![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
13 Cards in this Set
- Front
- Back
|
Which of the following elements in NOT responsible for a significant portion of the mass of living organisms? |
S |
|
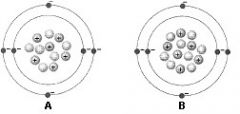
Use the figure to answer the corresponding question: The difference between the two atoms in the figure is? |
The number of neutrons |
|
|
The cohesiveness between water molecules is due largely to |
hydrogen bonds |
|
|
In living things, hydrogen bonds typically form between hydrogen and nitrogen or hydrogen and oxygen. What property must oxygen or nitrogen have for this bond to occur? |
They must have a partial negative charge |
|
|
A solution having a pH of 6 would: |
be slightly acidic |
|
|
A pH of 4 is ___________ times more ____________ than a pH of 7. |
1000; acidic |
|
|
The chemical reactivity of an atom is determined most directly by the: |
number of valence electrons |
|
|
Chlorine has seven electrons in its valence shell. The number of electrons it must gain to complete its valence shell is: |
one |
|
|
A covalent bond |
may be polar or non-polar depending on the atoms involved |
|
|
Which of the following choices correctly identifies a reactant in the following chemical equation? C6H12O6 + O2 -->6CO2 + 6H2O + ENERGY |
C6H12O6 AND O2 |
|
|
Which covalent bond involves only 2 electrons (1 pair) |
Single |
|
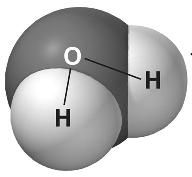
In a water molecule, because oxygen is more electronegative than hydrogen, the shared electrons are more commonly found around the __________ nucleus than the ______________nucleus.
|
oxygen, hydrogen |
|
|
When a small amount of hydrochloric acid (HC1) is added to a solution of Na2HPO4, the pH of the solution does not change markedly. The pH also does not change drastically when a small amount of sodium hydroxide (NaOH) is added to this same solution. Based on these observations, the compound Na2HPO4 is: |
acting as a buffer |

