![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
49 Cards in this Set
- Front
- Back
|
What is an enzyme?
|
A protein catalyst that increases the rate of reactions w/o being changed in the overall process
|
|
|
What is a synthetase?
|
Requires ATP to function
|
|
|
What is a synthase?
|
no ATP required to function
|
|
|
What is a phosphatase?
|
Uses water to remove a phosphoryl group
|
|
|
What is a phosphorylase?
|
Uses Pi to break a bond and generate a phosphorylated product
|
|
|
What is a dehydrogenase?
|
NAD/FAD is electron acceptor in redox reactions
|
|
|
What is a oxidase?
|
O2 acceptor but oxygen atoms are not incorporated into substrate
|
|
|
What is a oxygenase?
|
One or both oxygen atoms are incorporated
|
|
|
What are RNAs w/ catalytic activity?
|
Ribozymes
|
|
|
What does the active site contain?
|
Amino acid side chains that participate in substrate binding and catalysis
|
|
|
What is the induced fit model?
|
The enzyme undergoes a conformational change that allows catalysis of the enzyme
|
|
|
What is the holoenzyme?
|
The active enzyme w/ its nonprotein component
|
|
|
What is a apoenzyme?
|
The enzyme w/o its nonprotein moiety, the inactive form
|
|
|
What is a cofactor?
|
If the nonprotein moiety of an enzyme is a metal ion such as Fe3+ or Zn2+
|
|
|
What is a coenzyme?
|
IF the nonprotein moiety of an enzyme is small organic molecule
|
|
|
What are cosubstrates?
|
Coenzymes that only transiently associate w/ the enzyme
|
|
|
What are coenzymes frequently derived from?
|
Vitamins
|
|
|
What is the purpose of enzyme compartmentalization?
|
Serves to isolate the reaction substrate or product from other competing reactions
Also provides a favorable environment for the reaction, and organizes the thousands of enzymes present in the cell into purposeful pathways |
|
|
What is the free energy of activation?
|
The energy difference b/e that of the reactants and a high-energy intermediate that occurs during the formation of product
|
|
|
What do molecules need in order to react?
|
Enough energy to overcome the energy barrier of the transition state
|
|
|
What is accomplished by the enzyme stabilizing the transition state?
|
Greatly increases the concentration of the reactive intermediate that can be converted to product and accelerates the reaction
|
|
|
What is the enzymes maximal velocity?
|
The rate of a reaction is the number of substrate molecules converted to product per unit time
|
|
|
What enzymes do not follow michaelis-menten kinetics?
|
Allosteric enzymes
|
|
|
What effect on reaction velocity does temperature have?
|
Increased temperature increases the rate of the reaction by adding more energy
|
|
|
What happens to reaction velocity if you heat it too hot?
|
The enzymes denature and reaction velocity decreases
|
|
|
What is the michaelis-menton equation?
|
v0 = (Vmax[S])/(Km + [S])
v0 = initial reaction velocity vmax = maximum reaction velocity [S] = substrate concentration Km = constant |
|
|
What assumptions are made by the michaelis-menton equation?
|
1. [S] >>> [E]
2. [ES] does not change w/ time 3. The rate of the reaction is measured as soon as enzyme and substrate are mixed |
|
|
What is Km equal to?
|
Numerically equal to the substrate concentration at which the reaction velocity is equal to 1/2Vmax
|
|
|
What does Km reflect about the enzyme? what does a low Km mean? high Km?
|
The affinity of the enzyme for that substrate
low Km = high affinity of enzyme for substrate high Km = a low affinity of enzyme for substrate |
|
|
What is the relationship of velocity to enzyme concentration?
|
The rate of the reaction is directly proportional to the enzyme concentration at all substrate concentrations
*i.e. when [E] is halved, v0 is halved too |
|
|
What is the order of a reaction?
|
first order: When [S] is much less than Km, the velocity of the reaction is approximately proportional to the substrate concentration
zero order: When [S] is much greater than Km, the velocity is constant and equal to Vmax, and rate is independent of [S] |
|
|
What is competitive inhibition?
|
When the inhibitor binds reversibly to the same site that the substrate would normally occupy
|
|
|
What is competitive inhibition's effect on Vmax? Km? lineweaver-burk plot?
|
![Vmax = effect of competitive inhibitor is reversed by increasing [S], so reactions eventually reach the Vmax
Km = increases the apparent Km, so more substrate is required to reach 1/2Km
L-B = shows a characteristic plot where the inhibited and u...](https://images.cram.com/images/upload-flashcards/61/57/88/2615788_m.jpg)
Vmax = effect of competitive inhibitor is reversed by increasing [S], so reactions eventually reach the Vmax
Km = increases the apparent Km, so more substrate is required to reach 1/2Km L-B = shows a characteristic plot where the inhibited and uninhibited plots intersect on the y-axis at 1/Vmax |
|
|
What is noncompetitive inhibition? what is its effect on Vmax? Km? L-B?
|
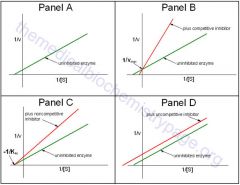
When inhibition occurs at a site other than the active site
Vmax = decrease the apparent Vmax of a reaction Km = does not interfere w/ binding, so same Km L-B = Apparent Vmax decreases |
|
|
What is the function of allosteric enzymes?
|
Alter the affinity of the enzyme for its substrate, or modify the maximal catalytic activity of the enzyme or both
|
|
|
What role does alanine play in the liver?
|
Transfers ammonia equivalents to the liver for processing into urea
|
|
|
What is the function of the ALT enzyme? What does it require to function?
|
Plays a critical role in amino acid catabolism
The vitamin B6 derivative PLP ("pyridoxal-5'-phosphate") |
|
|
What cofactor do many neurotransmitters require for their synthesis?
|
PLP
|
|
|
Which Michaelis-Menten kinetic parameters correlate with enzyme concentration?
|
v0
|
|
|
Which Michaelis-Menten parameter depends on substrate concentration? why?
|
Vmax;
b/c the Vmax is determined by how much substrate you can add before you reach maximum reaction speed |
|
|
Which Michaelis-Menten kinetic parameter indicates relative substrate preferences? why?
|
Kcat/Km
b/c Kcat/Km is the specificity constant and represents the enzymes ability to catalyze a particular substrate, so the lower Km and the higher Kcat the better the substrate binds to the enzyme |
|
|
What are the kinetics of indicator reactions characterized by?
|
low Km values and high activity
|
|
|
What is the effect of pH on enzymes?
|
There is a very narrow pH range where enzymes function w/o denaturing, so the pH must be strongly buffered
|
|
|
What would you consider a compound that binds to an enzyme w/o being modified?
|
A potential inhibitor
|
|
|
Where do Competitive, noncompetitive and mixed inhibitors intersect on a lineweaver-burk plot?
|
Competitive inhibitors intersect on the y-axis
noncompetitive inhibitors intersect on the x-axis mixed inhibitors intersect somewhere in outerspace and not on a axis |
|
|
What effect do mixed inhibitors have on binding to the enzyme-substrate complex?
|
Mixed inhibitors change the conformation of the active site and prevent substrate binding. SO it has the characteristics of competitive inhibition, but does not bind to the active site
|
|
|
What effect will binding of a substrate to part of an allosteric effector have?
|
Will change the conformation of all sites
|
|
|
What is the most common enzyme that is inhibited by uncompetitive inhibition?
|
bisubstrate enzymes
|
|
|
What effect on apparent Km do competitive inhibitors have?
|
Make the apparent Km larger
|

