/BMP Ventral - SHH |
|
|
What's the split between excite and inhib in the cortex |
80% excitatory, glutamatergic, one type of pyramidals, derived from Pax6/Mgm2 20% inhibitory, GABA interneurons, multiple cell types (difficult to classify), specified in subpallium → migrate to cortex |
|
|
What order are the cortical layers produced, and how do you know when they're done dividing? |
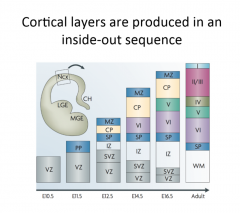
Inside out i.e. 1 on the outside comes last. Add BrdU → if still dividing → dilutes, if not → stays same. Layers produced temporally distinctly, one after the other. |
|
|
What distinguishes the layers and what makes them so distinct? |
They all have different transcriptional profiles and they mutually repress |
|
|
How do Radial Glia in ventricular zone divide? |
They divide symmetrically→ lots of progenitors Or divide asymmetrically - when determinants/spindle polarised→ differentiate→ migrate to cortical plate→ branching Or give rise to intermediate progenitors in SVZ (marked by Tbr2 TF)→ differentiates into 2 neurons (in any layer). (KO Tbr2→ smaller layers) Or generate outer radial glia in outer SVZ if cleavage horizontal/oblique→ self renew or produce intermediates |
|
|
What do Outer Radial Glia do? |

In Outer SVZ, produced by horizontal cleavage of radial glia Either self renew or produce intermediate progenitors Expanded outerSVZ / oRG→ probably underlies expanded cortex because allow many more neurons |
|
|
How are inhibitory interneurons specified? |
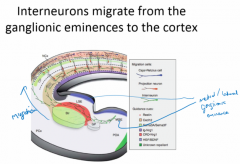
Shh patterns the subpallium→ split into lateral and medial ganglionic eminances (become striatum and pallidum later) (and telenchephalic stalk). LGE/MGE come through Hox genes Dlx1/2 expression, and if KO → Gsx1/2 expand, but if KO Gsx1/2→ partial rescue Interneurons then migrate from LGE/MGE→ cortex |
|
|
What determines and how can you classify interneurons? |
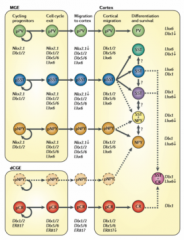
Determined by time/place of birth. Early → deep layers (makes sense because layer 6 formed first) (Ciceri et al 2013 - lentivirus dye) Can classify by morphology, electrical response, molecular markers, |
|
|
How do interneurons and pyramidals coord? |
Temporal competence! Key to specific connections and controlling firing Without that→ epilepsy |
|
|
What are the models for how layers could be specified from progenitors |
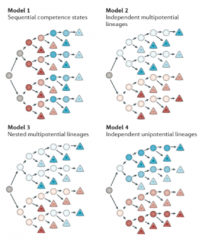
Sequential competence = temporal competence = supported by most of the evidence Although independent multipotential i.e. some are lineage restricted is supported by Franco (2013) who thinks Cux2 neurons are fate limited from the start, Guo (2013) suggests most of those that are fate limited are migrating interneurons from LGE/MGE, and those from the VZ go to all layers. Other two, just theoretical |
|
|
How do we know progenitors in SVZ/oSVZ populate all layers of cortex and how do we know sequential? |
Use retrovirus → label S-phase cells ∴ all progeny inherit retrovirus If labelled early → labels all layers If labelled late → only upper layers (Walsh and Cepko 1988) + if you take them out in vitro → first preplate, then deep, then upper |
|
|
How is the sequential progeny order specified in progenitors? |
Transplant early progenitors into late→ change to late. BUT Late→ transplant to early→ no change ∵ already specified. (Desai & McConnell 2000)
This change happens after birth, but if pre-birth, mid (not late) progenitors can be reset with ShRNA→ less FoxG1 = early progenitor (Shen et al 2008)
Mouse ES cells also shown to be intrinsic too - grown in vitro w/o morphogen→ regular temporal competence (Gaspard et al 2008)
Ikaros is a TF (hunchback homologue too!) that encourages early differentiation → if ectopic in early progenitors→ more early progeny (i.e. deep layer neurons), but no effect in later progenitors because they're expressing other factors... (Alsio et al 2013) |
|
|
How do sensory neurons from the body via the thalamus migrate to make connections in the cortex? |
Early stage guided by Netrin, Late stage guided by Eph/Ephrin interactions |
|
|
Does the lineage affect connectivity as well as which layer? |
Clones from the same progenitor are more likely to be connected to each other than non-clones (although circuit still matches canonical circuit described before i.e. in → 4 → 2/3 → 5/6 → out)(Yu et al 2009) |
|
|
How do the different neurons in each layer know where to target (assuming they're targeting outside local layers i.e. projection neurons to subcortical or callosally)? |
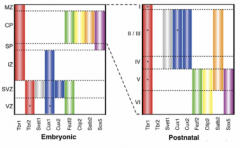
Distinct transcriptional identies in each layer means the neurons are unique and also means that each layer is unique so that it can be a specific target.
Postmitotic gene network of 4 genes ensures stereotyped neuronal projections i.e. Fezf2,Ctip2, Tbr1, Satb2 = complex genetic switch and mutually repress e.g. in layers 2-5, Satb2 and Ctip2 regulate Tbr1 (Srinivasan 2012) e.g. Ctip2 mutant→ corticospinal defects e.g. Fezf2→ responsible for callosal neuron reprogramming to corticospinal tract
|
|
|
3 ways in which cortical functions have been mapped?
|
|
|
|
Protomap vs Protocortex |
Protomap: Progenitors have already specified cortical area Protocortex: Stimuli from thalamus specifies cortex, so early cortex = tabula rasa Gbx2 mutants→ no thalamocortical innervation→ cortical areas stay specified ∴ protomap (Miyashita-lin et al 1999). BUT thalamic input refines areas e.g. no VI w/o input (Chou et al 2013) |
|
|
How is the protomap set up? |
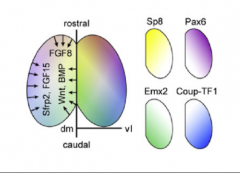
Cortical Hem = Wnts/BMP Antihem = Sfrp2, Fgf7, Tfgα Anterior Neural Ridge = Fgf8, Fgf17, Fgf18 → downstream gradients of Pax6, CoupTF1, Emx2, Sp8 (clockwise). No sharp boundaries except maybe V1. Increased Fgf8 → increased rostral. CoupTF1 mutant, expands frontal ∵ frontal can take over Whole network → mutual repression (between) and enhancement (within) → predictable perturbations |
|
|
Are there sharp cortex area boundaries? |
Eventually yes → specific markers for each cortex e.g. Lmo4 in Motor Cortex, Bhlhb5 in sensory/visual cortices. Loss of these TFs → loss of cortical arealisation |
|
|
Why bother modelling human brain development in vitro? |
Allows disease modelling and drug testing? |
|
|
How do you make human brains in vitro? |
HESCs or iPSCs→ Retinoids, SMAD inhibs→ cortical neural rosettes → IKNM→ generate RGs, IPs, oRGs→ neurons in correct order/number (Shi et al 20112) + correct 3D architecture (kinda because no thalamic inputs)→ 3D mini brains |
|
|
What do you see in models of downs or microcephaly? |
Downs (similar to alzheimers) = Aβ peptides→ plaques, phospho tau→ intracellular tangles Microcephaly → can be modelled with centrioler mutants → see if spindle orientation key e.g. mutant CDK 5R-AP2 → fewer progenitors → smaller brain |

