![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
5 Cards in this Set
- Front
- Back
|
Measuring the Rate of reaction |
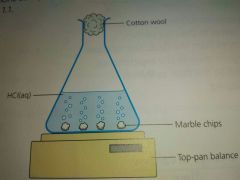
|
|
|
How can the rate if reaction be calculated? |
Rate of reaction= change of concentration/time Rate of reaction= change in quantity/ change in time |
|
|
What's the Collision theory? |
For a successful collision to occur, the reactants must collide with the correct geometry and possess a minimum energy before a successful reaction occurs |
|
|
Draw a diagram of an experiment on how gas can be collected |
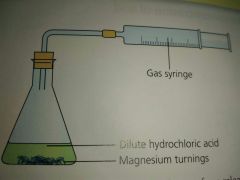
|
|
|
Concentration and pressure |
Increasing the concentration or pressure increases the rate of reaction because more particles are in the same place. As more particles are moving about, more collisions are likely to occur. If the particles colliding have suffient energy, a successful reaction will occur. |

