![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
11 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Solubility rules |

|
|
|
|
How many millimeters are required for a solution that contains .115 moles of HBr and has the concentration of .183 M |
L soln= (.115 moles Hbr)/(.183 M)=.628 L*(1000mL)/(1L)=628 mL |
M=Mol HBr/ L soln |
|
|
Give the concentration of each of the ions in the following solutions: 0.75 M iron (II) chloride |
[Fe^2+]->Fe^2+ + 2Cl- -> .75 |
|
|
|
What volume of potassium hydroxide (0.365 M) would be needed to neutralize 15.5 milliliters of .219 moles of h2so4 |

|
|
|
|
Write the net ionic equation for the following reaction aqueous solutionI Anna key equation for the following reaction a quiz solution: HNO3+KOH->KNO3+H2O |
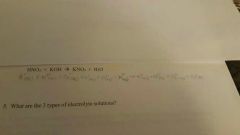
|
|
|
|
What are STP conditionswhat are a stupid conditions |
273k 1 atm= 760 mmHg =760 torr |
|
|
|
Calculate the molar mass of a gas with a density of 1.72 g/L at 1.50 atm and 25° C Which diatomic gas with this be? |
Molecular hydrogen |
|
|
|
Using heat of formation calculate change of enthalpy reaction for the following reaction: 2H2 (g) + O2(g) -> 2H2 (l) |
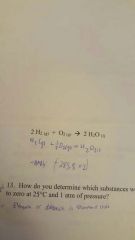
|
|
|

|

|
|
|
|
The rate of change of effusion of CO gas is 3.40 moles/min. What would the rate of ammonia gas very under the same conditions? |

|
|
|
|
6 types of chemical reactions |

|
|

