![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
86 Cards in this Set
- Front
- Back
|
The Scientific Method |
Purpose Hypothesis Procedure Experiment Observations Conclusions |
|
|
Scientific Notation |
When adding or subtracting with scientific Notation. Make sure that the powers of 10 remain the same and the other numbers are the ones subtracted. When multiplying, add the two powers of 10 and multiply the other numbers When dividing, subtract the powers of 10 and multiply the other numbers |
|
|
Significant figures |
Addition and Subtraction: digits after decimal Multiplication and Division : all digits |
|
|
What is chemistry? |
The study of all matter. |
|
|
How is the periodic table organised 9 |
Periods are Rows Groups are columns Organised by atomic number (number of protons in the nucleus) |
|
|
What are the alkali metals? |
Most reactive Very conductive Group 1 in the periodic table Shiny |
|
|
What are the alkaline earth metals? |
Less conductive than Alkali Less reactive than alkali Dull Used in fireworks Group 2 on the periodic table |
|
|
Metalloids |
Group of solids Semi-Conductors Make a staircase between non metals and metals |
|
|
Halogens |
Very reactive gases except Bromine (liquid) Group 17 in the periodic table Not very conductive Fairly brittle when solid |
|
|
Noble Gases |
All are gases Not very conductive Nor very reactive Colourless and odourless Most stable valence electrons Group 18 in the periodic table Brittle Non ductile Non malléable |
|
|
Difference between physical and chemical properties |
A physical property looks at whatever you can see already without making the element react. I. E: colour, taste, malleability, ductility Chemical properties are reactions with other things I. E:flammability, reaction with acid, reaction with water |
|
|
Quantitative and Qualitative Properties |
Quantitative have numerical value and are measured usually through measuring tools Example : mass, volume, Density Qualitative are measured using the senses and have no numerical value: colour, taste, smell |
|
|
Physical and Chemical Changes |
Physical changes are when no other substances are created. Often reversible Dissolving and Change of state are physical Chemical changes are when 1 or more new substances are created Often unreversible |
|
|
Clues to finding a chemical change |

|
|
|
Pure substance |
Contains only kind of particles Either an element or a compound Can't be separated by physical means |
|
|
Elements and Compounds |
Éléments Composed of identical atoms Found on periodic table Compounds: Made of two or more different elements in a fixed proportion Ex: salt (NaCl) |
|
|
Mixtures |
Variable combination of 2 or more pure substances |
|
|
Homogeneous |

Aka Solutions Only one visible phase (dissolved substance in solvent |
|
|
Heterogenous |

Mixture with two or more visible phases |
|
|
Atoms |
Building blocks of all matter. Made up of subatomic particles which include Protons, Electrons and Neutrons |
|
|
Proton |

|
|
|
Electron |

|
|
|
Electrons |

|
|
|
Chemical symbols |
Tells what atoms are Short form ( C=Carbon) Sometimes in Latin short form Always a capital letter, sometimes follow by one or more lower case letters |
|

|
Biohazardous infectious materials |
|

|
Harmful or Fatal |
|

|
May cause fire |
|

|
Flammable |
|

|
Health Hazard |
|

|
Harmful to environment |
|

|
Harmful |
|

|
Gas under pressure |
|

|
Causes severe skin and eye damage |
|

|
Explosion hazard |
|
|
What is Astronomy? |
The study of what is beyond the earth |
|
|
The universe |
Everything that exists, including all energy, matter and space |
|
|
Constellations |
Groups of stars that seem to form shapes or patterns Many are names after animals, therefore called constellations |
|
|
Solar System |
The Sun and all the objects that travel around it, including all the 8 known planets, the moons, of those planets and some planetoids |
|
|
Order of the planets |

|
|
|
Non luminous objects |
Do not emit their own light such as planets and moons The sunlight reflects off of them |
|
|
Luminous |
Objects that do emit their own light, such as the sun |
|
|
Stars |
Massive collection of gases ; held together by its own gravity and emitting huge amounts of energy (heat and light) Appear to twinkle |
|
|
Planets |
A large round celestial object that revolves around a star |
|
|
Moons |
Are satellites. Celestial objects that travel around a planet |
|
|
Asteroid |
Asteroid: small rocky objects. Many orbit around the sun and this is called an asteroid belt |
|
|
Meteoroid |
Small piece rock/metal that gets pulled to Earth |
|
|
Meteor |
Burns up in the earth's atmosphere |
|
|
Meteorite |
Hits the earth's surface causing a massive explosion |
|
|
Comets |
Chunk of frozen gases, rock and dust that travels in a very long orbit around the sun Some have regular orbits and can be seen at regular intervals Tails come from burning by the son |
|
|
Galaxies |
à HUGE collection of gas, dust and billions of stars, planets and other celestial objects |
|
|
The Sun |

|
|
|
Orbits |
Elliptical paths around a much larger object such as the sun |
|
|
Orbital period |
The time it takes to complete to complete revolution around the the sun. I. E: 365 1/4 for Earth |
|
|
Inner Planets |
Closest to the sun Termed terrestrial planets Made of minerals and rocky materials |
|
|
Mercury |
Closest to the sun Rarely seen, sun blocks the view Surface is smooth but covered in craters Day temp :400 degrees Celsius -180 degrees Celsius |
|
|
Venus |

|
|
|
Earth |

|
|
|
Mars |

|
|
|
Outer planets |
4 largest Contain mostly hydrogen and helium and are called the Gas Giant Lack solid surfaces Pluto used to be considered a planet but is no longer |
|
|
Jupiter |

|
|
|
Saturn |

|
|
|
Uranus |

|
|
|
Neptune |

|
|
|
Pluto |

|
|
|
Solar system calculations |
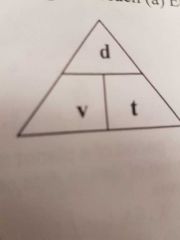
|
|
|
Old idea |
Earth centered universe Stars are just beyond planets Tranistion : 500 years ago telescope invented experiments conducted |
|
|
Modern idea |
Sun centered universe . Sun is one of many stars The milky-way Galaxy |
|
|
Rotation |
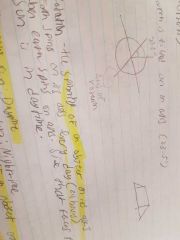
|
|
|
Revolution |
Movement of an object around another object |
|
|
Moon dance |
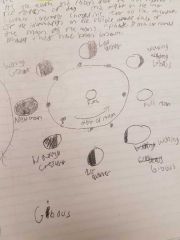
|
|
|
Stage 1 : Nebulas |
Composed of gases (hydrogen and helium) and dust Gas and dust particles swirl around and contract under their own gravity |
|
|
Stage 2 : Protostar |

|
|
|
Stage 3 Nuclear Fusion |

|
|
|
Stage 4 : old age |

|
|
|
What is Ecology |

|
|
|
Abiotic factor |
Non living things Light, heart, water, soil, wind Atmosphere and landscape. |
|
|
Biotic factors |
Planets, animals, microorganisms in soil Two main types of ecosystems 1. Terrestrial 2. Aquatic |
|
|
Ecology Hierarchy |
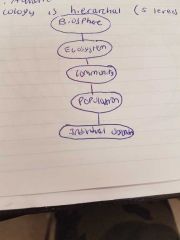
|
|
|
More on Hierarchy |

|
|
|
Competition |
Interaction between two or more organisms competing for the same resource in a given habitat. Competition can occur between members of the same species |
|
|
Predation |

|
|
|
Mimicry |
Species looks like another species |
|
|
Symbiosis |
Close interaction between different species live in, on or near members of another species |
|
|
Mutualism |
Both species benefit from the symbiotic partnership |
|
|
Commensalism |
Benefits from relationships with another species with no harm or benefit to the other species |
|
|
Parasitism |
When one species |

