![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
144 Cards in this Set
- Front
- Back
|
Test:
Glucose Fermentation Durham Tube - Gas Production Test |
Media:
Phenol Red Glucose Broth Reactions: Glucose ----------------> acids Positive results: Yellow media |
|
|
Test:
Lactose Fermentation |
Media:
Phenol Red Lactose Broth Reactions: Lactose ----------------> acids Positive results: Yellow media |
|
|
Test:
Mannitol Fermentation |
Media:
Phenol Red Mannitol Broth Reactions: Mannitol ----------------> acids Positive results: Yellow media |
|
|
Test:
Mixed Acid Fermentation |
Media:
MR - VP Broth Reactions: Glucose ----------------> acids ADD: 4 drops of methyl red Positive results: Red media |
|
|
Test:
Butanediol Fermentation |
Media:
MR - VP Broth Reactions: 2 Pyruvate --> Butanediol + Barritt's reagent Positive results: Pink to Red media |
|
|
Test:
Starch Hydrolysis |
Media:
TSI Slant Reactions: Starch ---------> maltose + glucose + Gram's iodine Positive results: CLEAR ZONE along streak |
|
|
Test:
Catalase Production (Test) |
Media:
TSA slant Reactions: 2H O ----------> O + 2H O Positive results: Bubbles media |
|
|
Test:
Urea Hydrolysis |
Media:
Urea broth Reactions: Urea ----------> ammonia Positive results: Bright pink media |
|
|
Test:
Tryptophan Hydrolysis (Degradation) |
Media:
Tryptone broth Reactions: Tryptophan----------> Indole + Kovac's reagent Positive results: Red media |
|
|
Test:
Citrate Utilization (Test) |
Media:
Simmons Citrate Agar Slate Reactions: Citrate ----------> alkaline products Positive results: Prussian blue media |
|
|
Test:
Nitrate Reduction |
Media:
Nitrate broth Reactions: NO ----------> NO + Reagent A + Reagent B Positive results: Deep red media |
|
|
Test:
Sugar Fermentation |
Media:
TSI Slant Reactions: Glucose -------------> acids Positive results: Red slant/Yellow butt |
|
|
Test:
Sugar Fermentation |
Media:
TSI Slant Reactions: Glucose -------------> acids Lactose -------------> acids Sucrose -------------> acids Positive results: Yellow slant/Yellow butt |
|
|
Test:
CO Production |
Media:
TSI Slant Reactions: Gas Production Positive results: Bubbles/cracks |
|
|
Test:
Hydrogen Sulfide Production |
Media:
TSI Slant Reactions: Cysteine -------------> H S Positive results: Black butt |
|
|
Test:
Casein Hydrolysis |
Media:
Skim milk agar plate Reactions: Casein -----------> peptides + amino acids Positive results: CLEAR ZONE along streak |
|
|
Aerobic
|
Requires oxygen
|
|
|
Agar
|
Complex polysaccharide derived from a marine alga (red) and used as a soldifying agent in culture media (agar by itself is NOT a nutrient)
|
|
|
Aseptic technique
|
To inoculate the sterile medium with a pure culture
of microorganisms without outside contamination. |
|
|
Autoclave
|
A machine used to sterilize by the use of steam under pressure. Insures that the spores are destroyed. It will go up to fifteen pounds of pressure per square inch. It will be kept at 121 degrees centigrade for 15 to 20 minutes.
|
|
|
Broth
|
Medium without agar
|
|
|
Brownian Movement
|
Vibrations of an object seen in a microscope, not true motility.
|
|
|
CFU
|
Colony-forming units, i.e. colonies.
|
|
|
Colony
|
A macroscopically visible population of cells growing on solid medium, arising from a single cell.
|
|
|
Culture medium
|
Those nutrients essential for growth of a given microorganism.
Provides suitable surroundings for growth = the proper pH, osmotic pressure, oxygen, temperature, etc. |
|
|
Differential Stain
|
Uses 2 or more dyes which allow differentiation between different bacterial groups or structures.
|
|
|
Counterstain
|
The 2nd dye added to a smear, taken in after the wall is decolorized.
|
|
|
Decolorizer
|
The reagent used to remove the primary dye from the cell wall in a differential stain, e.g. acid alcohol, acetone-alcohol.
|
|
|
Deep
|
Agar media that solidifies in a vertical tube.
|
|
|
EMB aqar
|
Eosin-methylene blue agar; contains bile salts and the dyes eosin and methylene blue, all inhibitory to Gram (+) bacteria, therefore, this medium selects for Gram (-) bacteria; it differentiates lactose-fermenting bacteria from non-lactose-fermenting bacteria.
|
|
|
Facultative anaerobe
|
Uses oxygen when present but can either ferment or anaerobically respire without it.
|
|
|
Fastidious
|
Hard-to-grow bacteria, requiring grow factors or particular nutrients.
|
|
|
Flaqella
|
A structure for motility.
|
|
|
Genus
|
Category of organisms with like features and closely related, divided into species.
|
|
|
Growth
|
The development of a population of cells from one or a few cells.
This is not growth in size of the cells, but growth with respect to multiplication of the cells. |
|
|
Inoculum
|
Material used to initiated a microbial culture.
|
|
|
Inoculate
|
The transfer of the cells you want to grow into a culture medium.
|
|
|
Halophilic
|
Salt-tolerant or salt-loving (salt-requiring).
|
|
|
Incubate
|
Cells placed in an environment providing suitable growth conditions.
|
|
|
MAC
|
MacConkey agar; contains bile salts and crystal violet, both inhibitory to Gram(+) bacteria, therefore, this medium selects for Gram(-) bacteria; it differentiates lactose-fermenting bacteria from non-lactose-fermenting bacteria.
|
|
|
MSA
|
Mannitol salt agar; contains 7.5% NaCI, which is inhibitory to most bacteria, therefore it selects for halophillic (halotolerant) bacteria; it differentiates mannitol-fermenting bacteria from non-mannitol
fermenting bacteria. |
|
|
Mixed Culture
|
More than one species of microorganisms.
|
|
|
Obligate Aerobe
|
Requires oxygen to grow.
|
|
|
Obligate Anaerobe
|
Does not use oxygen to grow, may even be killed by it.
|
|
|
Pathogenic
|
Disease-causing.
|
|
|
Petri dish
|
Shallow dishes with a lid that nest over the bottom to prevent contamination.
|
|
|
Petri (or culture) Plate
|
Petri dish that is filled.
|
|
|
PFU
|
Plaque-forming units produced by bacterial viruses when infecting host bacterial cells.
|
|
|
Plaque
|
Destruction of the bacterial lawn by a bacteriophage
as the lytic infection progresses. |
|
|
Pour Plate
|
Procedure where liquefied agar has been poured into a Petri dish after being mixed with bacteria.
|
|
|
Primary Stain
|
The 1st dye used in a differential stain,
e.g. malachite green, crystal violet, carbolfuchsin. |
|
|
Pure culture
|
Single species of microorganisms.
|
|
|
Slant
|
Test tubes with agar media that are allowed to solidify with
the tube held at an angle so that a large surface area for growth is available. |
|
|
Species
|
A subdivision of a genus.
|
|
|
Sterile
|
Heated to a point where all organisms present
are destroyed. It may not be heated, but basically the word sterile means absolutely no organisms. |
|
|
Streak Plate
|
Procedure where a bacterial specimen is placed on a pre-made plate and diluted out using flame and multiple sections.
|
|
|
TSB
|
Trypticase soy broth
|
|
|
TSA
|
Trypticase soy agar
|
|
|
Turbidity
|
Cloudiness in liquid broth. The more bacteria,
the more turbidity there is. |
|
|
Ubiquitous
|
Bacteria are universally distributed; they are everywhere.
|
|
|
Zone of Inhibition
|
Area of no bacterial growth around a chemical on a disc, indicates sensitivity.
|
|
|
Know the function of the streak plate technique
|
Streak plate for isolation
|
|
|
Type of media used for streak plate.
|
TSB
|
|
|
Understand the differences in cell wall structure of
gram(+) VS. gram(-) bacteria |
Gram(+):
THICK peptidoglycan Teichoic acid Gram (-): THIN peptidoglycan OUTER membrane Periplasm |
|
|
Be able to identify gram reaction under microscope or in diagram
|
Gram(+) Purple
Gram(-) Pink |
|
|
Staphylococcus aureus
|
Gram(+), Coccus, Staphylococci
Habitats: Lives on skin/in nose Can cause skin infections, pneumoniae, Toxic Shock Syndrome |
|
|
Escherichia coli
|
Gram Reaction:
Gram(-), Bacillus, Single bacillus Habitats: Lives in the lower intestines of mammals Can cause food poisoning; UTIs |
|
|
Micrococcus luteus
|
Gram(+), Coccus, Tetrads
Habitats: Found in soil, dust, water, air and normal flora of the mammalian skin Non-pathogenic but a contaminant |
|
|
Serratia marcescens
|
Gram(-), Bacillus, Coccobacillus
Habitats: Lives in damp conditions(bathroom, pink discoloration) Pathogenic (involved in nosocomial infections) UTI's |
|
|
Enterococcus faecalis
|
Gram(+),Coccus,Streptococci
Habitats: Inhabits the GI tracts of human and other mammals Can cause infections in humans especially in nosocomial (hospital) environment |
|
|
Pseudomonas aeruginosa
|
Gram(-),Bacillus, Single bacillus
Habitats: Common inhabitants of soil & water; occur regularly on surfaces of plants and occasionally on the surfaces of animals |
|
|
Bacillus subtilis
|
Gram(+),Bacillus,Single bacillus
Habitats: Able to form tough, protective endospores wihich allows organism to tolerate extreme environmental conditions Not considered human pathogens MAY contaminate foods |
|
|
Be able to determine CPUs
|
Organisms/ml =
# colonies X inverse of dilution 1: 100,000 80 colonies X 100,000 = 8,000,000 8.0 x 10 CFU/ml |
|
|
Antimicrobic Sensitivty Test
What method was used? |
Kirby-Bauer Method
|
|
|
What type of AGAR?
|
Mueller-Hinton II agar
|
|
|
What antiobiotics were used in lab?
|
Tetracycline
Ampicillin Pencillin Sulfamethoxazole Chloramphenicol Tobramycin |
|
|
What are the modes of actions of antibiotics?
Tetracycline |
Inhibits protein synthesis
|
|
|
What are the modes of actions of antibiotics?
Ampicillin |
Inhibits cell wall synthesis
|
|
|
What are the modes of actions of antibiotics?
Pencillin |
Inhibits cell wall synthesis
|
|
|
What are the modes of actions of antibiotics?
Sulfamethoxazole |
Inhibits nucleic acid synthesis
|
|
|
What are the modes of actions of antibiotics?
Chloramphenicol |
Inhibits protein synthesis
|
|
|
What are the modes of actions of antibiotics?
Tobramycin |
Inhibits protein synthesis
|
|
|
How did our species respond to the six different antibiotics?
Staphylococcus aureus |
Sensitive to all 6 antibiotics.
|
|
|
How did our species respond to the six different antibiotics?
Escherichia coli |
RESISTANT to Penicillin; sensitive to Ampicillin, Sulfamethoxazole, Tetracycline, Tobramycin, and Chloramphenicol.
|
|
|
How did our species respond to the six different antibiotics?
Pseudomonas aeruginosa |
RESISTANT to Penicillin, Ampicillin, Sulfamethoxazole, & Chloramphenicol; INTERMEDIATE to Tetracycline; sensitive to Tobramycin.
|
|
|
What is an antiseptic?
|
Chemicals applied to animal surfaces (skin/mucous membrane)to prevent infection by killing or inhibiting the growth of pathogens
|
|
|
What antiseptics were tested?
|
Iodine
Alcohol Listerine |
|
|
ANTISEPTIC
What type of agar was used? |
TSA
|
|
|
How did our species respond to the three different antiseptics?
Staphylococcus aureus Most effective? |
Greatest to least:
Iodine, Alcohol, Listerine. I A L MOST EFFECTIVE: Iodine |
|
|
How did our species respond to the three different antiseptics?
Pseudomonas aeruginosa Most effective? |
Greatest to least:
Alcohol, Iodine, Listerine. A I L Most effective: ALCOHOL |
|
|
How did our species respond to the three different antiseptics?
Escherichia coli Most effective? |
Greatest to least:
Alcohol, Iodine, Listerine. A I L Most effective: ALCOHOL |
|
|
What is a disinfectant?
|
Agents, usually chemicals, used to disinfect inanimate objects
|
|
|
DISINFECTANT
What species of bacteria were tested? |
Escherichia coli
Bacillus subtilus |
|
|
DISINFECTANT
Compare and contrast the two species tested. What were their growth patterns like and why? |
Bacillus subtilis (gram(+)) was more resistant to disinfectants than Escherichia coli because of its ability to form endospores in the harsh chemical environment and Gram(-).
|
|
|
What was the MOST EFFECTIVE disinfectant against each species?
Escherichia coli |
PINE SOL, Lysol, Bleach
|
|
|
What was the MOST EFFECTIVE disinfectant against each species?
Bacillus subtilis |
LYSOL, Bleach, Pine sol
(Effectiveness was the same at 1 min, 5 min, & 10 min) |
|
|
What disinfectants were tested?
|
Lysol
Pine sol Bleach |
|
|
What is a phage?
|
Virus
|
|
|
Be able to determine PFUs (organism/ml = # of plaques x inverse of dilution)
|
Plate 1:
Phage particles/mL = # of plaques X dilution factor Phage particles/mL = 500 X 1,000,000 Phage particles/mL = 500,000,000 -> 5.0 X 10 PFU/mL |
|
|
Be able to determine PFUs (organism/ml = # of plaques x inverse of dilution)
|
Plate 2:
Phage particles/mL = # of plaques X dilution factor Phage particles/mL = 50 X 10,000,000 Phage particles/mL = 500,000,000 -> 5.0 X 10 PFU/mL |
|
|
Be able to determine PFUs (organism/ml = # of plaques x inverse of dilution)
|
Plate 3:
Phage particles/mL = # of plaques X dilution factor Phage particles/mL - 5 X 100,000,000 Phage particles/mL - 500,000,000 -> 5.0 X 10 PFU/mL Phage particles/mL - 500,000,000 -> 5.0 X 10 PFU/mL *In this example, the bacteriophage titer (number of phage particles in the original stock of 74) is 5.0 X 108 PFU/mL. |
|
|
Differential Stain:
Primary stain Mordant Decolorizer Counterstain |
Gram Stain:
Crystal Violet Gram’s Iodine Ethanol Safranin |
|
|
Microaerophilic
|
Likes a reduced oxygen concentration.
|
|
|
Shapes:
Coccus (cocci) Bacillus (bacilli) Spiral (curve rods) |
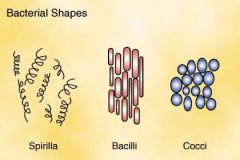
Shapes
|
|
|
What colony characteristics can be used for differentiation of bacterial species? As an example, compare the properties of colonies of Serratia marcescens and Micrococcus luteus on your streak plate. (EX. 9)
|
Color, shape of colonies
|
|
|
Coccus Arrangements:
Single coccus Diplococci Tetrads Sarcinae Streptococci Staphylococci |
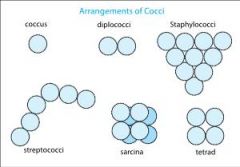
`
|
|
|
Staphylococcus epidermis
|
Gram(+),Cocci,Staphylococci
Habitats: Occurs frequently on the skin of humans & animals and in mucous membranes |
|
|
Myobacterium smegmatis
|
NOT truly gram(+) NOR gram (-)
Bacillus, Single bacillus Habitats: Found in normal genital secretions |
|
|
Proteus mirabilis
|
Gram(-), Bacillus, Single bacillus
Habitats: Cause UTIs, wound infections, septicemia & pneumonias Habitat is human intestinal tract, soil, water & plants |
|
|
Klebsiella pneumoniae
|
Gram(-), Bacillus, Single bacillus
Habitats: Can cause pneumonia due to capsule Found in the normal flora of mouth, skin & intestines |
|
|
T4 Figure
Know Structure |
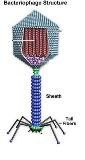
Structure
|
|
|
What is a bacteriphage?
|
Viruses that infect bacteria
|
|
|
Species specificity of T4
|
E. Coli, Strain B
|
|
|
Pseudomonas aeruginosa after primary stain is added.(EX.14)
|
Purple
|
|
|
Bacterial colonies (CFU)
|
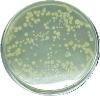
`
|
|
|
Test:
Antibiotic Sensitivity Testing |
Media:
Mueller Hinton Agar plate |
|
|
Test:
Antiseptic Sensitivity Testing |
Media:
Trypticase Soy Agar plate |
|
|
Test:
Disinfectant Sensitivity Testing |
Media:
Trypticase Soy Broth |
|
|
Test:
Gram Stain |
Media:
Trypticase Soy Agar plate |
|
|
Spiral Arrangments:
Vibrio Spirillum Spirochete |

Arrangements
|
|
|
Test:
Bacteriophage Titer determination |
Media:
Soft Agar/TSA plate |
|
|
In regards to bacterial growth on solid media, define the term "colony".(EX. 9)
|
A macroscopically visible population of cells growing on solid medium, rising from a single cell.
|
|
|
Why is the loop flamed before it is placed in a culture tube? Why is it flamed after completing the inoc¬ulation?(EX. 9)
|
To sterilize the loop for aseptic technique
|
|
|
Provide two reasons why plates should be inverted during incubation.(EX. 9)
|
1.The problem of moisture on the cover lid is eliminated.
2. Moisture can become deposited on the agar surface causing the organisms to spread over the surface (defeating the isolation process.) |
|
|
How does smear preparation of cells from a liquid medium differ from preparation of cells from a solid medium? (EX.10)
|
You must add a drop of water to suspend the organism in when obtaining it from
a solid medium |
|
|
Why is it important to limit the quantity of cells used
to prepare a smear? (EX.10) |
To see individual cells
|
|
|
For preparation of a smear on a slide, what is the purpose of heat fixation? What problems can arise when the slide is heated in a flame? (EX.10)
|
1.To adhere to a slide
2.To kill m.o. destroy/denature part or all of the cell microorganisms |
|
|
Which of the three differential stains would likely be the first
used when identifying an unknown bacterium? Explain. (EX.14) |
Gram stain – it tells you if it is gram (+) or (-) and tells you the shape/arrangement of cells.
|
|
|
What is the function of a mordant? (EX.14)
|
(To adhere stain) Form a crystal complex with crystal violet so that
gram(+) bacteria retain the purple stain. |
|
|
For differential staining, how does a counterstain differ from
a primary stain? (EX.14) |
It is a less intense stain
|
|
|
How do gram-positive and gram-negative bacteria differ in cellular structure? How does this contribute to their differential staining properties? (EX.14)
|
Cell walls differ:
gram (+) - thick layer of peptidoglycan and teichoic acids gram (-) – thin layer of peptidoglycan, periplasm, outer membrane |
|
|
Which is the most critical step in the Gram-stain procedure? Why?
If this procedure is done incorrectly, how might that affect the final results? (EX.14) |
Decolorizing with ethanol; if left too long it can discolor the gram (+) cells
|
|
|
How does culture age affect the results of a Gram-stain? (EX.14)
|
Grams (+) can convert to gram-variable or gram (-)
|
|
|
Why must smear thickness be considered before
performing a Gram-stain? (EX.14) |
If a smear is too thick you will not see individual cells
|
|
|
Bacteriophage plaques(PFU)
|

Plaques are clear zones formed in a lawn of cells due to lysis
by phage. |
|
|
What color are bacterial endospores after a
Gram-stain is performed? What does this tell you about the physical properties of endospores?(EX.14) |
Colorless
Endospores are highly resistant to stains, heat, etc… |
|
|
Staphylococcus aureus before primary stain is added.(EX.14)
|
No color
|
|
|
Bacillus megaterium after the mordant is added.(EX.14)
|
Purple
|
|
|
Staphylococcus aureus cells after the decolorizer is used. (EX.14)
|
Purple
|
|
|
Moraxella (Branhamella) catarrhalis after the decolorizer is used. (EX.14)
|
No color-gram(-)
|
|
|
Bacillus megaterium after the counterstain is added. (EX.14)
|
Purple
|
|
|
Bacillus Arrangements:
Single Bacillus Coccobacillus Diplobacilli Streptobacillus |

`
|
|
|
Pseudomonas aeruginosa after the counterstain is added.(EX.14)
|
Pink
|
|
|
Why has the classification of slime molds been difficult? (Ex. 20)
|
`
|

