![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
44 Cards in this Set
- Front
- Back
|
polar covalent |
0 - 0.5
|
|
|
non polar covalent |
0.6 - 1.9
|
|
|
ionic |
2.0 or higher
|
|
|
electron |
-same as proton
- |
|
|
proton |
-atomic number
|
|
|
neutron |
-mass number - proton
|
|
|
kilo (k) |
1000 = 10^3 |
|
|
deci (d) |
0.1 = 10^-1 |
|
|
centi (c) |
0.01 = 10^-2 |
|
|
milli (m) |
0.001 = 10^-3 |
|
|
micro (u) |
0.000001 = 10^-6 |
|
|
nano (n) |
0.000000001 = 10^-9 |
|
|
multiplication sig fig |
same number sig fig with the fewest |
|
|
division sig fig |
same number sig fig with fewest decimal points |
|
|
Fahrenheit |
F = 1.8(C) + 32 |
|
|
Kelvin |
K = C + 273 |
|
|
ammonium |
NH4+ |
|
|
hydroxide |
OH- |
|
|
combination |
two or more substance combine to form one product |
|
|
decomposition |
one substance breaks down into two or more simpler substance |
|
|
single replacement |
one element trades places with a different element in a compound |
|
|
double replacement |
element in two compound switch places |
|
|
combustion |
a compound containing carbon reacts with oxygen to form carbon dioxide |
|
|
1 mole |
6.022 x 10^23 |
|
|
boyle law |
pressure and volume P1 X V2 = P2 X V2 & inversely |
|
|
gay lussac law |
pressure and temperature P1 / T1 = P2 / T2 *must be in kelvin & directly |
|
|
charles law |
volume and temperature V1/T1 = V2/T2 *must be in kelvin & directly |
|
|
Avogadros law |
volume and number of mole V1/ n1 = V2/n2 *must be in kelvin & directly (both do exactly the same) |
|
|
combined gas law |
volume, temperture, and pressure P1xV1 / T1 = P2xV2 / T2 *must be in kelvin |
|
|
percent yield |
percent yield = actual yield (given in the problem ) / theoretical yield x 100 |
|
|
stp |
1 atm ( 760 mm Hg) & 22.4 L |
|
|
mass/volume percent |
grams of solute / mL of solution x 100 ___ g of solute / 100 mL of solution |
|
|
mass / mass percent |
grams of solute / grams of solution x 100 __ g of solute / 100 g of solution |
|
|
volume / volume percent |
mL solute / mL solution x 100 __ml of solute / 100 mL solution |
|
|
molarity concentration |
moles of solute / liters of solution (given mole) mole solute / 1 L solution |
|
|
1 gram = ? mg |
1000 mg |
|
|
Dilution formula |
C1xV1 = C2xV2 c=concentration & v=volume |
|
|
osmosis = movement of solvent particles |
Higher concentration = crenation lower concentration = hemolysis same concentration = isotonic |
|
|
alkane ending |
-ane |
|
|
aklene ending |
-ene |
|
|
alkyne |
-yne |
|
|
cis |
same side |
|
|
trans |
opposite |
|
|
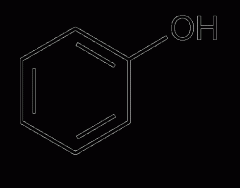
phenol |
|

