![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
32 Cards in this Set
- Front
- Back
|
Whatare the masses and charges of these parts of an atom? · Proton · Neutron · Electron |
Protons= +ve (mass1) Neutrons= neutral (mass1) Electrons= -ve (mass1/2000th) |
|
|
Why do atoms usually haveno overall electrical charge? |
Atomsusually have no overall charge because they have equal numbers of protons (+ve)and electrons (-ve), which cancel one another out
|
|
|
Allatoms of a particular element (e.g. carbon) all have the same number of what?
|
Protons |
|
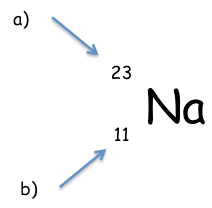
Whatare the following numbers called? |
a= relative atomic mass b= atomic number
|
|
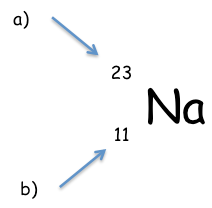
Whatdo the numbers tell you about the atom? |
a= relative atomic mass – the number of protons and neutrons in the atom
b= atomic number – the number of protons (usually the same as the number ofelectrons) |
|
|
Whatis an isotope?
|
Anisotope is an atom with a different amount of neutronsY9
|
|
|
Usethe periodic table to draw the electron structure of the following: -
· Neon · Calcium · Sodium Howcan you tell which group these atoms are in? |
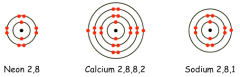
Thenumber of electrons in the outer shell is the same as their group number
|
|
|
Theelectron structure of sodium can be represented as 2,8,1 – what does this mean? Represent these atoms: -
· Fluorine · Oxygen · Potassium · Magnesium |
2,8,1means 2 electrons in the 1st shell, 8 electrons in the 2ndshell and 1 electron in the 3rd shell
Fluorine= 2,7 Oxygen= 2,6 Potassium= 2,8,8,1 Magnesium= 2,8,2 |
|
|
How many electrons canoccupy the 1st and 2nd shell (energy level) of an atom?
|
2in the 1st shell and 8 in the 2nd shell
|
|
|
Whathappens to the electrons of 2 atoms when they chemically react?
|
Duringa chemical reaction electrons are either given away, taken, or shared_ |
|
|
Definethese terms: -
· Element · Compound · Mixture |
Element– a pure substance made from 1 type of atom only Compound– 2 or more elements chemically bound Mixture– 2 or more elements or compounds mixed together which are not chemically boundlass9^f |
|
|
Whatis an ion? Howare +ve ions formed? Howare -ve ions formed?
|
Anion is an atom with a charge +veions have lost electrons -veions have gained electrons |
|
|
Explainhow the following atoms become ions (will they be +ve or –ve)?
· Potassium · Magnesium · Chlorine · Oxygen |
Potassium– loses 1 electron (K+) Magnesium– loses 2 electrons (Mg2+) Chlorine– gains 1 electron (Cl-) Oxygen– gains 2 electrons (O2-) |
|
|
Use diagrams to show howthese atoms for ionic bonds: -
Sodium + Chlorine |
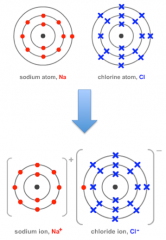
|
|
|
Describeand use a diagram to show the structure of ionic compounds
|
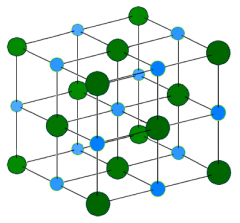
Ioniccompounds have a giant structure – they are bonded to 6 other ions byelectrostatic bonds
|
|
|
Whatare the melting and boiling points of ionic compounds like? Whyis this? |
Ioniccompounds have high melting points and boiling points – to melt them you haveto supply enough energy to break the 6 bonds attached to each ion |
|
|
Whathappens to the electrons when atoms form ionic bonds and covalent bonds? Whichis the strongest?
|
Ionicbonds – 1 atom loses electrons and another gains electrons
Covalentbonds – electrons are shared Anindividual covalent bond is stronger than an ionic bond |
|
|
Usediagrams to show how the following compounds are formed: -
· Hydrogen · Oxygen · Hydrogen Chloride |
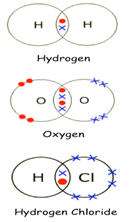
|
|
|
Usea diagram to show how water is formed
|
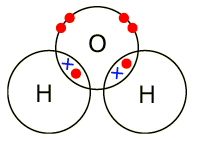
Water(H2O)
|
|
|
Howare the elements of the periodic table arranged?
Whatare the rows called? Whatare the columns called? |
Elementsare arranged in order of atomic number
Rowsare called periods Columnsare called groups |
|
|
What are the melting andboiling points of covalent compounds like? Explain why this is
|
Covalentcompounds have low melting and boiling points (are often gases) are there is noattraction between the molecules
|
|
|
Whydo elements in the same group have similar properties?
|
Elementsin the same group have similar properties as they all have the same number ofelectrons in their outer shell'>CE1
|
|
|
Which2 elements would be out of order in the periodic table if they were arranged byatomic mass instead of atomic number?
|
Potassiumand ArgonA
|
|
|
Whatare the symbols for these compounds?
· Calcium carbonate · Sodium chloride · Hydrochloric acid |
Calciumcarbonate – CaCO3
Sodiumchloride – NaCl Hydrochloricacid - HCl |
|
|
Why do covalent compoundsnot conduct electricity when dissolved?
|
Covalentcompounds do not conduct because they do no break down into ions
|
|
|
Givethe names of 3 covalent compounds which have giant ionic structures? Whatis the structure of each like?
|
Diamond– 4 carbon-carbon covalent bonds Graphite– 3 carbon-carbon covalent bonds Silicondioxide – 2 double silicon-oxygen covalent bonds |
|
|
Whydoes graphite conduct electricity?
|
Graphiteconducts electricity because it is only bonded 3x (meaning there is adelocalised electron to pass the charge)
|
|
|
Explainwhy ionic compounds have a giant structure
|
Ioniccompounds have giant structures because the ions keep their charge and attractup to 6 other ions – forming a giant lattice
|
|
|
Whatdoes monatomic mean and why are noble gases monatomic?
|
Monatomic means an atom which only exists on its own (does not react) Noblegases are monatomic as they have a full shell and are un-reactive
|
|
|
Whatdoes diatomic mean and why are elements such as the halogens, hydrogen andoxygen diatomic?
|
Diatomic– atoms in pairs, e.g. H2 and O2
Halogenswill form diatomic molecules because they covalently bond with each other,forming pairs |
|
|
What is the bonding inmetals like?
|
The bonding in metalscreates delocalised electrons (free to move and pass on electrical current)
|
|
|
Whydo metals conduct electricity?
|
|

