![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
57 Cards in this Set
- Front
- Back
|
If two identical nonmetal atoms bond together (like Chlorine-Chlorine) what type of bond is it if the electrons are shared equally? |
nonpolar covalent
nonpolar because the electrons are shared equally.
covalent because it is two nonmetals bonded together. |
|
|
How many electrons does a phosphorus atom need to satisfy the octet rule? |
3 |
|
|
How many electrons does a nitrogen atom need to satisfy the octet rule? |
3 |
|
|
How many electrons does a sulfur atom need to satisfy the octet rule? |
2 |
|
|
How many electrons does an oxygen s atom need to satisfy the octet rule? |
2 |
|
|
How many electrons does a chlorine atom need to satisfy the octet rule? |
1 |
|
|
How many electrons does a bromine atom need to satisfy the octet rule? |
1 |
|
|
What is the name for a neutral group of atoms held together by covalent bonds? |
molecule
or
molecular formula |
|
|
What type of bond is composed of nonmetals bonded with nonmetals? |
covalent |
|
|
What type of bond features the sharing of valence electrons? |
covalent |
|
|
What type of bond is composed of a metals bonded with a nonmetal |
ionic |
|
|
What type of bond features the transfer of valence electrons? |
ionic |
|
|
Draw the correct Lewis Dot diagram for hydrogen.
(or any other element in group 1) |
. |
|
|
Draw the correct Lewis Dot diagram for Rubidium.
(or any other element in group 1) |
. |
|
|
Draw the correct Lewis Dot diagram for calcium.
(or any other element in group 2 |
. |
|
|
Draw the correct Lewis Dot diagram for Aluminum.
(or any other element in group 3) |
. |
|
|
Draw the correct Lewis Dot diagram for silicon.
(or any other element in group 4) |
. |
|
|
Draw the correct Lewis Dot diagram for nitrogen.
(or any other element in group 5) |
. |
|
|
Draw the correct Lewis Dot diagram for sulfur.
(or any other element in group 6) |
. |
|
|
Draw the correct Lewis Dot diagram for bromine.
(or any other element in group 7) |
. |
|
|
Draw the correct Lewis Dot diagram for helium.
(or any other noble gas) |
. |
|
|
Draw the correct Lewis Dot diagram for argon.
(or any other noble gas) |
. |
|
|
What make a polar molecule polar? |
if the atom shares electron unequally and there is a region of positive charge and a region of negative charge. |
|
|
When drawing a Lewis structure every nonmetal (except hydrogen) should be surrounded by _________ electrons. |
8, an octet |
|
|
When drawing a Lewis structure containing hydrogen, each hydrogen atom should be surrounded by _________ electrons. |
2 |
|
|
Compare and contrast ionic and covelent bonds.
(Hint: Look at your foldable) |
Ionics are M + NM, the transfer electrons, called formulas units, very high melting points.
Covalent are NM + NM, the share electrons, called molecules, and have lower melting points. |
|
|
Which electrons are involved in the formation of chemical bonds? |
the valence electrons
valence electrons are the outer electrons.
(specifically the s & p orbitals) |
|
|
What can be said about the electrons in a metallic bond? |
*they form the sea of electrons
*they are delocalized and can go anywhere in the substance.
*they no longer belong to an individual atom |
|
|
Which elements can form an +1 ion when they bond? |
Alkali metals & hydrogen
(H, Li, Na, K, Rb, Cs, Fr) |
|
|
Which elements can form an +2 ion when they bond? |
Alkaline Earth Metals
(Be, Mg, Ca, Sr, Ba, Ra) |
|
|
Which elements can form an +3 ion when they bond? |
Boron's Group
(B, Al, Ga, In, Tl) |
|
|
Which elements can form an +4 or -4 ion when they bond? |
Carbon's group
(C, Si, Ge, Sn, Pb) |
|
|
Which elements can form an -3 ion when they bond? |
Nitrogen's group
(N, P, As, Sb, Bi) |
|
|
Which elements can form an -2 ion when they bond? |
Oxygen's group
(O, S, Se, Te, Po) |
|
|
Which elements can form an -1 ion when they bond? |
Halogens
(F, Cl, Br, I, At) |
|
|
Which elements do not form ions? |
Noble Gases
The have no electronegativity (attraction for an electron) and their ionization energy is so high they will not release an electron. |
|
|
Malleability, luster, ducility and conductivity are properties of what bond type? |
metallic bonds |
|
|
What is a molecular formula? |
The formula of a molecule or a covalently bonded (only nonmetals) compound. |
|
|
What happens to the electrons in a metallic bond which features a sea of electrons? |
electrons are delocalized and the mobile electrons are shared by all atoms
|
|
|
What does VSEPR stand for? |
Valence Shell Electron Pair Repulsion |
|
|
How can you determine bond type using electronegativity values? |
solve for teh absolute value of the difference of the electronegativity and compare:
ionic bonds have an electronegativity difference above 1.7 and covalent bonds have an electronegativity below 1.7/ |
|
|
Why do atoms combine to form bonds? |
they are more stable |
|
|
In a compound, what is the term for the electrical attraction to an electron called? |
electronegativity |
|
|
What is the Lewis Structure for lithium chloride, LiCl? |
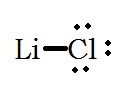
|
|
|
What is the Lewis Structure for hydrogen chloride, HCl? |
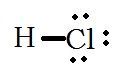
|
|
|
What is the Lewis Structure for Carbon tetrachloride, CCl4? |
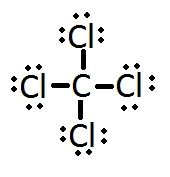
|
|
|
What is the Lewis structure for carbon tetraiodide, CI4? |
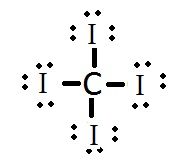
|
|
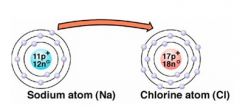
What type of bond is illustrated in the image? Be able to explain why. |
that is an ionic bond
Na is metal and Cl is nonmetal
Electrons are transferred |
|
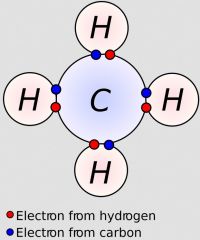
What type of bond is illustrated in the image? Be able to explain why. |
that is a covalent bond
C and H are both nonmetals
Electrons are shared |
|
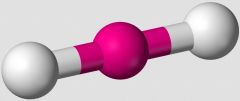
In the VSEPR Theory, which shape is illustrated. |
linear |
|
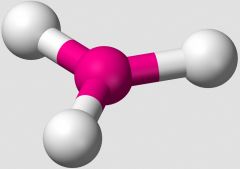
In the VSEPR Theory, which shape is illustrated. |
trigonal planar |
|
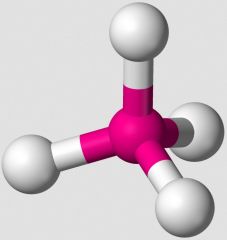
In the VSEPR Theory, which shape is illustrated. |
tetrahedral |
|
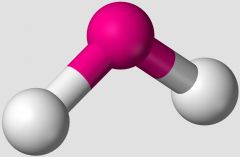
In the VSEPR Theory, which shape is illustrated. |
bent |
|
|
What VSEPR shape does an AB2 compound take, such as BeF2? |
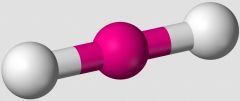
Linear |
|
|
What VSEPR shape does an AB3 compound take, such as BF3? |
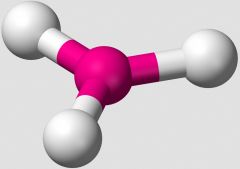
Trigonal Planar |
|
|
What VSEPR shape does an AB4 compound take, such as CH4? |
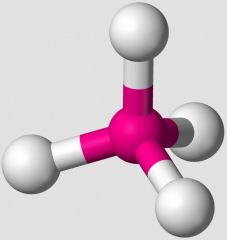
tetrahedral |
|
|
What VSEPR shape does an A2B compound take, such as H2O? |
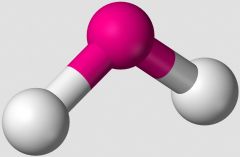
bent |

