![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
150 Cards in this Set
- Front
- Back
|
Oppositely charged ions are attracted and held together in an ___________ bond. |
Ionic |
|
|
This type of chemical bond occurs between what two types of elements? |
A metal on the left-side of the periodic table and a non-metal on the right-side of the periodic table. |
|
|
Sometimes atoms will _________ their valence electrons to achieve 8 electrons in their outermost energy shell. |
Share |
|
|
This occurs between what two elements on the periodic table? |
A non-metal atom found on the right side of the right side of the periodic table, and another non-metal atom on the same right side of the periodic table. |
|
|
What is that bond called? |
Covalent bond. |
|
|
Sharing two pairs of electrons is called a _________ covalent bond. |
Double |
|
|
Sharing three pairs of electrons is called a _____________ covalent bond. |
Triple |
|
|
Put these covalent bonds from shortest to longest. |
Triple covalent bonds, Double covalent bonds, and then single covalent bonds. |
|
|
What is it called when electrons are equally shared between atoms? |
Non-polar covalent bond. |
|
|
What is it called when electrons are not equally shared between atoms? |
It is recognized as a polar covalent bond. |
|
|
What is an isotope? |
An isotope is different versions of the same element due to a different number of neutrons in the nucleus. |
|
|
Some isotopes of an element are unstable and ____________. |
Radioactive. (Cue music here... hahaha) |
|
|
Low levels of radiation is used in __________ medicine. |
Nuclear |
|
|
What is a bad thing about these medical tracers? |
They can harm cells, damage DNA, and cause cancer. |
|
|
Why must all chemical equations be balanced? |
It must comply with the law of conservation, meaning that matter cannot be destroyed or created. |
|
|
What is the pH scale? |
The pH scale is an inverse and logarithmic scale. |
|
|
What does the scale measure? |
The scale measures hydrogen ion concentrations. |
|
|
H+ |
This is a hydrogen ion. |
|
|
[ ] |
This means concentration. |
|
|
The lower the number [On the pH scale], the _________ the [H+]. |
higher |
|
|
The higher the number [on the pH scale], the ____________ the [H+] |
lower |
|
|
Each pH unit is a ten-fold ____________ or ___________ of the [H+]. |
increase...decrease |
|
|
Solutions with greater the amounts of hydroxide ions than hydrogen ions are classified as ______________. |
basic solutions |
|
|
Solutions with greater concentrations of hydrogen ions than hydroxide ions are classified as _______________. |
acidic solutions |
|
|
Solutions with an equal concentration of hydrogen ions and hydroxide ions are classified as ________________. |
neutral solutions |
|
|
What is Bromothymol Blue an indicator of? |
Acidity |
|
|
What is Phenolphthalein an indicator of? |
If it is basic or not. (HAHAH...) |
|
|
Why does water have such unique qualities? |
It is because of hydrogen bonding between the water molecules. |
|
|
Water has a __________ heat capacity. This is seen when water takes a long time to heat up and to cool down. |
high |
|
|
Water also has a _______ heat of vaporization. |
high |
|
|
Water also has a __________ surface tension. |
high |
|
|
What is cohesion? |
Water molecule sticking to each other. |
|
|
What is adhesion? |
Water molecules sticking to polar/other surfaces. |
|
|
Water is known as the universal __________ of life. |
solvent |
|
|
What is a solvent? |
A dissolving agent. |
|
|
What is a solute? |
A substance that has already dissolved. |
|
|
Solid water (ice) is _________ dense than liquid water because of ______________________. |
less...hydrogen bonding |
|
|
When water ionizes, it releases an equal number of ___________ ions and ____________ ions. |
negative...positive |
|
|
Name the prefix for each of the numbers:
1 _____________ 2 _____________ 3 _____________ 4 _____________ 5 _____________ |
1: mono- 2: di- 3: tri- 4: tetra- 5: penta- |
|
|
Name the prefix for each of the numbers: 6 _______________ 7 _______________ 8 _______________ 9 _______________ 10 ______________ |
6: hexa- 7: hepta- 8: octa- 9: nona- 10: deca- |
|
|
H2O |
Water |
|
|
O3 |
Ozone |
|
|
NH3 |
Ammonia |
|
|
NO |
Nitric Oxide |
|
|
NO2 |
Nitrous Oxide |
|
|
What are rows called on the periodic table? |
Periods. |
|
|
What are columns called on the periodic table? |
Groups or families. |
|
|
What is an element? |
A substance that cannot be broken down to other substances using ordinary chemical means. |
|
|
What is the atomic number of an atom? |
The number of protons the atom has. |
|
|
What is the atomic mass or mass number in an atom? |
The mass of the protons and neutrons in the nucleus. |
|
|
What is an ion? |
A charged atom with unbalanced # of protons to # of electrons. |
|
|
What is a cation? |
A positively charged ion. |
|
|
What is an anion? |
A negatively charged ion. |
|
|
How many electrons to each electron shell. |
2 in the first shell 8 in the second shell 18 in the third shell 32 in the fourth shell |
|
|
What are valence electrons? |
Electrons in the outermost shell. |
|
|
What do valence electrons determine? |
The chemical reactivity of the atom. (Only the valence electrons are involved in the bonding between atoms. |
|
|
What is the octet rule? |
All atoms need to have a full outer shell of 8 electrons to be stable. |
|
|
When atoms share electrons to achieve a full outer shell, what does it create? |
A covalent bond between the atoms. (Nonmetal + Nonmetal). |
|
|
When atoms give or receive atoms to achieve a full outer shell, what is it called? |
This is an ionic bond between the atoms. (Metal + Nonmetal). |
|
|
What do columns indicate in the periodic table? |
The number of valence electrons in the element's outermost shell. |
|
|
What do rows indicate in the periodic table? |
The number of electron shells in the element. |
|
|
Would changing the number of protons in the element change it into a different element? |
Yes. |
|
|
What are the six elements common to all life? |
Sulfur, oxygen, nitrogen, carbon, hydrogen, and phosphorus. |
|
|
Name P2O5. |
di-phosphorus pentoxide. |
|
|
Name N2O3 |
di-nitrogen trioxide |
|
|
Name CBr4 |
carbon tetrabromide |
|
|
What is the three carbon monomer of carbohydrate's chemical formula? |
C3H6O3 |
|
|
True or false, carbon atoms can bond together in straight chains, branched chains, or rings. |
True |
|
|
True or false, large molecules containing carbon atoms are called micromolecules. |
False |
|
|
True or false, polymers are formed by hydrolysis. |
False |
|
|
True or false, cells use carbohydrates for energy. |
True |
|
|
What is matter and give examples? |
Matter is anything that has mass and takes up space.
Gas Liquid Solid |
|
|
What is a compound? |
A substance consisting of two or more different elements combined in a fixed ratio. |
|
|
What are trace elements? |
Trace elements are essential to the human life but are needed in only minute quantities. |
|
|
What is an atom? |
The atom is the smallest unit of matter that retains properties from element. |
|
|
What is a proton? |
A proton is a subatomic particle with one positive charge. |
|
|
What is a electron? |
An electron is a subatomic particle with one negative charge. |
|
|
What is a neutron? |
A neutron is a subatomic particle with no charge. |
|
|
What is a nucleus? |
A nucleus is a combination of protons and neutrons tightly packed together to form the central core. |
|
|
What are reactants? |
The starting materials before the chemical reaction. |
|
|
What are products? |
The material resulting from the chemical reaction. |
|
|
What is thermal energy? |
Thermal energy is associated with the random movement of atoms and molecules. |
|
|
What is heat? |
Heat is thermal energy transfer from warmer to cooler bodies of matter. |
|
|
What is temperature? |
Temperature is the average speed of molecules in a body of matter. |
|
|
What is a solution? |
A liquid consisting of uniform mixture of two or more substances. |
|
|
What are buffers? |
Buffers are substances that minimize exchanged in pH. |
|
|
What are organic compounds? |
Organic compounds are carbon-based molecules. |
|
|
What are isomers? |
Isomers are compounds with the same chemical formula but different structural arrangement. |
|
|
What are hydrocarbons? |
They contain only hydrogen and carbon atoms. |
|
|
What are functioning groups? |
They are chemical groups that affect a molecule's function. |
|
|
Hydrophilic? |
Water-loving. |
|
|
Hydroxyl group? |
A hydrogen atom bonded to an oxygen atom.
-OH |
|
|
Carbonyl group? |
A carbon atom double bonde to an oxygen atom.
-C=O |
|
|
Carboxyl group? |
A carbon atom double bonded to an oxygen atom and also bonded to a hydroxyl group.
-C=O | OH |
|
|
Amino Group? |
Nitrogen bonded to two hydrogen atoms and a carbon skeleton.
H | -N-H |
|
|
Phosphat group? |
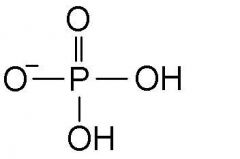
A phosphorous atom bonded to four oxygen atoms. |
|
|
Methyl Group? |
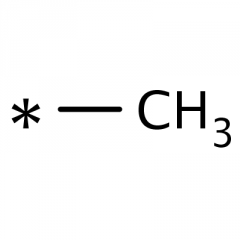
carbon bonded to three hydrogen atoms attached to a carbon skeleton. |
|
|
Macromolecules? |
gigantic molecules. |
|
|
polymers? |
small chains of molecules. |
|
|
monomers? |
building blocks of polymers. |
|
|
Dehydration reaction? |
Reaction that removes a molecule of water as two monomers bond together. |
|
|
Hydrolysis? |
Reaction that adds water to a polymer to break it up into two monomers. |
|
|
Enzymes? |
Specialized macromolecules that speed up chemical reaction in cells. (Protein) |
|
|
Carbohydrates? |
The class of molecules that range from small sugar molecules to large polysaccharides like starch molecules. |
|
|
Monosaccharides? |
Monomers of carbohydrates. |
|
|
Glucose? |
A common monosaccharide if central importance in the chemistry of life.
C6H12O6 |
|
|
What is a disaccharide? |
A disaccharide is two monosaccharides bonded together. |
|
|
Trans Fats |
Trans fats are harmful polysaccharides with detrimental effects on cardiovascular health. |
|
|
What are polysaccharides? |
Polysaccharides are macromolecules, polymers of hundreds to thousands of monosaccharides linked together by dehydration reaction. [poly saccharides are structural and storage molecules]. |
|
|
Starch? |
A storage polysaccharide in plants with long glucose monomers. |
|
|
Glycogen? |
A polysaccharide that stores glucose in animals. |
|
|
Cellulose? |
An abundant polysaccharide on earth that helps make up the tough outer wall in plant cells. |
|
|
What is chitin? |
Chitin is a structural polysaccharide that is used by insects and crustaceans to build exoskeletons. |
|
|
What are lipids? |
Lipids are a diverse group of molecules classified by a sole common trait, they do not mix well with water. |
|
|
Hydrophopic? |
Water-fearing. |
|
|
Fat? |
A large lipid made from two different kinds of smaller molecules: glycerol and fatty acids. |
|
|
What is an unsaturated fatty acid? |
An unsaturated fatty acid is a fatty acid with one or more double bonds. |
|
|
What is a saturated fatty acid? |
A saturated fatty acid is a fatty acid with no double bonds and thus has more hydrogen packed into it. |
|
|
What are phospholipids? |
Phospholipids are a major component of cell membranes. Only two fatty acids are connected to the glycerol. |
|
|
What are steroids? |
Steroids are lipids with a carbon skeleton that has four fused rings. Steroids are made from cholesterol. |
|
|
Cholesterol? |
Cholesterol is common in an animal cell membrane, it is the precursor of making other steroids. |
|
|
What is protein? |
Protein is a polymer of small building blocks of amino acids. |
|
|
Amino acids? |
Are composed with an amino group and a carboxyl group. |
|
|
What is a peptide bond? |
It is a bond between two amino acids with water removed. |
|
|
What is a polypeptide? |
A polypeptide is a chain of amino acids. |
|
|
What is a primary structure? |
It is a precise structure of amino acids in a sequence. |
|
|
What is a secondary structure? |
It is segments that coil and fold into patterns. |
|
|
What is a tertiary structure? |
The tertiary structure is the three dimensional shape. |
|
|
What is the quaternary structure? |
It is proteins with one or more polypeptide chains. |
|
|
What is a gene? |
It is a unit of inheritance. |
|
|
What is RNA |
Ribonucleic Acid It builds proteins out of intermediary nucleic acids. |
|
|
What are nucleotides? |
They are the monomers of nucleic acids. |
|
|
What is a double helix? |
A double helix is two polynucleotides that wrap around each other. |
|
|
What are some properties of carbon? |
It has four valence electrons It can form four covalent bonds It can be single, double, or triple bonded covalently with other atoms. |
|
|
What determines function? |
Shape. |
|
|
What is a keytone group? |
A keytone group is a carbonyl group in the center on the carbon skeleton. |
|
|
What is the function of carbohydrates? |
Short-term energy storage Quick fuel Structural polysaccharides. |
|
|
Monomers of Carbohydrates? |
glucose, fructose, and galactose. |
|
|
Disaccharides of carbohydrates? |
maltose, sucrose, and lactose. |
|
|
Energy storing polysaccharides? |
Starch -- Plant Glycogen -- Animal |
|
|
Structural polysaccharides? |
Cellulose -- plant cell wall Chitin -- fungal cell wall and exoskeleton of some arthropods. |
|
|
What is the function of lipids? |
Long-term energy storage insulation cushioning |
|
|
Examples? |
Fats (Animals) Oils (plants) |
|
|
What are two types of nucleic acids? |
DNA & RNA |
|
|
DNA is the molecule of ____________. |
Heredity. |
|
|
What are the nitrogenous bass for DNA |
A T G C |
|
|
RNA is _____________ stranded. |
Single |
|
|
What are the nitrogenous bases for RNA? |
A U G C |
|
|
How to make nucleotide? |
5-C sugar Nitrogenous base phosphate group |
|
|
ATP? |
Energy currency of the cell. Needed for all cell work. |

