![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
48 Cards in this Set
- Front
- Back
|
How can solid materials differ (in terms of properties)? |
They can differ in melting points strength (tension or compression) stiffness hardness density |
|
|
Meaning of strong ? |
if the material needs a large force to break it |
|
|
Meaning of tensile strength ? |
How much of a pull force is needed to break it |
|
|
Meaning of compressive strength ? |
how much push force is needed to break it ? |
|
|
Meaning of stiff ? |
Opposite to flexible, difficult to bend or stretch |
|
|
meaning of hard ? |
difficult to scratch |
|
|
Properties of plastics ? (5) |
hard strong stiff fairly low density mouldable |
|
|
Properties of rubber ? (4) |
strong soft flexible mouldable |
|
|
what are material we use? |
Chemicals or mixtures of chemicals, these include - metals -ceramics -polymers |
|
|
Materials be made or obtained from .... |
living things |
|
|
Name Natural polymers ? (4) |
cotton paper silk wool |
|
|
Name Synthetic polymers ? (5) |
Polythene Polypropylene PVC Polyester PET |
|
|
What can raw materials from the earths crust be used to make ? |
Synthetic materials |
|
|
During a chemical reaction the number of atoms at the start of the reaction is .... |
the same as the number of atoms at the end of the reaction |
|
|
What does crude oil mainly consist of ?
|
hydrocarbons |
|
|
What is a hydrocarbon ? |
A chain molecule of varying lengths made from carbon and hydrogen atoms only |
|
|
A small percentage of crude oil is used for .... ? |
chemical synthesis and most is used as fuels |
|
|
What does the petrochemical industry do ? |
it refines crude oil by fractional distillation |
|
|
How is crude oil separated ? |
by Fractional Distillation |
|
|
How does fractional distillation work ? |
1. hydrocarbons are separated into fractions of different boiling points 2. to produce fuels 3. to produce lubricants 4. and raw materials for chemical synthesis |
|
|
Relate the size of the forces between hydrocarbon molecules to the size of the molecules ? |
The longer the size of the molecule the stronger the force so the higher the boiling point (thick and sticky) |
|
|
Boiling point of short chain hydrocarbon molecules ? |
low boiling points - often gases |
|
|
Boiling point of long chain hydrocarbon molecules ? |
high boiling points- (thick and sticky) |
|
|
Name the two types of important bonds in crude oil (hydrocarbon) and explain them? |
1. strong covalent bond- between the hydrogen and carbon atoms (within each hydrocarbon) 2. intermolecular forces- attraction between different hydrocarbon molecules in the mixture |
|
|
What happens when crude oil when it is heated ? |
The molecules are supplied with extra energy, eventually the molecules can gain enough energy to overcome their intermolecular forces, it then turns into a gas |
|
|
What is stronger...the covalent bonds holding each molecule together or the intermolecular forces ? |
the covalent bonds holding each molecule together are much stronger than the intermolecular forces. |
|
|
Describe the intermolecular forces in small hydrocarbons (crude oil) ? |
the intermolecular forces break more easily in small molecules than in bigger molecules. The intermolecular attraction is weaker in small molecules |
|
|
what kind of boiling points do big molecules have ? |
they have high boiling points- more energy is needed for them to break out of a liquid and form a gas |
|
|
What are small molecules called ? |
Monomers |
|
|
What happens during polymerisation ? |
small molecules called monomers join together to make very long molecules called polymers |
|
|
Name examples of materials, that because of their superior properties, have replaced materials used in the past ? |
cotton, wool, silk ---> nylon and polyester wood-----> PVC |
|
|
What do the properties of polymers depend on ? |
how their molecules are arranged and held together |
|
|
What modifications in polymers have produced changes to their properties ? (4) |
1. increased chain length 2. cross - linking 3. the use of plasticisers 4. increased crystallinity |
|
|
cross links properties (5) |
1. less flexible 2. stronger 3. harder 4. higher melting point 5. stops rubber molecules uncoiling and sliding past each other |
|
|
cross links |
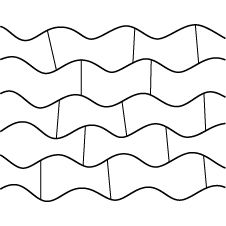
|
|
|
Plasticisers- properties (5) |
1. flexible 2. softer 3. low melting point 4. oily liquid 5. Makes PVC softer |
|
|
Plasticiser |
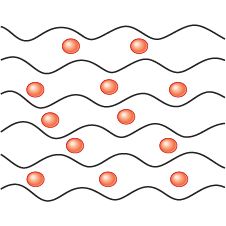
|
|
|
Branching chain properties (6) |
1. stretchy 2. elastic 3. rustle and noisy 4. plastic bags 5. line up irregularly 6. LDPE |
|
|
Branching chain |
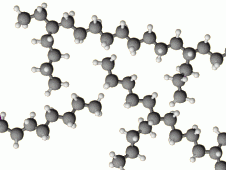
|
|
|
Crystalline polymer properties (5) |
1. stronger 2. denser 3. arranged in neat lines 4. plastic milk bottles 5. HDPE |
|
|
Crystalline polymer |

|
|
|
What does nanotechnology involve ? |
it involves structures that are about the same size as some molecules |
|
|
what is nanotechnology ? |
it is the use and control of structures that are very small (1-100nm) |
|
|
how can nanoparticles occur ? |
They can occur -naturally - seaspray -by accident- smallest particles from combustion of fuels -by design |
|
|
What do nanoparticles of a material show ? (properties) and give the reason |
they show different properties compared to larger particles of the same material. A reason of this is much larger surface area of the nanoparticles compared to their volume |
|
|
What can nanoparticles be used for ? |
They can be used to modify the properties of materials |
|
|
Examples of nanoparticles being used to modify the properties of materials ? |
1. the use of silver nanoparticles to give fibres antibacterial properties 2. adding nanoparticles to plastics for sport equipment to make them stronger (tennis rackets) |
|
|
Risks of nanotechnology ? |
1. Nobody knows the long term effects 2. large surface area to their volume, so they can react very quickly, this could speed up reactions such as illnesses 3. particles are so small they may be able to enter the brain from the bloodstream 4. nanoparticle have different properties, they may have different effects on plants/ animals. |

