![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
24 Cards in this Set
- Front
- Back
|
paraffins
|
a word sometimes used for alkanes
Latin for "little affinity" |
|
|
What does hν mean?
|
ultraviolate light
(where ν is the Greek letter nu) |
|
|
Alkanes show regular ______ in both boiling point and melting point as molecular ____ increases
|
increases; weight
|
|
|
dispersion forces increase as molecular _____ increases
|
size
This accounts for the higher melting and boiling points of larger alkanes |
|
|
stereochemistry
|
the branch of chemistry concerned with the three-dimensional aspects of molecules.
|
|
|
conformations
|
the different arrangements of atoms that result from bond rotation
|
|
|
conformers
|
(conformational isomers) -- molecules that have different arrangements
|
|
|
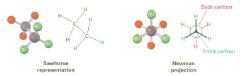
Newman projection
|
views the carbon-carbon bond directly end-on and represents the two carbon atoms by a circle
|
|
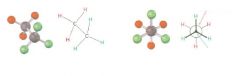
|
|
|
|
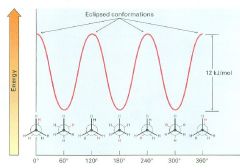
The lowest-energy, most stable conformer is the one in which:
This is called what in a Newman projection? |
all six C-H bonds are as far away from one another as possible
Staggered when viewed end-on in a Newman projection |
|
|
The highest-energy, least stable conformer is the one in which:
This is called what in a Newman projection? |
the six C-H bonds are as close as possible
Eclipsed in a Newman projection |
|
|
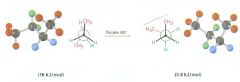
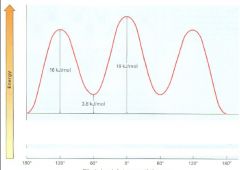
The extra 12 kJ/mol of energy present in the eclipsed conformer of ethane is called:
|
torsional strain
|
|
Fill in the Newmans!
|
|
|
|
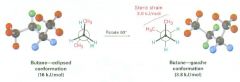
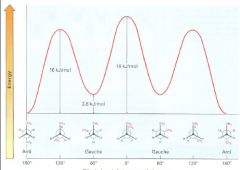
anti conformation
|
the lowest-energy arrangement
one in which the two methyl groups are as far apart as possible (e.g., in butane, 180 degrees away) |
|
|
gauche conformation
|
as a bond rotation continues, this energy minimum is reached at the staggered conformation where the methyl groups are 60 degrees apart
|
|
|
steric strain
|
the repulsive interaction that occurs when atoms are forced closer together than their atomic radii allow
result of trying to force two atoms to occupy the same space |
|
|
|
|
|
|
|

Which is more stable?
|

|
|
|
Make a graph of potential energy versus angle of bond rotation for propane, and assign values to the energy maxima
|
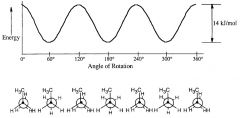
|
|
|
natural gas
|
primarily methane (but also contains ethane, propane, and butane)
|
|
|
petroleum
|
complex mixture of hydrocarbons
must be separated into fractions and then further refined before use |
|
|
engine-knock
|
an uncontrolled combustion that can occur in a hot engine
|
|
|
octane number (of fuel)
|
the measure by which its antiknock properties are judged
Straight chains have a higher tendency to knock e.g., heptane is terrible |

