![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
22 Cards in this Set
- Front
- Back
|
Tera (T) |
10^12 |
|
|
giga (G) |
10^9 |
|
|
mega (M) |
10^6 |
|
|
Hecto(h) |
10^2 |
|
|
Deka(da) |
10^1 |
|
|
Deci(d) |
10^-1 |
|
|
Micro(μ) |
10^-6 |
|
|
Nano(n) |
10^-9 |
|
|
Pico(p) |
10^-12 |
|
|
Femto(f) |
10^-15 |
|
|
Diatonic molecules |
H2, N2, F2, O2, I2, Cl2, and Br2. |
|
|
Heterogeneous |
Not the same throughout (oil and water, for example). |
|
|
Homogeneous |
Same throughout, constant composition, constant properties. |
|
|
Law of conservation of mass |
Mass is neither created or destroyed. Substances change but the total amount of matter doesn't. Mass of reactants = mass of products |
|
|
Law of definite proportions |
Different samples of a pure substance always contain the same proportion of elements. Elements do not combine in random proportions. |
|
|
Law of multiple proportions |
If two elements combine in different ways to form different substances, the mass ratios are small whole-number multiples of each other (in other words, it's possible for the same elements to combine in different ratios). |
|
|
Isotope behavior |
Isotopes behave almost identically in chemical reactions to their regular counterpart. It is electrons that determine chemical reactions. |
|
|
Ions to memorize |
Cr = 2+, 3+, 6+ Fe = 2+, 3+ Co = 2+, 3+ Cu = +, 2+ |
|
|
When are the hypo-ite, -ite, -ate, per-are prefixes and suffixes used? |
On oxoanions (cation is positive ion, anion is negative ion). |
|
|
What is an Oxoacid? |
An acid that contains H, O and other elements. It dissolves in water to make H+ ions and a Polyatomic oxoanion. |
|
|
Rules for naming acids |

|
|
|
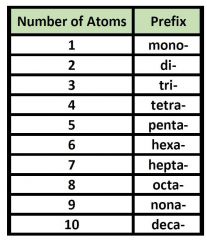
Prefixes for covalent bonds |
|

