![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
24 Cards in this Set
- Front
- Back
|
Define a nucleophile |
An atom or group of atoms which is attracted to an electron deficient centre or atom, where it donates a pair of electrons to form a new covalent bond |
|
|
Define an electrophile |
An atom or group of atoms that which is attracted to an electron rich centre or atom, where it accepts a pair of electrons to form a new covalent bond |
|
|
What are examples of some nucleophiles? |
Water molecules, H2O: Hydroxide ions, :OH- Ammonia molecules, :NH3 |
|
|
How do you convert an alkene to an alcohol |
Using H3PO4 and Steam (H2O) Hydration reaction |
|
|
How do you convert an alcohol to an alkene |
Using concentrated H3PO4 Dehydration reaction |
|
|
How do you get from an alkene to a dihaloalkane? |
Using a halogen Electrophilic addition reaction |
|
|
How do we convert an alkene into an alkane |
Using H2 and a nickel catalyst 150C Hydrogenation ( a type of addition reaction ) |
|
|
How do we convert an alkane into a haloalkane? |
G UV and a halogen Halogenation |
|
|
How do we convert a haloalkane into an alcohol? |
Ethanol solvent NaOH-/ or KOH Nucleophilic addition |
|
|
How do we convert an alcohol into a haloalkane? |
Nucleophilic substitution H2SO4 Na Halide |
|
|
How do you convert an alkene into a haloalkane |
Using a hydrogen halide Electrophilic addition (halogenation) |
|
|
Give an example of a electrophilic addition reaction |
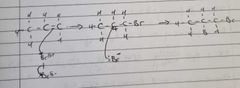
The electron pair in the c=c goes to the Br (it's an electrophile - accepts the electron pair)
Halogen bonds to the carbon in the double bond with the most Hydrogens already bonded to it |
|
|
Give an example of nucleophilic substitution |
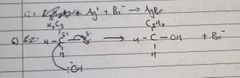
Charges on the c and halogen Nucleophile attacks on side opposite of the halogen Nucleophike donates an electron pair to the carbon Halogen gets booted off |
|
|
How do we get from a primary alcohol to an Aldehyde |
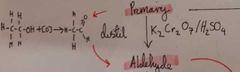
Distil Use K2Cr2O7 and H2SO4 |
|
|
How do we go from an Aldehyde to a carboxylic acid? |
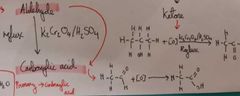
Reflux K2Cr2O4 /H2SO4 |
|
|
How do we go straight from a primary alcohol to a carboxylic acid? |
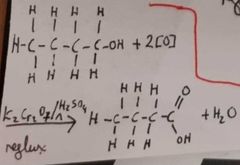
|
|
|
How do we go from a secondary alcohol to a ketone |
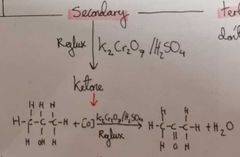
Reflux K2Cr2O7 / H2SO4 |
|
|
What is the functional group for Aldehydes? |
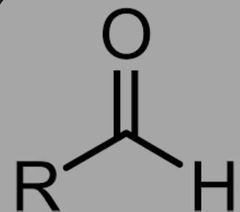
|
|
|
What is the functional group for ketones? |
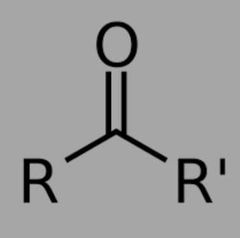
|
|
|
What are the different ways you can get a haloalkane? |
Alcohol to haloalkane Alkene to haloalkane Alkane to haloalkane Alcohol to alkene to haloalkane Alkene to alcohol to haloalkane Alcohol to alkene to alkane to haloalkane |
|
|
What are the different ways to grt an alcohol |
Haloalkane to alcohol Alkene to alcohol |
|
|
How do we go from an alkane to an alkene |
Dehydrogenation |
|
|
How do we go from a dihaloalkane to an alkene |
Dehalogenation |
|
|
How to go from haloalkane to alkene |
Elimination reaction |

