![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
81 Cards in this Set
- Front
- Back
|
What is limestone made of |
Calcite |
|
|
Solid materials that do not pessess an orderly arrangement of atoms are called _ |
glasses |
|
|
Which of the following substances is not considered a mineral? 1) Coal 2) Diamond 3) Gypsum 4) Rock salt |
1) Coal |
|
|
Isotopes of a given element have _ |
The same number of protons but different atomic masses |
|
|
Which subatomic particle has a positive charge?
|
Proton |
|
|
Sodium has an atomic number of 11. HOw many electrons will the sodium ion Na+ have? |
10 |
|
|
Isotopes of an element have different numbers of _ |
Neutrons |
|
|
What is the term for atoms of the same element that have different numbers of neutrons? |
Isotopes |
|
|
Carbon has an atomic number of 6 and an atomic weight of 12.011. This means that carbon atoms have _ |
6 protons and varying numbers of neutrons |
|
|
The atomic mass of an element is equal to the number of _ |
Protons plus neutrons |
|
|
What will be the charge of an atom containing 8 protons, 9 neutrons, and 10 electrons |
-2 |
|
|
How are the elements organized in the periodic table? |
In order of increasing number of protons |
|
|
What does the symbol Na+ represent? |
A sodium cation |
|
|
In order to make a sodium (Na) chlorine (CL) bon in NaCl, _ |
One electron is transferred from the sodium atom to the chlorine atom |
|
|
Which of the following elements tends to share electrons? 1) carbon 2) helium 3) oxygen 4) sodium |
1) Carbon |
|
|
What is the dominant type of bonding in minerals? |
Ionic bonding |
|
|
Diamond is an example of what type of bonding? |
Covalent |
|
|
The growth of a solid from a gas or liquid whose atoms can come together in the proper chemical proportions and crystalline arrangement is called _ |
Crystallization |
|
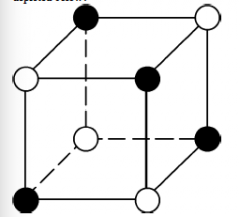
What elements could be represented by the open and solid spheres in the mineral structure depicted? |
Sodium and chlorine |
|
|
What is true about the relationship in size between Cations and Anions? |
Cations are generally smaller than anions |
|
|
The chemical formula (Mg,Fe)2SiO4 describes which mineral? |
Olivine |
|
|
Iron and magnesium ions are similar in size and both have a +2 positive charge. Therefore, we would expect iron and magnesium to _ |
substitute for each other in minerals |
|
|
Large crystals with well-formed crystal faces tend to form when _ |
Minerals have space to grow |
|
|
Chemical substances that have exactly the same chemical formula but different crystal strucures are calles _ |
Polymorphs |
|
|
Which of the following statements about graphite and diamond is false?
1) Graphite and diamond have the same density 2) Graphite and diamond have different mineral structures 3) Graphite and diamond are both made of carbon atoms 4) Graphite is stable in the Earth's crust, whereas diamond is stable in the Earth's mantle |
1) Graphite and diamond have the same density |
|
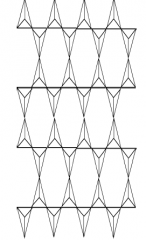
What silicate mineral contains tetrahedra linked togehter as depicted? |
Mica |
|
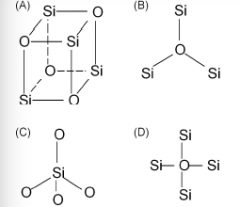
Which best depicts a silicate ion? |
Diagram C |
|
|
The most common rock-forming minerals in the crust are _ |
Silicates |
|
|
The two most common elements in the Earth's crust are _ |
Oxygen and silicon |
|
|
What mineral is a common clay mineral used for making pottery? |
Kaolinite |
|
|
Which of the following statements about feldspar is false? Feldspar is 1) harder than calcite 2) softer than corundum 3) the most abundant mineral in the Earth's crust 4) a sheet silicate |
4) Feldspar is a sheet silicate |
|
|
What type of minerals are calcite and dolomite? |
Carbonates |
|
|
Mica is a common example of a _ silicate |
Sheet |
|
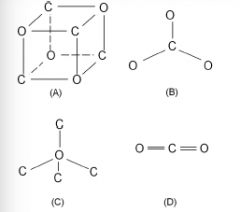
Which of the following atomic structures represents a carbonate ion? |
Diagram B |
|
|
Which mineral does not contain carbon? |
Gypsum |
|
|
What type of mineral is gypsum, the primary component of plaster? |
Sulfate |
|
|
What type of mineral is pyrite, also known as "fool's gold"? |
Sulfide |
|
|
What form of iron is the chief iron ore? |
Iron oxide |
|
|
Which metal is commonly mined as an oxide? |
Iron |
|
|
Which mineral fizzes when it comes in contact with hydrochloric acid (HCl)? |
Calcite |
|
|
What is the hardest mineral? |
Diamond |
|
|
Which of the following minerals has the greatest hardness? |
Quartz |
|
|
Which of the following minerals is not a form of Al2O3? |
Diamond |
|
|
The mineral property "cleavage" refers to _ |
The splitting of a mineral along planar surfaces |
|
|
When cleaved, mica breaks through planes of _ |
Interlayer cations |
|
|
Which of the following minerals has the best cleavage? |
Mica |
|
|
Which mineral has two good cleavage planes oriented at right angles to each other? |
Pyroxene |
|
|
Which of the following minerals does not exhibit cleavage? 1) Amphibole 2) Calcite 3) Mica 4) Quartz |
4) Quartz |
|
|
Which of the following statements regarding the density of minerals is false? 1) Density depends on the atomic weight of the ions in a mineral. 2) Density depends on the closeness of the atomic packing 3) Density increases with increasing pressure 4) Density increases with increasing temperature |
4) Density increases with increasing temperature |
|
|
Minerals with the same chemical forula but different crystal lattice structure are referred to as _ |
Polymorphs |
|
|
_ and _ are examples of polymorphs |
Diamond, graphite |
|
|
If a mineral is allowed to grow without space impediments, then it will develop _ |
Crystal faces |
|
|
Which of the following statements is false? 1) Minerals are crystalline 2) 90% of minerals are ionic compounds 3) Minerals are also called grains 4) The most common mineral in the Earth's crust is quartz |
4) The most common mineral in the Earth's crus is quartz |
|
|
Which of the following minerals is a framework silicate? 1) Quartz 2) Orthoclase feldspar 3) Plagioclase feldspar 4) They are all framework silicates |
4) They are all framework silicates
|
|
|
The silica tetrahedra readily bonds with cations when available because _ |
It has a net negative charge |
|
|
How many cleavage planes do sheet silicates tend to have? |
one |
|
|
How the surface of a mineral reflects light is referred to as _ |
luster |
|
|
Which is the least reliable physical property? |
Color |
|
|
The shape in which individual minerals or an aggregate of minerals grow is called _ |
habit |
|
|
Hardness is referred to as a mineral's _ |
resistence to scratching |
|
|
Which minerals are commonly found in igneous, sedimentary, and metamorphic rocks? |
Feldspar and quart |
|
|
The identity of a rock is dependent on its mineralogy and _ |
texture |
|
|
Rocks that form from the solidification of molten rock are called _ |
igneous |
|
|
The concept that identifies that the three basic groups of rocks are all related is called _ |
the rock cycle |
|
|
Minerals that contain metallic elements that can be recovered profitably are called _ |
Ore Minerals |
|
|
A common metal sulfide mineraal is _ 1) Galena 2) Pyrite 3) Cinnabar 4) They are all metal sulfide minerals |
4) They are all metal sulfide minerals
|
|
|
Ore depostits are associated with _ 1) Igneous rocks 2) Sedimentary rocks 3) Metamorphic rocks 4) All of the above |
4) all of the above |
|
|
Ore deposits that are formed when minerals are concentrated by the mechanical sorting action of river currents are called _ deposits. |
Placer |
|
|
The sulfate mineral found on the surface of Mars that supports the presence of liquid water is called _ |
Jarosite |
|
|
Sulfide minerals react with groundwater to produce _ |
Sulfuric acid Sulfate Ions (SO42-) Iron ions (Fe3+) |
|
|
What is not a mineral in the strictest geologic sense? |
Gall stone |
|
|
Which mineral is commonly referred to as "fool's gold"? |
Pyrite |
|
|
If hardness is a measure of the strength of chemical bonds, then which of the following minerals has the weakest bonds? 1) talc 2) mica 3) diamond 4) graphite |
1) Talc |
|
|
Calcite (CaCO3) fizzes when it comes in contract with hydrochloric acit (HCl). What are the products of this reaction? |
CaO and CO2 |
|
|
_ is the most common mineral or mineral group in the Earth's crust. |
Feldspar |
|
|
_ is the most common mineral or mineral group in the Earth as a whole |
Perovskite |
|
|
As we go deeper in the Earth and the pressure grows we encounter minerals with _
|
More compact crystal structures |
|
|
Which mineral or mineral group occurs exclusively in sedimentary rocks? |
Clay minerals |
|
|
Which mineral or mineral group occurs exclusively in a metamorphic rocks? |
Kyanite |
|
|
Which mineral or mineral group occurs exclusively in igneous rocks? |
Olivine |
|
|
The rock cycle describes how the three basic rock groups _ |
form, break down, and relate to one another |

