![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
80 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
How many carbon atoms does the hydrocarbon 2-butene contain? -2 -4 -6 -8
|
4 |
|
|
|
Which law relates the volume, pressure, temperature, and number of moles of a gas? -Avogadro's law -Combined gas law -Gay-Lussac's law -Ideal gas law
|
Ideal gas law |
|
|
|
In a reversible chemical reaction represented A+B<-->AB, how would adding more of substance A to system affect the equilibrium? -shift left -shift right -no effect |
|
|
|
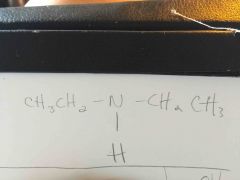
|
C. Amine
|
|
|
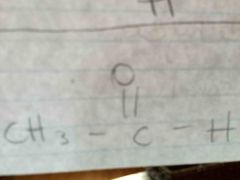
|
B. Aldehyde |
|
|
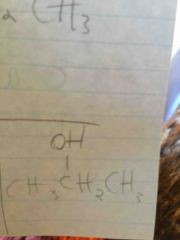
|
D. Alcohol
|
|
|

|
C. |
|
|
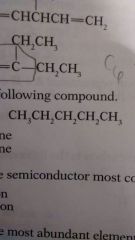
|
C. Pentane |
|
|
|
What is the semiconductor most commonly used to make computer chips? |
silicon |
|
|
|
What is the most abundant element in the universe?
|
hydrogen
|
|
|
|
What element is ozone a form of? |
Oxygen |
|
|
|
What is the pH of 1x 10(⁻³) M solution of HCl? |
3 |
|
|
|
What is the oxidation number of carbon in the iron ore siderite (FeCO₃)? |
+4 |
|
|
|
What type of substance consists of two or more elements chemically combined? |
Compound |
|
|
|
What group of lipids includes cholesterol, bile salts, and vitamin D?
|
steroids
|
|
|
|
rubbery polymers made from silicon-oxygen chains rather than carbon chains? |
Silicones |
|
|
|
Biological polymers composed of long chains of sugars? |
|
|
|
|
protein catalysts found in living things? |
Enzymes |
|
|
|
substituted hydrocarbons containing one or more hydroxyl groups (-OH)? |
alcohols |
|
|
|
ionic compounds composed of any cation except H+ and any anion except OH-? |
Salts |
|
|
|
compounds with the same molecular formula but different arrangements of atoms? |
Isomers |
|
|
|
sodium or potassium salts of fatty acids? |
Soaps |
|
|
|
hydrocarbons consisting of rings containing delocalized electrons? |
Aromatic hydrocarbons |
|
|
|
hydrocarbons characterized by one or more triple bonds between carbon atoms? |
Alkynes |
|
|
|
alkanes and other organic compounds consisting of only hydrogen and carbon? |
Hydrocarbons |
|
|
|
The outermost electrons in an atom, which are involved in chemical bonding, are called the atom's ________ electrons. |
valence |
|
|
|
The complex mixture of liquid hydrocarbons found in the earth's crust is called ________? |
Petroleum |
|
|
|
The reactions of alkanes and other hydrocarbons with oxygen to produce CO₂, H₂O, and heat is called ________? |
Combustion |
|
|
|
The study of chemical compounds containing carbon is called _______? |
Organic chemistry |
|
|
|
An atom that gains electrons in a chemical reaction is said to undergo _________? |
Reduction |
|
|
|
The minimum kinetic energy that a molecule must possess to undergo a chemical reaction is called the _______ energy. |
Activation |
|
|
|
A distance of 3.2 km is equal to ______m. |
Move decimal 3x to the right 3,200 meters |
|
|
|
If the forward and reverse reactions of a reversible chemical reaction are proceeding at the same rate, then the system is said to be in a state of chemical ____________. |
Equilibrium |
|
|
|
Plastic and other materials made from many small molecules that have been linked together to form much larger molecules are called _____________? |
Polymers |
|
|
|
The reaction HNO₃ aq + NaOH(aq)----> H₂O(L) + NaNO₃ (aq) is a _____________ reaction? |
|
|
|
|
An atom of a particular element is distinguished from atom of other elements by the number of ________ in the nucleus. |
Protons |
|
|
|
protein molecules consist of long chains of smaller molecules called _______? |
Amino acids |
|
|
|
Nuclear radiation that consists of high-speed electrons is called _______ radiation. |
Beta |
|
|
|
Elements with the largest atoms are located in the ________ region of the periodic table. |
Lower left |
|
|
|
The elements helium, argon, and xenon belong to the group of elements known as the __________? |
Noble gases |
|
|
|
What is the functional group of aldehydes? |
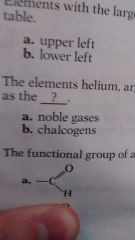
A |
|
|
|
Nylon a common name for a group of polymers called __________? |
|
|
|
|
Citric acid, acetic acid, and lactic acid are examples of _____________? |
|
|
|
|
An important 6-carbon sugar found in both animals and plants is ________? |
Glucose |
|
|
|
More compounds are formed from ________ than from all other elements combined. |
Carbon |
|
|
|
DNA and RNA are polymers of ________? |
Nucleotides |
|
|
|
The strong intermolecular force responsible for the unexpectedly high boiling points of water, ammonia, and hydrogen fluoride is the _________. |
Hydrogen bond |
|
|
|
As a rule of thumb, the rate of a given chemical reaction doubles when the temperature is increased by __________? |
|
|
|
|
A chemical bond characterized by the sharing of one or more electron pairs between two atoms in a molecule is called a __________? |
|
|
|
|
A substance that dissociates or ionizes in water to produce ions that can conduct an electric current is called an ELECTRODE. |
Electrolyte |
|
|
|
Chemical reactions and physical changes that release heat to their surroundings are ENDOTHERMIC processes. |
|
|
|
|
The process of oxidation in an electrochemical cell always occurs at the ANODE. |
True |
|
|
|
Electrons that circulate freely throughout a molecule instead of being bound to a single atom or pair of atoms are said to be DELOCALIZED. |
True |
|
|
|
Kinetic energy is energy associated with an object's MOTION. |
True |
|
|
|
A list of metals arranged by their relative ease of oxidation is an OXIDATION series. |
Activity |
|
|
|
The study of the chemistry of living things is called BIOCHEMISTRY. |
True |
|
|
|
A substance that produces hydroxide ions when dissolved in water is an ACID. |
Base |
|
|
|
The shorter the half-life of a radioactive substance, the GREATER its activity. |
true |
|
|
|
compounds that contain the functional group --N-- and can be thought of as derived from ammonia are called ________? |
|
|
|
|
The second law of thermodynamics states that ______? |
|
|
|
|
The elements lithium, sodium, potassium, and cesium belong to the group of elements know as the _______ metals. |
Alkali |
|
|
|
The amount of fissionable material at a given density required to sustain a chain reaction is known as the __________? |
Critical mass |
|
|
|
The precious metal, more expensive than gold, that is commonly used in industrial and automotive catalysts is _________? |
|
|
|
|
The uncertainty principle states that.......?
A. The position and momentum of an electron may be precisely described using four quantum numbers B. When a p,d,or f sublevel is being filled, each orbital is filled singly before pairing begins C. No two electrons in an atom can have identical quantum numbers D. The position and momentum of small particles such as electrons cannot be precisely measured simultaneously |
D. The position and momentum of small particles such as electrons cannot be precisely measured simultaneously. |
|
|
|
A triglyceride is a(n) ___________ molecule. |
Fat |
|
|
|
Hydrocarbons containing one or more double bonds usually have names ending in _________. |
|
|
|
|
A mixture containing tiny clumps or particles that remain suspended and do not settle out over time is a ___________. |
Colloid |
|
|
|
Sulfur is extracted from petroleum and natural gas using the _______process. |
Claus |
|
|
|
If a strip of blue litmus paper turns red when dipped into a solution, the pH must be ______? |
Less than 7 |
|
|
|
A nuclear reaction in which a single large nucleus splits into two smaller nuclei is __________? |
Nuclear fission |
|
|
|
The concentration of a solution in terms of moles of solute per liter of solution is known as ________? |
|
|
|
|
When a carboxylic acid reacts with an alcohol, a(n) ________ is formed. |
B. Ester |
|
|
|
Individual "packets" or quanta of light are called _________? |
photons |
|
|
|
write the formula for phosphorus trichloride. |
PCl₃ |
|
|
|
write the name of MgSO₄. |
|
|
|
|
Write the equilibrium constant expression for the reaction H₂SO₄(aq)+ CaF₂(s) <-------->2 HF(g)+ CaSO₄(s). |
|
|
|
|
Calculate the molar mass of carbon tetraflouride (CF₄) in grams per mole, rounding to proper significant figures, if M(c)= 12.01 u and m(f)= 19.00u. |
88.01 grams per mol |
Page 54 |
|
|
Balance the following equation: Fe₃O₄(s)+H₂----> Fe(s)+ H₂O (liquid) |
Fe₃O₄(s) + 4H₂ --> 3Fe(s) + 4H₂O(l)
|
|
|
|
Methane, the chief component of natural gas, burns in oxygen according to the equation CH₄(g) + 2O₂(g)------->CO₂(g) +2 H₂O(g) If 7.0 mol of CH₄ react with an unlimited amount of oxygen, what amount (in moles) of H₂O (g) will be produced? |
14 moles of H₂O |
|
|
|
What volume of air at 25 degrees C and 1.00 atm can be stored in a 10.0 L high-pressure air tank if compressed to 25 degrees C and 175 atm. |
Either will |
|

