![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
24 Cards in this Set
- Front
- Back
|
Rate of reaction |
Change in concentration if a reactant or a product in a given time |
|
|
Rate = |
Change in concentration / change in time |
|
|
What's order? |
It shows how rate is affected by concentration Rate is proportional to the concentration raised to the power of the order |
|
|
Zero order? First order? Second order effects? |
● zéro - rate not affected by conc ● first - rate changes by same factor as conc change ● second - rate changes by the same factor as conc change raised to the power of 2 |
|
|
What's a half life? (In concentration time graphs) |
Time for the concentration to decrease by a half |
|
|
Zero order and first order conc time graphs |
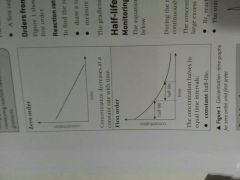
Zéro order - straight line downwards First order - curve downwards |
|
|
How can the concentration of a substance be monitored? |
Using a colorimeter |
|
|
How do you find the rate constant from a first order conc time graph? |
● measure gradient of tangent of curve (to find rate) and divide by the concentration corresponding to that time ● find the half life and do ln2/ t1/2 Where t1/2 = symbol for half life |
|
|
Explain the shapes of the 3 rate concentration graphs? |

Zero order - straight horizontal line 1st order - line through origin 2nd order - upwards curve through origin |
|
|
What does increasing temperature do on the rate constant? |
It increases the rate constant |
|
|
What's the rate determining step? |
The slower step in the reaction mechanism of a one step reaction |
|
|
With increasing temperature in an exothermic reaction what happens to the value of the equilibrium constant |
The equilibrium constant decreases so the of the product decreases as the equilibrium position shifts to the left |
|
|
With increasing temperature in an endothermic reaction what happens to the value of the equilibrium constant |
The value of the equilibrium constant increases to the aquarium position shifts the right |
|
|
What's a homogeneous equilibrium |
It contains equilibrium species that all have the same state |
|
|
What is a heterogeneous equilibrium |
It is an equilibrium that contain species with different states |
|
|
What is included in the Kc expression? |
Only gas or aqueous states |
|
|
What does a larger Kc value mean? |
It shows the equilibrium position being further to the right |
|
|
What affects the value of equilibrium constants? |
Température only. |
|
|
In an exothermic reaction, what happens to the Kc if you increase temperature? |
The equilibrium constant decreases since the rate of the backward reaction (endothermic) increases |
|
|
What happens when you increase the temperature of an endothermic reaction |
▪ it causes an increase in the Kc value ▪ the ratio of products/reactants must therefore increase to meet this new value of Kc ▪ the products increase ▪ the reactants decrease ▪ the position of equilibrium shifts right |
|
|
What happens when the concentration of a reactant is increased? |
▪ the ratio of products/reactants is now less than the Kc value (Kc value doesn't change!) And the system is no longer in equilibrium ▪ the reactants decrease ▪ the products increase ▪ the position of equilibrium shifts right |
|
|
If thé concentration of product increases, what happens? |
▪ the ratio of products/reactants is now more than the Kc value and so the system is no longer in equilibrium ▪ the products decease ▪ the reactants increase ▪ the position of equilibrium shifts left |
|
|
What happens when you change the pressure? |
It has a similar effect to changes in concentration where equilibrium shifts to side with less gaseous moles if pressure increases |
|
|
What does a catalyst do to the equilibrium constant |
It does nothing Since it only affects reaction rates, the position of equilibrium doesn't shift since the rate of the forward and backward reaction both increase by the same amount |

