![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
197 Cards in this Set
- Front
- Back
|
What are some old drugs that are still around today (at least in some form)?
|
alcohol, opium, castor oil, nightshade, cinchona bark, foxglove, willow bark, cocaine
|
|
|
True or false: drugs have been on the market for only roughly a century?
|
True.
|
|
|
What can be said about the cost of drug discovery relative to the number of new drugs approved?
|
costs are rising but new drugs are falling
|
|
|
What percent of drugs will make it through Phase I?
|
70%
|
|
|
When did the FDA require new drugs to show efficacy?
|
1962 (after thalidomide)
|
|
|
What are the reasons a drug under discovery may be terminated (4)?
|
lack of desired level of efficacy, unacceptable toxicity, poor pharmaceutical properties, poor market potential/saturated competition
|
|
|
What are some specific examples of poor pharmaceutical properties that may lead to drug termination (5)?
|
instability, low aqueous solubility, poor cell, penetration, unacceptably high clearance
|
|
|
How many years do drug manufacturers have patent protection in Canada?
|
17 years of patent protection from the date on which the patent is issued or 20 years from the filing date, whichever term expires later
|
|
|
What is Phase I study characteristics? Who are the subjects?
|
first time drug used in humans. Usually investigated in healthy volunteers, establish PK profile, safety examination
|
|
|
Under what conditions would healthy volunteers not be used in Phase I trials?
|
chemotherapeutics. Patients will receive these in Phase I too.
|
|
|
What are the characteristics of Phase II trials?
|
drug testing in patients, but still has strict inclusion/exclusion criteria (can’t have any co-morbidities, or other medication). ‘Proof of concept’ study for efficacy.
|
|
|
What are the characteristics of Phase III trials?
|
thousands of patients, compared to placebo or standard of care, looking for safety and efficacy. Still have very strict inclusion/exclusion criteria.
|
|
|
True or false: Phase I and Phase II clinical trials have strict inclusion/exclusion criteria, but Phase III does not?
|
False. Phase III will also have strict inclusion/exclusion criteria, albeit not as strict as Phase I and Phase II.
|
|
|
What is the document that allows a pharmaceutical company to sell a drug in Canada?
|
Notice of Compliance (NOC)
|
|
|
What is the difference between a placebo and a nocebo?
|
placebo: therapeutic effect is observed from administering a sugar pill; nocebo: negative adverse effects are observed from administering a sugar pill
|
|
|
What is the body that reviews the pricing of newly approved drugs under patent protection?
|
Patented Medicine Prices Review Board (PMPRB)
|
|
|
True or false: all drugs sold in Canada, including radiopharmaceuticals, blood, and blood products, must have a drug identification number (DIN)?
|
False. All drugs sold in Canada with the EXCEPTION of radiopharmaceuticals, blood, and blood products, must have a drug identification number (DIN).
|
|
|
What are characteristics of Phase IV clinical trials?
|
not a formal process, mostly post-marketing surveillance for safety, represents a real world example (no inclusion/exclusion criteria anymore)
|
|
|
What is the difference between a cohort design study and a case-controlled study?
|
for cohort design, you follow a cohort with unknown effects (i.e. you don’t know the outcome). For case-control design, you know the outcome so you look to see if it’s correlated with the drug.
|
|
|
What document must be submitted to Health Canada to apply for approval of a generic drug? What must the generic drug company show about its drug?
|
abbreviated NDS; they must show that the pharmacokinetic profile of the drug is very similar to the trade name drug
|
|
|
What is the Special Access Program (SAP)?
|
a program that allows patients to access drugs generally unavailable for sale in Canada on the basis of compassionate grounds
|
|
|
With regards to DDI, what is it called when you take two drugs with the same active ingredient and what is an example?
|
duplications; e.g.Tylenol + NeoCitran
|
|
|
With regards to DDI, what is it called when you take two drugs with the physiological action and what is an example?
|
additive; e.g. propranolol + timolol (1+1=2)
|
|
|
With regards to DDI, what is it called when you take two drugs and interaction causes an increase in the effects of one or both of the drugs, and what is an example?
|
synergistic; ethanol + diazepam (1+1=3)
|
|
|
With regards to DDI, what is it called when you take two drugs whose effects act against each other, and what is an example?
|
antagonistic; e.g. morphine + naloxone
|
|
|
How can DDI lead to pharmacokinetic interactions?
|
ADME (absorption, distribution, biotransformation, excretion)
|
|
|
What are some ways that DDI can effect absorption of a drug (6)?
|
physicochemical interactions, gastrointestinal motility, changes in bacterial flora, mucosal damage, changes in gastric pH, drug transporters
|
|
|
What are the two main types of drug transporters (with respect to directionality of transport)?
|
uptake, efflux
|
|
|
What is the overall class, and what are some examples of uptake drug transporters (4)?
|
Solute-linked carrier (SLC) superfamily: OATs, OATPs, OCTs, OCTNs
|
|
|
What is the overall class, and what are some examples of efflux drug transporters (3)?
|
ABC superfamily: MDRs, MRPs, BCRPs
|
|
|
Which class of drug transporter has fruit juice been shown to inhibit?
|
OATP (uptake channel)
|
|
|
What drug is a substrate of the OATP drug transporter?
|
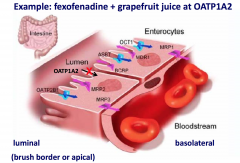
fexofenadine
|
|
|
P-gp is an efflux transporter. Which anatomical compartments will receive/not receive a drug that is a substrate for P-gp?
|
|
|
|
What are some examples of P-gp substrates (3+)?
|

|
|
|
What are some ways that DDI can effect the distribution of a drug (5)?
|
displacement from plasma proteins, displacement from tissue binding sites, changes in blood flow, alterations in local tissue barriers, interactions at drug transporters
|
|
|
Describe the DDI between clarithromycin and pravastatin?
|
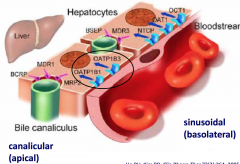
i) pravastatin is a substrate for OATP uptake into liver hepatocytes. ii) pravastatin uptake by OAPTs is inhibited by clarithromycin. iii) in the presence of clarithromycin, plasma levels of pravastatin increase and less reaches the hepatocytes, its therapeutic site of action
|
|
|
What adverse effect was observed in some young adults taking the antihistamine terfenadine and how was this caused?
|
Torsades de Pointes; terfenadine is a prodrug of fexofenadine and is biotransformed by CYP3A4. Inhibition of CYP3A4 by erythromycin or ketoconazole (or others) inhibits this conversion leading to high levels of terfenadine. Terfenadine leads to long Q-T interval and death.
|
|
|
What are two other examples of adverse DDI involving biotransformation?
|
i) failure of oral contraceptives caused by induction of CYP3A4 (antibiotics, anti-seizure medication). ii) reduced effect of warfarin due to induction of CYP2C9 by phenobarbital
|
|
|
What are some ways that DDI can effect excretion of a drug (3)?
|
pH (uncharged drugs can be reabsorbed), inhibition of drug transporters, induction of drug transporters
|
|
|
What drug can be co-administered with penicillin to decrease its excretion into the urine, and what is the mechanism of action?
|
probenecid; inhibits the renal tubular secretion of penicillin by blocking OAT1
|
|
|
What other application has probenecid inhibition of OAT1 been used for?
|
increasing the plasma concentrations of oseltamivir (active compound of Tamiflu)
|
|
|
How does taking St. John’s wort contribute to organ transplant rejection?
|
transplant patients are taking cyclosporine as an immunosuppressant. St. John’s wort induces both CYP3A4 and P-gp. This leads to lower concentrations of cyclosporine.
|
|
|
What are some examples of beneficial drug-drug interactions (5)?
|
elevate plasma concentrations, reverse of multidrug resistance, penetrate the blood‐brain barrier or maternal‐fetal barrier, prevent toxicity, treat overdoses
|
|
|
What categories do autonomic nerves belong to: CNS vs. PNS, sensory vs. motor, etc?
|
peripheral nervous system, motor, autonomic
|
|
|
What are the three types of motor neurons?
|
sympathetic, parasympathetic, somatic
|
|
|
For which motor neuron does the propagating signal reach the target without first passing through a ganglion?
|
somatic
|
|
|
Which neurotransmitter acts on skeletal muscle and which receptor is activated by binding of this neurotransmitter?
|
acetylcholine binds to muscular nicotinic receptors
|
|
|
What neurotransmitter signals between pre-ganglionic and post-ganglionic neurons for sympathetic and parasympathetic systems, respectively?
|
acetylcholine for both
|
|
|
What neurotransmitter signals between the pre-synaptic post-ganglionic neurons and their target for sympathetic and parasympathetic systems, respectively?
|
sympathetic: norepinephrine, acetylcholine (for sweat glands only); parasympathetic: acetylcholine
|
|
|
Which receptors are located on post-ganglionic neurons that receive the signal from preganglionic neurons?
|
nicotinic cholinergic receptors
|
|
|
From where in the CNS do sympathetic neurons arise? From where in the CNS do parasympathetic neurons arise?
|
Sympathetic: T1 through L2; parasympathetic: brain stem (CN III, VII, IX, X) and sacral (S2-S4)
|
|
|
What connects parasympathetic fibres arising from the sacral regions of the spinal cord to the pelvic plexus and descending colon?
|
pelvic splanchnic nerves
|
|
|
Between sympathetic and parasympathetic systems, which effects are more localized and why?
|
effects of the PNS tend to be more local than widespread because there are fewer number of synaptic connections than SNS. (Note: vagus nerve is an exception).
|
|
Fill in the blanks:
|
|
|
|
With respect to neuronal signalling, what potential drug targets exist (6)?
|
synthesis of neurotransmitter, storage of neurotransmitter, release of neurotransmitter, uptake of neurotransmitter, degradation of neurotransmitter, receptors (agonists and antagonists)
|
|
|
What drug blocks the transport of choline into cells and what is the effect (i)? What drug blocks the uptake of acetylcholine into synaptic vesicles (ii)? What drug blocks the release of acetylcholine from the nerve terminal (iii)?
|
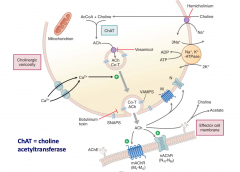
i) hemicholinium, leading to reduced ACh synthesis. ii) vesamicol. iii) botulinum toxin
|
|
|
What category of drugs bind directly to muscarinic/nicotinic receptors and what category of drugs bind to other targets with subsequent effects on the receptors?
|
direct-acting vs. indirect-acting
|
|
|
What enzyme converts choline to acetylcholine?
|
choline acetyltransferase (ChAT)
|
|
|
Where are M1 muscarinic receptors found and what are the effects at each location (4)?
|
CNS, autonomic ganglia, glands (gastric and salivary), enteric nerves; effect is to increase secretions
|
|
|
Where are M2 muscarinic receptors found and what are the effects at each location (4)?
|
CNS, heart, smooth muscle, autonomic nerve terminals; slows HR at SA node, slows conduction velocity at AV node, lowers force of contraction at myocardium
|
|
|
Where are M3 muscarinic receptors found and what are the effects at each location (4)?
|
CNS, smooth muscle, glands, heart; smooth muscle contraction (iris, bronchi, GI tract)
|
|
|
Which GPCR is coupled to the various muscarinic receptors?
|
M1, M3, M5: Gq. M2, M4: Gi/o
|
|
|
What is the biochemical mechanism of Gq GPCR pathway (5 points)?
|
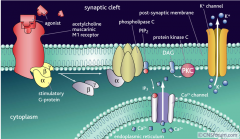
i) acetylcholine binds to M1 muscarinic receptor (or M3/M5). ii) Gq protein activates phospholipase C (PLC). iii) phosphatidylinositol phosphate (PIP2) is hydrolyzed to inositol triphosphate (IP3) and diacylglycerol (DAG). iv) IP3 releases stored calcium into the cytoplasm leading to smooth muscle contraction. v) DAG activates protein kinase C (PKC) leading to phosphorylation of various targets
|
|
|
What is the mechanism of action for M2 and M4 Gi-coupled receptors?
|
i) acetylcholine binds to M2 receptor and activates the Gi proteins. ii) Gi activation leads to inhibition of adenylate cyclase. iii) decreased activity of adenylate cyclase leads to a decrease in cytosolic cAMP
|
|
|
What are the effects of low cAMP on membrane ion channels and what is the overall result (2)?
|
i) activation of inwardly rectifying potassium channels. ii) inhibition of voltage-gated calcium channels; both of these lead to a hyperpolarized cell with suppressed cellular excitability. Note: a good way to remember this is to think of M2 receptor activation slowing down the heart (i.e. less excitable).
|
|
|
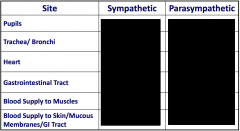
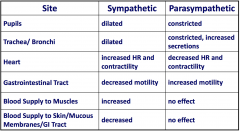
What are the effects of muscarinic receptor activation at the following organs: eye, secretory glands, heart, lungs, GI, gall bladder, urinary bladder?
|

|
|
|
Which types of neuronal receptors are found on the adrenal medulla?
|
neuronal nicotinic receptors
|
|
|
What is the mechanism of action that occurs due to acetylcholine binding on nicotinic receptors?
|
opening of the Na/K channels
|
|
|
What are the effects of nicotinic receptor activation at the following sites: autonomic ganglion, adrenal medulla, skeletal muscle?
|

|
|
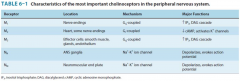
Here is a summary table.
|
M4 and M5 are the forgotten receptors.
|
|
|
Why is acetylcholine an unrealistic drug (3)?
|
not well absorbed, does not cross BBB, rapidly degraded by cholinesterases
|
|
|
What receptors does methacholine act upon and what is this drug used for clinically?
|
nonselective for muscarinic receptors; used in the diagnosis of bronchial hyper-reactivity and asthma
|
|
|
What is carbachol and what is it used for clinically?
|
muscarinic receptor agonist used locally for glaucoma to reduce intraocular pressure
|
|
|
What are some examples of nicotinic agonists (4)?
|
nicotine, lobeline, arecoline, succinylcholine
|
|
|
What is succinylcholine used for clinically and what is the mechanism of action?
|
can be used to induce paralysis by causing continuous depolarization of skeletal muscle
|
|
|
What are the physiological effects of nicotine (5)?
|
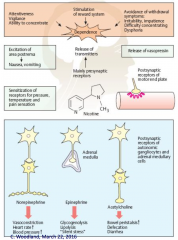
i) activation of both sympathetic and parasympathetic pathways. ii) release of norepinephrine within vascular walls of the heart. iii) release of epinephrine from adrenal medulla. iv) release of ADH. v) tremor and nausea through passage over BBB
|
|
|
What is varenicline used for and what is its mechanism of action?
|
used for smoking cessation; it is a partial nicotinic receptor agonist that can compete with nicotine. It reduced cravings and also decreases the pleasurable effects of smoking
|
|
|
What is the name of a reversible cholinesterase inhibitor that can cross the BBB? What is the name of a reversible cholinesterase inhibitor that cannot cross the BBB? What is the net result with respect to levels of acetylcholine at the synaptic cleft?
|
physostigmine; neostigmine (or pyridostigmine); this leads to higher levels of acetylcholine at the synaptic cleft
|
|
|
How come physostigmine can cross the BBB but neostigmine cannot?
|
neostigmine is a quaternary ammonium salt (charged) so it is not very lipophilic, while physostigmine is neutral and lipid soluble
|
|
|
What conditions can reversible cholinesterase inhibitors be used to treat (2)?
|
myasthenia gravis, Alzheimer’s disease
|
|
|
What is an example of an environmental toxin that can act as an irreversible acetylcholinesterase inhibitor?
|
organophosphates
|
|
|
What drug can be used in the acute treatment of organophosphate overdose?
|
pralidoxime (pyridine-2-aldoxime, 2-PAM)
|
|
|
What are three organophosphates?
|
echothiophate, sarin, parathion
|
|
|
What is the most common clinical use of cholinomimetics?
|
locally in the treatment of glaucoma
|
|
|
What is a classic feature of naturally occurring muscarinic receptor antagonists?
|
alkaloids, selective for muscarinic receptors over nicotinic
|
|
|
Which class of drugs is atropine, and what plant contains atropine naturally?
|
muscarinic receptor antagonist; deadly nightshade (Atropa belladonna)
|
|
|
Aside from atropine, which muscarinic receptor antagonist has CNS activity and which has no CNS activity?
|
CNS activity: scopolamine; no CNS activity: ipratropium, tiotropium
|
|
|
What is ipratropium used for?
|
used in the treatment of COPD
|
|
|
What is the difference between ipratropium and triotropium?
|
triotropium has less affinity for M2 receptors
|
|
|
Would you expect ipatropium to increase or decrease general secretions?
|
decrease. It is deactivated the parasympathetic system. In fact, dry mouth is the most common adverse effect.
|
|
|
What drugs act as selective antagonists for M3 receptors and what are these drugs clinically used for?
|
darifenacin, solifenacin; used in the treatment of overactive bladder
|
|
|
What drugs is NOT used in the treatment of Parkinson’s disease? a) benztropine. b) galantamine. c) procyclidine. d) trihexyphenidyl.
|
B. Galantamine is a reversible acetylcholinesterase inhibitor used in the treatment of Alzheimer’s disease (leads to greater cholinergic response). The drugs for treatment of Parkinson’s are muscarinic receptor antagonists (leads to less cholinergic response).
|
|
|
What is the primary clinical utility of nicotinic receptor antagonists?
|
non‐depolarizing (competitive) neuromuscular blockade
|
|
|
True or false: the nicotinic receptors in autonomic ganglia are not identical to those found at the neuromuscular junction?
|
True
|
|
|
How can the muscular paralytic effects of nicotinic receptor antagonists be reversed?
|
acetylcholinesterase inhibitors (leads to more ACh stimulation of nicotinic receptors)
|
|
|
How are neuromuscular blockers (i.e. nicotinic receptor antagonists) further classified?
|
duration of action (rapid, intermediate, long)
|
|
|
What is the suffix of neuromuscular receptor blockers?
|
curonium
|
|
|
How does the mechanism of non-depolarizing neuromuscular blockers differ at low doses compared to at high doses?
|
low doses: prevent depolarization of muscle cell membrane. high doses: can also enter the pore of the ion channel, making it more difficult to overcome by acetylcholinesterase inhibitors
|
|

|

|
|
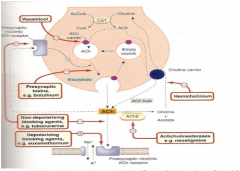
Here is a summary slide of drugable targets along the nicotinic pathway.
|

Here is a picture of the Jose Bautista bat flip
|
|
|
What neurotransmitters can be released by postganglionic sympathetic neurons, and what is the primary neurotransmitter of the sympathetic system?
|
norepinephrine (primary neurotransmitter), acetylcholine (for sweat glands), dopamine
|
|
|
Where are dopamine receptors of the sympathetic nervous system located?
|
renal vascular smooth muscle
|
|
|
Where are catecholamines synthesized (2)?
|
sympathetic nerve endings, chromaffin cells
|
|
|
What amino acid are catecholamines derived from?
|
tyrosine
|
|
|
What converts norepinephrine into epinephrine?
|
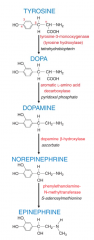
phenylethanolamine N-methyltransferase
|
|
|
How is tyrosine transported into neurons?
|
co-transported with sodium through the aromatic L-amino acid transporter
|
|
|
Which receptors are activated by norepinephrine and where are these receptors located?
|
on postsynaptic cell: alpha1, beta1, beta2; on presynaptic adrenergic neuron: alpha2
|
|
|
What class of drugs turn on the sympathetic nervous system (2)? What class of drugs turn off the sympathetic nervous system (3)?
|

|
|
|
What G protein are alpha1 receptors coupled to? What G protein are alpha2 receptors coupled to?
|
alpha1: Gq. alpha2: Gi
|
|
|
What G protein are beta1 receptors coupled to? What G protein are beta2 receptors coupled to? What G protein are beta3 receptors coupled to?
|
all beta receptors are coupled to Gs
|
|
|
Where are alpha1 receptors located? Where are alpha2 receptors located?
|
alpha1: vascular smooth muscles, glands, genitourinary smooth muscle, intestinal smooth muscle, heart, liver. alpha2: nerve endings, some smooth muscles
|
|
|
Where are beta1 receptors located? Where are beta2 receptors located? Where are beta3 receptors located?
|
beta1: cardiac muscle, juxtaglomerular apparatus. beta2: pulmonary smooth muscle, liver, heart. beta3: adipose cells
|
|
|
What are the physiological effects of alpha1 activation (6)?
|
contraction of vascular smooth muscle, contraction of genitourinary smooth muscle (sphincter contraction), relaxation of intestinal smooth muscle, increased inotrophy/chronotrophy of the heart, glycogenolysis/gluconeogenesis in liver, pupil dilation (mydriasis)
|
|
|
What are the physiological effects of alpha2 activation (4)?
|
decreased insulin secretion, platelet aggregation, decreased norepinephrine released from adrenergic nerves, contraction of vascular smooth muscle
|
|
|
What is the net cellular result of alpha1 activation (and Gq activation)?
|
increased intracellular calcium concentration (causing contraction and secretion)
|
|
|
What is the net cellular result of alpha1 activation (and Gi activation)?
|
decreased intracellular cAMP concentration (inhibit release of neurotransmitters and calcium)
|
|
|
What are the physiological effects of beta1 activation (3)?
|
increased inotrophy/chronotrophy, increased AV node conduction, increased renin secretion
|
|
|
What are the physiological effects of beta2 activation (3)?
|
smooth muscle relaxation (pulmonary, vascular, uterine), glycogenolysis and potassium uptake in skeletal muscle, glycogenolysis and gluconeogenesis in liver
|
|
|
What is the physiological effects of beta3 activation (1)?
|
lipolysis in adipose tissue
|
|
|
What is the net cellular result of beta1 activation (and Gs activation)? What is the mechanism?
|
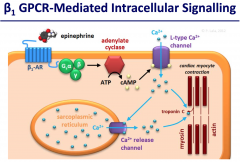
leads to activation of adenylyl cyclase and increased intracellular cAMP concentrations. This in turn leads to increased calcium concentrations because of the stimulatory action of cAMP on L-type calcium channels.
|
|
|
What is the net cellular result of beta2 activation (and Gs activation)? What is the mechanism?
|
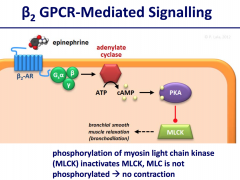
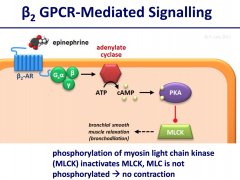
leads to activation of adenylyl cyclase and increased intracellular cAMP concentrations. This in turn leads to activation (phosphorylation) of protein kinase A (PKA). PKA inactivates myosin light chain kinase (MLCK), so myosin light chain is not phosphorylated. Net result is no contraction.
|
|
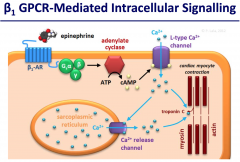
Convince yourself that high levels of cAMP lead to contraction in cardiac muscle (beta1 activation)…
|
but high levels of cAMP lead to smooth muscle relaxation in bronchi, uterus, etc. Depends whether L-type Ca channels are activated by high cAMP or if PKA is activated by high cAMP.
|
|
|
Norepinephrine (and all catecholamines) is an adrenergic agonist. Which receptors respond more strongly to norepinephrine?
|
in order of strength: alpha > beta1 >> beta2,3
|
|
|
What are the physiological responses of norepinephrine (2)?
|
increase in mean arterial pressure, decrease in HR (reflex bradycardia)
|
|
|
Describe the physiology of reflex bradycardia.
|
i) baroreceptors sense increased mean arterial pressure (MAP). ii) high MAP triggers vagus nerve activity and release of acetylcholine. iii) acetylcholine acts on M2 receptors to decreased HR
|
|
|
Which adrenergic receptors respond more strongly to epinephrine?
|
depends on the dose. At low doses beta effects predominate (vasodilation). At high doses, alpha effects predominate (vasoconstriction).
|
|
|
What are the physiological responses to epinephrine administration?
|

mixed response. Think of activating all adrenergic receptors.
|
|
|
What are the main effects of dopamine activation of D1 receptors?
|
vasodilation in peripheral renal, mesenteric, and coronary vascular beds
|
|
|
What drug is a selective alpha2 agonist and what is the physiological effect (2)
|
clonidine; CNS: inhibits excitatory outflow in regions that control cardiovascular activity. Peripherally: inhibit release of catecholamines from post-ganglionic sympathetic neurons via negative feedback system (due to presynaptic localization of alpha2)
|
|
|
What indirect agonist drugs can be used to increase norepinephrine release?
|
amphetamines, tyramine
|
|
|
What indirect agonist drugs can be used to decrease norepinephrine reuptake?
|
cocaine, tricyclic antidepressants
|
|
|
What is the mechanism of action for amphetamines?
|
they reverse the vesicular monoamine transporter (VMAT) leading to increased norepinephrine release
|
|
|
What is the mechanism of action for cocaine?
|
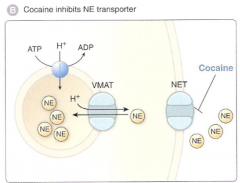
inhibits the norepinephrine transporter, reducing the reuptake or NE
|
|
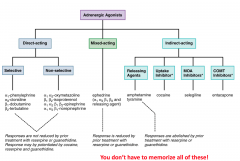
Here is a summary slide.
|
Remember that agonists can have opposite effects.
|
|
|
How can catecholamine activity be indirectly inhibited (5)?
|
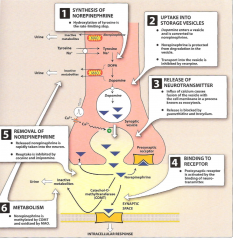
synthesis of NE, uptake of NE into storage vesicles, release of NE from presynaptic neuron, removal/reuptake of NE, metabolism of NE
|
|
|
What types of alpha-adrenergic antagonists can still exhibit their effects even when the drug is no longer present in the plasma?
|
irreversible antagonists (they can remain bound to the receptors)
|
|
|
Does antagonizing the alpha2-receptor lead to more or less norepinephrine release into the synaptic cleft?
|
it leads to more. Usually alpha-2 would prevent the release of norepinephrine through that negative feedback loop.
|
|
|
How does tamsulosin work in the treatment of benign prostatic hyperplasia? Why is this drug superior than a less selective one would be?
|
antagonizes the alpha-1A receptor leading to vasodilation of the smooth muscle of the prostate and urethra. Has less cardiac effects than other drugs because most cardiac receptors are alpha-1B
|
|
|
What class of drug is propranolol and what is the physiological effect (4+)?
|
nonselective beta-adrenergic antagonist; leads to decrease is inotropy/chronotropy, decrease cardiac output, peripheral vasoconstriction, bronchoconstriction, sodium retention, reduced glycogenolysis
|
|
|
How does nadolol differ from propranolol?
|
nadolol has a very long duration of action
|
|
|
Why would propranolol lead to sodium retention?
|
less renal blood flow means less filtration and ultimately excretion
|
|
|
Labetolol is a nonselective adrenergic antagonist that acts on alpha1, beta2 and beta2. What is labetolol used for clinically (2)?
|
i) treatment of pheochromocytoma. ii) treatment of hypertensive emergency
|
|
|
Why would selective beta1 antagonists be better than non-selective beta antagonists in the treatment of hypertension?
|
beta2 antagonists would cause bronchioconstriction, but beta1 antagonists do not
|
|
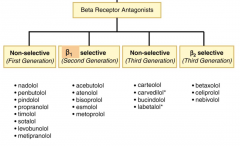
Here is a table showing the beta blockers organized by receptor target.
|
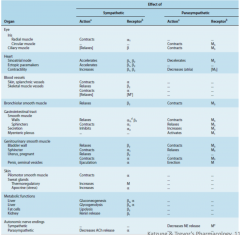
Here is a massive summary table of autonomic receptors and physiology
|
|
|
True or false: the cardiovascular system is made up of both the heart and the blood vessels?
|
True
|
|
|
True or false: many antiarrhythmic drugs are also proarrhythmic?
|
True
|
|
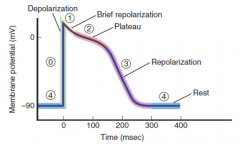
Here is a review of the cardiomyocyte action potential
|
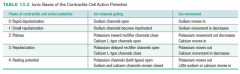
Here is an explanation of the 5 phases (0 through 4)
|
|
|
How does sympathetic activation of beta1 receptors lead to increased HR (i.e. what is the effect of beta1 activation on the SA node)?
|
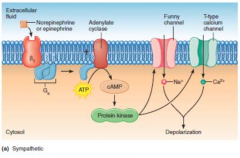
increased cAMP enhances the movement of sodium and calcium into the pacemaker cell
|
|
|
How does parasympathetic activation of muscarinic receptors lead to decreased HR (i.e. what is the effect of M2 activation on the SA node)?
|
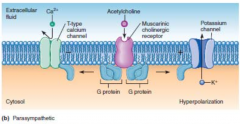
i) decrease movement of calcium and sodium (not shown) into the cell . ii) increases the movement of potassium out of the cell
|
|
|
What are the three major determinants of myocardial oxygen demand?
|
heart rate, contractility, vascular wall stress
|
|
|
True or false: preload is a function of venous tone while afterload is a function of arteriolar tone?
|
True
|
|
|
What two pathways are responsible for regulating vascular tone and what are the downstream effects of each initial substrate?
|
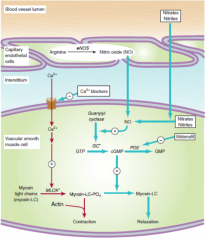
i) calcium, whereby high intracellular calcium concentrations leads to vasoconstriction. ii ) nitric oxide, whereby high NO concentrations lead to vasodilation
|
|
|
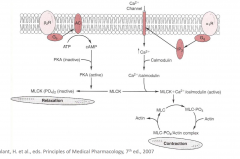
How does calcium entry into smooth muscle cell (due to depolarization) lead to contraction (3 points)?
|
i) calcium binds to calmodulin to form the calcium-calmodulin complex (Ca-CaM). ii) Ca-CaM activates MLCK which leads to phosphorylation of myosin light chains. iii) phosphorylated MLCs are capable of interacting with actin filament
|
|
|
How does activation of guanylate cyclase lead to smooth muscle relaxation (4 points)?
|
i) activated guanylate cyclase leads to high levels of cGMP. ii) cGMP activates cGMP-dependant protein kinase. iii) this kinase activates myosin light chain phosphatase which leads to dephosphorylation of MLC. iv) dephosphorylation of MLC inhibits the interaction of myosin with actin, leading to relaxation
|
|
|
How is nitric oxide related to the cGMP-mediated smooth muscle relaxation pathway?
|
nitric oxide activates guanylate cyclase, converting GTP to cGMP
|
|
|
What are the useful drug classes in the treatment of cardiovascular disease (including hypertension, angina heart failure, arrhythmias)? (7+)
|
diuretics, beta blockers, ACE inhibitors, ARBs, calcium channel blockers, vasodilators, antiarrhythmics (and hypolipidemics, anticoagulants, etc.)
|
|
|
What is the mechanism of action for thiazide diuretics?
|
inhibits the Na/Cl cotransporter in the DCT
|
|
|
What is the mechanism of action for loop diuretics?
|
inhibits NaK2Cl channels in the loop of Henle
|
|
|
What is the mechanism of action for spironolactone? What is another drug that has the same mechanism of action?
|
aldosterone antagonist; eplerenone
|
|
|
What is the mechanism of action for amiloride?
|
antagonist of sodium channels (ENaC) in the collecting tubules
|
|
|
What two classes of drugs used to treat hypertension have the side effect of cough, and what is the mechanism for both?
|
i) nonselective beta-adrenergic antagonists (bronchoconstriction due to beta2 inhibition). ii) ACE inhibitors (accumulation of bradykinin leading to pro-inflammatory factors)
|
|
|
Do ACE inhibitors and ARBs lead to increased or decreased levels of ADH release?
|
decreased. Even though you might expect an increase in ADH following reduction in blood pressure, angiotensin II stimulates ADH release, so you lose this pathway too.
|
|
|
What are the three classes of calcium channel blockers and what is an example of each?
|
i) diphenylalkylamines, verapamil, V site. ii) benzothiazepines, diltiazem, D site. iii) dihydropyridines, nifedipine, N site
|
|
|
Generally speaking, what are the two locations of L-type calcium channels?
|
heart, vascular smooth muscle
|
|
|
What two physiological situations manifest as angina? How do calcium channel blockers help to alleviate these symptoms?
|
increased oxygen demand of the heart or decreased oxygen supply; calcium channel blockers decrease oxygen demand by decreasing afterload and may also increase oxygen supply via dilation of the coronary arteries
|
|
|
Which subclass of calcium channel blockers has greater affinity for channels on the vascular walls? Which subclass of calcium channel blockers has greater affinity for channels in the heart?
|
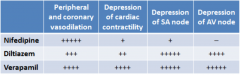
vascular wall: nifedipine. heart: verapamil
|
|
|
Which cells release nitric oxide?
|
endothelial cells
|
|
|
What classes of drugs would we use in the treatment of angina?
|
nitrates, calcium channel blockers, beta blockers
|
|
|
How are the nitrate class of drugs further subdivided?
|
short, intermedia and long duration
|
|
|
What are some limitations of nitrates (2)?
|
i) tolerance to the drug diminishes its clinical efficacy. ii) lots of contraindications
|
|
|
What volume distribution issue occurs from left ventricular heart failure? What volume distribution issue occurs from right ventricular heart failure?
|
left: pulmonary edema. right: peripheral edema
|
|
|
True or false: there are a large number of different agents that can be used to treat heart failure?
|
True.
|
|
|
What is the mechanism of action for digoxin (3 points)?
|
i) digoxin inhibits the Na/K ATPase leading to accumulation of intracellular sodium. ii) increased sodium concentration leads to decreased NCX activity (which pumps out calcium in exchange for sodium). iii) decreased NCX activity leads to an accumulation of intracellular calcium, increasing inotropy
|
|
Another summary slide.
|
let’s just treat with all of these drugs
|
|
|
What is the goal of an anti-arrhythmic drug (2)?
|
directly reduce the severity or prevent the occurrence of a cardiac arrhythmia
|
|
|
What is the precipitating factor leading to the development of most arrhythmias by number?
|
coronary heart disease
|
|
|
There are four classes of anti-arrhythmic drugs: Class I-IV. What is the mechanism of each class (4)?
|
Class I: sodium channel blockers. Class II: beta-receptor blockers. Class III: potassium channel blockers. Class IV: calcium channel blockers
|
|
|
What is an example of a sodium channel blocker and what is the electrophysiological action?
|
lidocaine; slows conduction velocity
|
|
|
What is the electrophysiological action of beta blockers?
|
anti-adrenergic action
|
|
|
What is the electrophysiological action of potassium channel blockers?
|
prolonged action potential duration and refractoriness
|
|
|
What is the electrophysiological action of calcium channel blockers?
|
heart rate slowing, slowing of AV nodal conduction
|
|
|
What specific conditions are antiarrhythmic agents most often used (4)?
|
i) prevent reoccurrence of atrial fibrillation/flutter. ii) prevent reoccurrence of supraventricular tachycardia. iii) as an adjunct in patients with implantable cardiac defibrillators. iv) treat ongoing arrhythmias
|
|
|
How can sodium channel blockers be further subdivided?
|
based on their speed of association/release from the channels
|
|
|
Propafenone and flecainide are Class 1c sodium channel blockers. They release slowly from sodium channels. Under what circumstances are these drugs particularly dangerous, and why?
|
dangerous in athletes when their heart rate increases to high levels. Every time the sodium channel opens, these drugs can enter and block the channel. If these channels are opening rapidly, there can be accumulated toxicity.
|
|
|
Sotalol is excreted by the kidneys. What adverse effect can occur in patients with kidney failure taking sotalol and why?
|
Torsades de Pointes. This is because sotalol can also act as a potassium channel blocker. Increased concentration due to poor excretion would lead to Torsades de Pointes.
|
|
|
What is the difference between ARP, RRP, and ERP?
|
Absolute refractory period: cannot initiate a new action potential. Relative refractory period: can initiate a new action potential, but it will be less effective (proper filling has not occurred). Effective refractory period: normal physiological time to reset
|
|
|
What are the differing effects of Class I antiarrhythmic drugs?
|
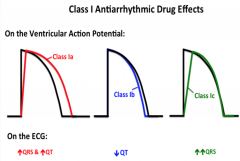
|
|
|
How do Class III antiarrhythmic drugs reduce the likelihood of reentry arrhythmias?
|
they prolong the refractory period so that the bypassed area cannot become stimulated on reentry
|
|
|
True or false: sodium channel blockers can make arrhythmias related to reentry circuits worse?
|
True.
|
|
|
What are two reasons why it is hard to assess efficacy of antiarrhythmic drugs in clinical trials?
|
i) arrhythmias are usually intermittent so it is hard to know whether the drug led to the result or if it would have happened anyways. ii) surrogate measures are hard to definitively correlate with outcomes
|
|
|
What is the most effective drug to use in preventing the reoccurrence of atrial fibrillation?
|
amiodarone
|
|
|
What are the most relevant risks of antiarrhythmic drugs by class (4)?
|
Class 1: sudden death (VT or VF). Class 2: bradycardia, fatigue. Class 3: Torsades de Pointes VT, sudden death. Class 4: worsening heart failure, bradycardia
|
|
|
What is meant by the term proarrhythmia?
|
tendency of antiarrhythmic drugs to facilitate emergence of new arrhythmias
|
|
|
True or false: it would have been better to be on a placebo than on encainide or flecainide during the 1991 clinical trial assessing incidence of sudden death?
|
True. There was a 3x greater risk of sudden death in the group taking the drugs.
|
|
|
What is the underlying cause of Torsades de Pointes (hint: can be seen on an ECG)?
|
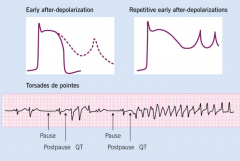
delayed action potential leads to prolonged Q-T interval. Because of the delayed action potential (specifically repolarization), cells fail to repolarize and begin to “flash” at 300 bpm
|
|
|
Which class of antiarrhythmic drugs can lead to Torsades de Pointes?
|
Class III (potassium channel blockers)
|
|
|
True or false: dose does not equal effect?
|
True.
|

