![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
16 Cards in this Set
- Front
- Back
|
Definition: Lewis electron-dot structures |
A drawing that represents chemical bonds between atoms as shared or transferred electrons (lines) and valence electrons around those elements as dots |
|
|
Definition: Octet |
Eight dots around an atom. Means it has a full outer layer |
|
|
Definition: Duet |
Two dots around an atom in a Lewis structure. Helium has this and is stable. |
|
|
Definition: Octet rule |
The bonding of atoms so they have a stable configuration, which usually means they have eight electrons in their outermost shell. |
|
|
Electron configuration for ionic bonds |
1. Identify the number of valence electrons for each atom 2. For the metal, take away the number of electrons that will make it isoelectronic with the previous noble gas. 3. Adjust the ratio of metal(s) to nonmetal(s) so that the electrons taken from the metal(s) fill the outer shell of the nonmetal(s) (octet rule) |
|
|
Example for Lewis structure of covalent bonds: Bonding of H2O |

|
|
|
Definition: Bonding pair |
A shared pair of electrons (represented with a line) |
|
|
Definition: Lone pair/nonbonding electrons |
A pair of electrons associated with only one atom |
|
|
Definition: Double bond |
When two electron pairs are shared between two atoms. (Triple bond is three bonds between two atoms) |
|
|
Lewis structure of cations |
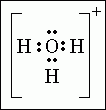
Remove one electron from the group for each positive charge. Ex: H3O+ has 9 total protons and 8 electrons (follows octet rule) |
|
|
Lewis structure of anions |
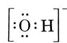
Add one electron to the group for each negative charge. Ex: OH- has 7 protons and 8 electrons |
|
|
Definition: Polar covalent bond |
Intermittent between a pure covalent bond and an ionic bond. The electrons are shared, but unevenly. |
|
|
Notation of a polar bond |
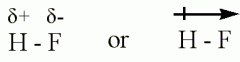
F is more electronegative, and has a more negative charge |
|
|
Definition: Electronegativity difference |
Symbol: ΔEN The difference between the electronegativity values of covalently bonded atoms |
|
|
Bond types by electronegativity difference |
ΔEN between 0-0.4 is covalent ΔEN between 0.4-2.0 is polar covalent ΔEN 2.0+ is ionic |
|
|
The type of bond formed by identical atoms or atoms with close electronegativities |
Nonpolar covalent bonds |

