![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
10 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
What is electrolysis and how does it happen? |
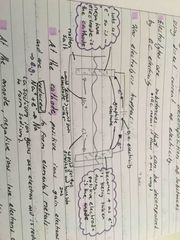
Electrolysis is the decomposition of substances using direct current of electricity At the cathode, positive ions gain electrons and are reduced to form elemental metals At the anode negative ions lose electrons and are oxidised to form diatomic molecules |
|
|
|
Electrolysis of aqueous salts (ionic solutions) |
-in water a small amount of water molecules hAve decomposed to form H+ and OH- - these ions wil compete at the electrodes with salt ions for oxidisation/reduction Rules: AT THE CATHODE- Ifthe metal is more reactive than copper; the hydrogen ions will be reduced to form H2 (only precious metals and copper will ‘out compete’ H+ AT THE ANODE- If sulfate ions (SO4 2-) are present, OH- will be oxidised to form O2. Otherwise the salt anion is always oxidised |
|
|
|
What is electroplating and how does it work? |
Electroplating is the coating of the surface of one metal with another metal. Done to cover cheap materials to improve finish, and to improve corrosion resistance. How it works: the anode- the plating metal/ the cathode- the metal you want covered (base metal)/ the electrolyte- palpitions containing the metal ions you want to plate on the cathode |
|
|
|
What happens when you use molten or aqueous salts in electrolysis |
If the salt is molten, the cathode will be plated (and will probably result in a colour change). If the salt is aqueous, the salt cation will have to compete with hydroxide ions. If the hydroxide ions are more reactive then copper, the hydrogen ions will turn into hydrogen and will become bubbles |
|
|
|
How to do half equations |

Remove spectator ions: -write down all ions present -what is formed at the cathode? -what is formed at the anode? -what are the half equations? -what is left over |
Molten half equations don’t involve H+ and OH-, only uses components from the salt, aqueous half equations do! |
|
|
What is oxidisation and reduction? |
OXIDISATION is the loss of electrons but a gain in oxygen (oxygen and the metal combine to form an ionic metal oxide. REDUCTION is the gain of electrons but a loss in oxygen. A redox reaction is when both happen in a reaction |
Think oilrig: Oxidisation Is Loss, Reduction Is Gain |
|
|
How to stop oxidisation |
-SACRIFICIAL PROTECTION: a more reactive metal is placed into a less reactive metal. The more reactive metal will oxidise whilst the other won’t. Note that the more reactive metal is only a block and will supply all of the other metal with electrons -GALVANISING: the coating (all of it) of a less reactive metal (usually iron in steel) with zinc. This protects inner layer from exposure. Note that if the outer layer is scratched, it will also act as a sacrificial anode to the inner layer. -COATING: this can be done with paint, plastic, oil ect and prevents oxygen and water getting to the metal. |
|
|
|
What are displacement reactions and how do you predict them? |
Displacement reactions are when metals lose electrons to form positive ions or vice versa. The more reactive the metal is, the stronger its ability to lose electrons to another metal is to become and ion. This means a displacement reaction is more like to occur. Displacement reactions are a type of redox reaction. |
|
|
|
What is an alloy? |
An alloy is a mixture of a metal with a other element (usually another metal) |
|
|
|
Strength of pure metal and an alloy |
PURE METAL: Ina a pure metal, all atoms are the same size and are arranged in a lattice structure. This means that when hit, the atoms can slide over each other easily making it WEAK. ALLOYS: In an alloy, atoms are différant sizes, this disrupts the regular arrangement. This means that when hit, the atoms cannot slide over each other, making it strong. |
|

