![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
67 Cards in this Set
- Front
- Back
|
3) Which of the following is the best choice of reagents to effect the electrophilic iodination of an aromatic ring?
A) KI, acetone B) I2, CH3CN C) KI, HNO3 D) I2, HNO3 E) none of the above |
D) I2, HNO3
|
|
|
4) How many distinct nodal planes which are perpendicular to the molecular plane are present in the π4* orbital of benzene?
A) 0 B) 1 C) 2 D) 4 E) 5 |
C) 2
|
|
|
5) How many pairs of degenerate π molecular orbitals are found in benzene?
A) 6 B) 5 C) 4 D) 3 E) 2 |
E) 2
|
|
|
6) Which of the following undergoes SN2 reaction with sodium methoxide most rapidly?
A) PhCH2Br B) Ph3CBr C) PhCH2CH2Br D) PhBr E) PhCH2CH2CH2Br |
A) PhCH2Br
|
|
|
7) Which of the following undergoes solvolysis in methanol most rapidly?
A) PhCH2Br B) Ph3CBr C) PhCH2CH2Br D) PhBr E) PhCH2CH2CH2Br |
B) Ph3CBr
|
|
|
8) The nitration of anisole:
A) proceeds more rapidly than the nitration of benzene and yields predominantly the meta product. B) proceeds more rapidly than the nitration of benzene and yields predominantly the ortho, para products. C) proceeds more slowly than the nitration of benzene and yields predominantly the meta product. D) proceeds more slowly than the nitration of benzene and yields predominantly the ortho, para products. E) proceeds at the same rate as the nitration of benzene and yields predominantly the meta product. |
B) proceeds more rapidly than the nitration of benzene and yields predominantly the ortho, para products.
|
|
|
9) Which of the following compounds will undergo bromination most rapidly using Br2, FeBr3?
A) p-methylacetanilide B) bromobenzene C) acetanilide D) benzenesulfonic acid E) dibromobenzene |
A) p-methylacetanilide
|
|
|
9) Which of the following compounds will undergo bromination most rapidly using Br2, FeBr3?
A) p-methylacetanilide B) bromobenzene C) acetanilide D) benzenesulfonic acid E) dibromobenzene |
A) p-methylacetanilide
|
|
|
11) Which of the following is the same as the tropylium ion?
A) cycloheptatrienyl cation B) cycloheptatrienyl anion C) cyclopentadienyl cation D) cyclopentadienyl anion E) cyclopropenyl anion |
A) cycloheptatrienyl cation
|
|
|
10) In electrophilic aromatic substitution reactions, a -NHCOCH3 substituent on the aromatic ring is:
A) a deactivator and a m-director. B) a deactivator and an o,p-director. C) an activator and a m-director. D) an activator and an o,p-director. E) none of the above. |
D) an activator and an o,p-director.
|
|
|
16) Nitrogen's lone pair electrons occupy what type of orbital in pyridine?
A) s B) sp C) sp2 D) sp3 E) p |
C) sp2
|
|
|
15) Describe the number of signals and their splitting in the 1H NMR spectrum of (CH3)2CHOCH3.
A) 3 signals: 2 doublets and a septet B) 2 signals: a doublet and a septet C) 3 signals: a doublet, a quartet, and a septet D) 4 signals: 2 doublets, a singlet, and a septet E) 3 signals: a singlet, a doublet, and a septet |
E) 3 signals: a singlet, a doublet, and a septet
|
|
|
12) Which of the following compounds will undergo Friedel-Crafts alkylation with
(CH3)3CCl, AlCl3 most rapidly? A) toluene B) iodobenzene C) acetophenone D) benzenesulfonic acid E) cyanobenzene |
A) toluene
|
|
|
14) What is the major organic product which results when cycloheptene is irradiated in the presence of N-bromosuccinimide?
A) 1-bromocycloheptene B) 2-bromocycloheptene C) 1,2-dibromocycloheptane D) 3-bromocycloheptene E) 4-bromocycloheptene |
D) 3-bromocycloheptene
|
|
|
13) Which of the following compounds will react most rapidly when treated with
CH3CH2Cl and AlCl3? A) benzene B) chlorobenzene C) nitrobenzene D) anisole E) toluene |
D) anisole
|
|
|
15) Including all possible stereoisomeric forms, how many distinct allylic bromides could be produced when 2-methylpent-1-ene is treated with NBS under irradiation by a sunlamp?
A) 2 B) 3 C) 4 D) 5 E) 6 |
D) 5
|
|
|
13) Which of the following compounds will react most rapidly when treated with
CH3CH2Cl and AlCl3? A) benzene B) chlorobenzene C) nitrobenzene D) anisole E) toluene |
D) anisole
|
|
|
21) If a signal is observed in the 170 to 200 ppm range in a C-13 NMR sectrum, what is the most likely type of functional group associated with that carbon atom?
A) carbonyl carbon B) allylic carbon C) aromatic carbon D) carbon/carbon triple bond E) carbon/carbon double bond |
A) carbonyl carbon
|
|
|
18) In the addition of an electrophile to acetophenone, which of the following best describes the expected mode of reaction?
A) The o,p-positions are most activated to attack by the electrophile. B) The m-positions are most activated to attack by the electrophile. C) The o,p-positions are most deactivated to attack by the electrophile. D) The m-positions are most deactivated to attack by the electrophile. E) All positions (o, m, and p) are equally activated to attack by the electrophile. |
C) The o,p-positions are most deactivated to attack by the electrophile.
|
|
|
17) How many electrons are present in the nonbonding π molecular orbital of the allyl cation?
A) 0 B) 1 C) 2 D) 3 E) 4 |
A) 0
|
|
|
22) What is the approximate chemical shift of an alkynyl carbon in 13C NMR spectroscopy?
A) 10 ppm B) 30 ppm C) 70 ppm D) 120 ppm E) 200 ppm |
C) 70 ppm
|
|
|
19) In electrophilic aromatic substitution reactions, a -CO2H substituent on the aromatic ring is:
A) a deactivator and a m-director. B) a deactivator and an o,p-director. C) an activator and a m-director. D) an activator and an o,p-director. E) none of the above. |
A) a deactivator and a m-director.
|
|
|
22) Which of the following compounds has the lowest boiling point?
A) 1,2,3-trichlorobenzene B) 1,2,4-tirchlorobenzene C) p-dichlorobenzene D) m-dichlorobenzene E) o-dichlorobenzene |
C) p-dichlorobenzene
|
|
|
21) Which of the following compounds will not undergo Friedel-Crafts acylation when treated with CH3CH2COCl, AlCl3?
A) toluene B) p-xylene C) anisole D) ethoxybenzene E) benzophenone |
E) benzophenone
|
|
|
21) Which of the following compounds will not undergo Friedel-Crafts acylation when treated with CH3CH2COCl, AlCl3?
A) toluene B) p-xylene C) anisole D) ethoxybenzene E) benzophenone |
E) benzophenone
|
|
|
23) Which of the following compound has the highest melting point?
A) benzene B) toluene C) o-dichlorobenzene D) m-dichlorobenzene E) p-dichlorobenzene |
E) p-dichlorobenzene
|
|
|
20) Which substrate would react most rapidly in an SN2 reaction?
A) CH3CH2CH=CHCH2Br B) BrCH2CH2CH=CHCH3 C) CH3CHBrCH=CH2CH3 D) CH3CH2CH2CH=CHBr |
A) CH3CH2CH=CHCH2Br
|
|
|
27) What type of carbon environment does not generate a signal in the DEPT-90 spectrum and gives a positive peak in the DEPT-135 spectrum?
A) quaternary B) methine C) methylene D) methyl E) carbonyl |
D) methyl
|
|
|
22) Which of the following compounds will undergo bromination least rapidly when treated with Br2 and FeBr3?
A) p-methylacetanilide B) bromobenzene C) acetanilide D) benzenesulfonic acid E) benzene |
D) benzenesulfonic acid
|
|
|
22) Which of the following compounds will undergo bromination least rapidly when treated with Br2 and FeBr3?
A) p-methylacetanilide B) bromobenzene C) acetanilide D) benzenesulfonic acid E) benzene |
D) benzenesulfonic acid
|
|
|
24) In the UV-visible spectra of the following compounds, in which does lambda max appear at the highest wavelength?
A) 3-phenylpropene B) 1-phenylpropene C) benzene D) n-octylbenzene E) isopropylbenzene |
B) 1-phenylpropene
|
|
|
28) What type of carbon environment does not generate a signal in the DEPT-90 spectrum and gives a negative signal in the DEPT-135 spectrum?
A) quaternary B) methine C) methylene D) methyl E) carbonyl |
C) methylene
|
|
|
23) Which of the following compounds will react least rapidly when treated with CH3CH2Cl and AlCl3?
A) o-xylene B) acetanilide C) toluene D) benzene E) bromobenzene |
E) bromobenzene
|
|
|
25) In the proton NMR, in what region of the spectrum does one typically observe hydrogens bound to the aromatic ring?
A) 1.0-1.5 ppm B) 2.0-3.0 ppm C) 4.5-5.5 ppm D) 7.0-8.0 ppm E) 9.0-10.0 ppm |
D) 7.0-8.0 ppm
|
|
|
29) Which compound generates positive peaks for the carbonyl in both its DEPT-90 and DEPT-135 spectra?
A) CH3CH2CHO B) CH3CH2COCH3 C) CH3CO2CH2CH3 D) CH3CH2CONH2 E) H2CO |
A) CH3CH2CHO
|
|
|
25) Which of the following compounds undergoes reaction with HNO3/H2SO4 at the fastest rate?
A) ethylbenzene B) benzenesulfonic acid C) nitrobenzene D) chlorobenzene E) acetophenone |
A) ethylbenzene
|
|
|
26) In the carbon NMR, in what region of the spectrum does one typically observe carbons which are part of the aromatic ring?
A) -10-0 ppm B) 40-60 ppm C) 80-100 ppm D) 120-150 ppm E) 200-220 ppm |
D) 120-150 ppm
|
|
|
30) What multiplicities are observed for the signals in the off-resonance decoupled 13C spectrum of 2-chloropropene?
A) 3 singlets B) 2 singlets and a doublet C) a singlet and 2 doublets D) a singlet, a doublet and a triplet E) a singlet, a triplet, and a quartet |
E) a singlet, a triplet, and a quartet
|
|
|
30) Which of the following compounds would most likely be used in the preparation of isobutylbenzene from benzene?
A) (CH3)2CHCOCl B) (CH3)2CHCH2Cl C) (CH3)2CHCH2Br D) CH3CH2CH2CH2Cl E) CH3CH2CH2COCl |
A) (CH3)2CHCOCl
|
|
|
30) Which of the following compounds would most likely be used in the preparation of isobutylbenzene from benzene?
A) (CH3)2CHCOCl B) (CH3)2CHCH2Cl C) (CH3)2CHCH2Br D) CH3CH2CH2CH2Cl E) CH3CH2CH2COCl |
A) (CH3)2CHCOCl
|
|

9) The protons marked Ha and Hb in the molecule below are __________.
A) chemically equivalent B) enantiotopic C) diastereotopic D) endotopic E) none of the above |
A) chemically equivalent
|
|
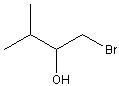
10) How many unique NMR signals (disregard splitting) would be predicted in the 1H spectrum of the following compound ?
A) 2 B) 3 C) 4 D) 5 E) 6 F) 7 |
F) 7
|
|
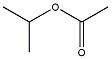
13) What 1H NMR spectral data is expected for the compound shown?
A) 3.8 (1H, septet), 2.1 (3H, s), 1.0 (6H, d) B) 3.8 (1H, septet), 3.3 (3H, s), 1.0 (6H, d) C) 3.3 (3H, s), 2.6 (3H, septet), 1.0 (6H, d) D) 2.6 (1H, septet), 2.1 (3H, s), 1.0 (6H, d) |
A) 3.8 (1H, septet), 2.1 (3H, s), 1.0 (6H, d)
|
|
|
32) When o-fluorotoluene is treated with sodium amide, the product is:
A) only o-toluidine. B) only m-toluidine. C) only p-toluidine. D) a mixture of o- and p-toluidine. E) a mixture of o- and m-toluidine. |
E) a mixture of o- and m-toluidine.
|
|
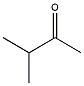
14) What 1H NMR spectral data is expected for the compound shown?
A) 3.8 (1H, septet), 2.1 (3H, s), 1.0 (6H, d) B) 3.8 (1H, septet), 3.3 (3H, s), 1.0 (6H, d) C) 3.3 (3H, s), 2.6 (3H, septet), 1.0 (6H, d) D) 2.6 (1H, septet), 2.1 (3H, s), 1.0 (6H, d) |
D) 2.6 (1H, septet), 2.1 (3H, s), 1.0 (6H, d)
|
|
|
33) Which of the following compounds is least reactive in the nucleophilic aromatic substitution reaction with NaOH?
A) 2,4-dinitrochlorobenzene B) m-nitrochlorobenzene C) o-nitrochlorobenzene D) p-nitrochlorobenzene E) 3,5-dinitrochlorobenzene |
B) m-nitrochlorobenzene
|
|
|
34) Which toluidine isomers are possible products when m-bromotoluene is treated with NaNH2?
A) ortho, meta, and para B) meta only C) para only D) ortho only E) meta and para only |
A) ortho, meta, and para
|
|
|
35) When 2,4-dinitrochlorobenzene is treated with sodium hydroxide at 100°C followed by protonation:
A) 2,4-dinitrophenol is formed via an addition-elimination nucleophilic aromatic substitution mechanism. B) 2,4-dinitrophenol is formed via an elimination-additon nucleophilic aromatic substitution mechanism. C) 3,5-dinitrophenol is formed via an elimination-addition nucleophilic aromatic substitution mechanism. D) 3,5-dinitrophenol is formed via an electrophilic aromatic substitution mechanism. E) 2,4-dinitrophenol is formed via an electrophilic aromatic substitution mechanism. |
A) 2,4-dinitrophenol is formed via an addition-elimination nucleophilic aromatic substitution mechanism.
|
|
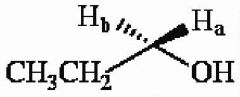
19) The protons marked Ha and Hb in the molecule below are __________.
A) chemically equivalent B) enantiotopic C) diastereotopic D) endotopic E) none of the above |
B) enantiotopic
|
|
|
38) p-Methoxybenzaldehyde can be prepared from anisole using the Gatterman-Koch formylation. What mixture of reagents is necessary for this process?
A) CO, HCl, AlCl3, CuCl B) CO, SO3, H2SO4 C) CO2, HCl, AlCl3 D) CO2, SO3, H2SO4 E) CO2, HNO3, H2SO4 |
A) CO, HCl, AlCl3, CuCl
|
|
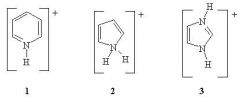
14) Rank the following in order of increasing pKa (from lowest to highest pKa)
A) 1 < 2 < 3 B) 3 < 2 < 1 C) 2 < 1 < 3 D) 3 < 1 < 2 E) 2 < 3 < 1 |
C) 2 < 1 < 3
|
|
|
26) Rank the following groups in order of increasing activating power in electrophilic aromatic substitution reactions: -OCH3, -OCOCH2CH3, -CH2CH3, -Br.
A) -CH2CH3 < -Br < -OCOCH2CH3 < -OCH3 B) -CH2CH3 < -OCOCH2CH3 < -Br < -OCH3 C) -Br < -OCOCH2CH3 < -CH2CH3 < -OCH3 D) -Br < -CH2CH3 < -OCOCH2CH3 < -OCH3 |
D) -Br < -CH2CH3 < -OCOCH2CH3 < -OCH3
|
|
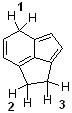
15) Which sequence ranks the indicated protons in order of increasing acidity?
A) 1 < 2 < 3 B) 2 < 3 < 1 C) 3 < 1 < 2 D) 3 < 2 < 1 |
D) 3 < 2 < 1
|
|
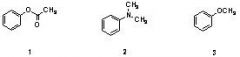
15) Which sequence correctly ranks the following aromatic rings in order of increasing rate of reactivity with chlorine and aluminum chloride
A) 1 < 2 < 3 B) 2 < 3 < 1 C) 3 < 2 < 1 D) 2 < 1 < 3 E) 1 < 3 < 2 |
E) 1 < 3 < 2
|
|
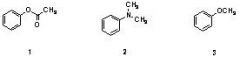
15) Which sequence correctly ranks the following aromatic rings in order of increasing rate of reactivity with chlorine and aluminum chloride
A) 1 < 2 < 3 B) 2 < 3 < 1 C) 3 < 2 < 1 D) 2 < 1 < 3 E) 1 < 3 < 2 |
E) 1 < 3 < 2
|
|
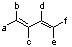
25) According to the 1,3-butadiene structure below, which positions would be best to place methoxy groups to yield the most reactive dimethoxy-1,3-butadiene isomer in the Diels-Alder reaction?
A) c and d B) a and c C) b and c D) a and d E) a and f |
D) a and d
|
|
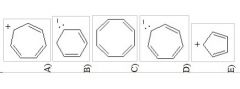
16) Which sequence ranks the aromatic rings in order of increasing reactivity in an electrophilic aromatic substitution reaction (slowest to fastest reacting)?
A) 1 < 2 < 3 B) 2 < 3 < 1 C) 3 < 2 < 1 D) 3 < 1 < 2 E) 2 < 1 < 3 |
E) 2 < 1 < 3
|
|
12) Which of the following structures is aromatic?
|
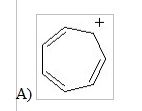
A
|
|
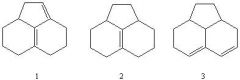
32) Which sequence correctly ranks the following conjugated systems in order of increasing UV 8max absorption values?
A) 1 - 2 - 3 B) 2 - 1 - 3 C) 2 - 3 - 1 D) 3 - 1 - 2 E) 3 - 2 - 1 |
C) 2 - 3 - 1
|
|
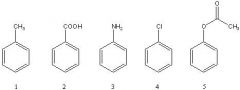
24) Rank the following compounds in order of increasing reactivity towards chlorination with Cl2/AlCl3 (slowest reacting to fastest).
A) 3 < 4 < 2 < 1 < 5 B) 2 < 4 < 1 < 3 < 5 C) 4 < 2 < 1 < 3 < 5 D) 2 < 4 < 5 < 1 < 3 E) 2 < 4 < 1 < 5 < 3 |
E) 2 < 4 < 1 < 5 < 3
|
|
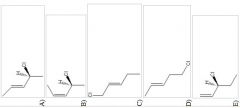
13) When 1 mole of anhydrous HCl is reacted with excess 1,3-pentadiene, both the 1,2 and the 1,4-addition products are formed. Which of the following structures shown below is the least likely to be one of these products? (Note: When a chiral carbon is formed in this reaction a racemic mixture results, only one of the two possible enantiomers is shown.)
|
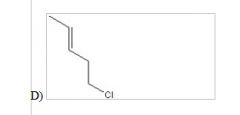
D
|
|
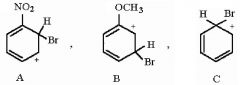
27) Rank the following sigma complexes in order of increasing stability.
A) A < B < C B) B < C < A C) B < A < C D) C < A < B |
A) A < B < C
|
|
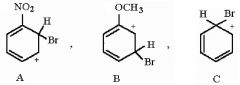
27) Rank the following sigma complexes in order of increasing stability.
A) A < B < C B) B < C < A C) B < A < C D) C < A < B |
A) A < B < C
|
|
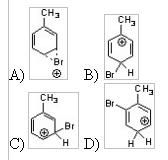
14) Which of the following is an intermediate in the bromination of toluene?
|
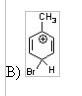
|
|
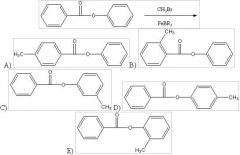
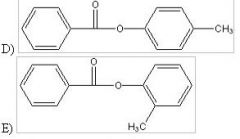
28) Predict the two major products from the following reaction. (Mark all correct responses.)
|
|
|
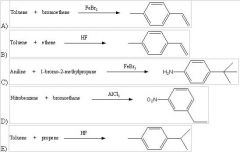
29) Which of the following reactions will actually yield the indicated product?
|

|
|
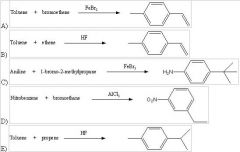
29) Which of the following reactions will actually yield the indicated product?
|

|

