![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
31 Cards in this Set
- Front
- Back
|
What type of bond is joining the two hydrogen atoms? |
covalent
A covalent bond is one in which electron pairs are shared |
|
|
A(n) _____ refers to two or more atoms held together by covalent bonds. |
molecule |
|
|
This atom can form up to _____ single covalent bond(s). |
4
There are four electrons in the outermost electron shell |
|
|
(n) _____ bond joins these two oxygen atoms. |
double covalent |
|
|
The innermost electron shell of an atom can hold up to _____ electrons. |
2 |
|
|
Which of these relationships is true of an uncharged atom? |
The number of protons is equal to the number of electrons |
|
|
What determines the types of chemical reactions that an atom participates in? |
the number of electrons in the outermost electron shell |
|
|
A phrase that applies to covalent bonding and not other kinds of bonds is ... |
electron-sharing |
|
|
When chemists and biologists want to show how atoms are bonded in a molecule, they usually use a ... |
structural formula. |
|
|
Which of the following statements most accurately describes the relative positions of the shared electrons in methane (CH4) and ammonia (NH3)? |
Electrons are shared equally in CH4 and unequally in NH3 |
|
|
Atoms of a specific element always have a constant number of _____. |
protons |
|
|
The brackets are indicating a(n) _____ bond. |
hydrogen
A hydrogen atom with a net positive charge is attracted to an oxygen atom with a net negative charge |
|
|
What name is given to the bond between water molecules? |
hydrogen |
|
|
Each water molecule is joined to _____ other water molecules by ____ bonds. |
four ... hydrogen |
|
|
The unequal sharing of electrons within a water molecule makes the water molecule _____. |
polar |
|
|
The tendency of an atom to pull electrons toward itself is referred to as its _____. |
electronegativity |
|
|
In this molecule, what type of bond is found between the oxygen and hydrogens? |
polar covalent |
|
|
Which of these bonds is weakest? |
A. Hydrogen Bonds are weaker than covalent bonds because they do not involve sharing of electrons, and they are weaker than ionic bonds because they involve the attraction of partial (not full) opposite charges |
|
|
Why isn't this insect drowning? |
Surface tension.
The high surface tension of water allows the insect to remain on the surface |
|
|
Which answer is a true statement about pH values? |
Pure water has a pH of 0. Stomach juice has a high pH. pH is important in life mainly because it affects the cohesion of water. A high pH corresponds to a high H+ concentration. None of the above. |
|
|
What conditions must be met in order for an aqueous solution to be called “neutral”? |
The concentrations of hydronium and hydroxide ions are equal |
|
|
The water molecule has a bent shape because ...
|
oxygen has two unbonded pairs of valence electrons |
|
|
Which statement is true of water? |
The H atoms in water have partial positive charges |
|
|
Which kinds of bonds are present in a single water molecule? |
polar covalent bonds |
|
|
The hydrogen bonds among water molecules endow water with which property? |
Water has a high specific heat. |
|
|
Which statement best explains the physical basis for why oil (molecules containing only carbon and hydrogen) and water do not mix? |
Oils are nonpolar molecules that cannot interact with water molecules via hydrogen bonding. |
|
|
What type of energy is stored in the chemical bonds of a molecule? |
potential energy |
|
|
The first law of thermodynamics states that _____. |
energy is neither created nor destroyed |
|
|
Carbon bonding Which molecules show an appropriate number of bonds around each carbon atom? |
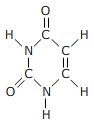
|
|
|
Which functional group contains nitrogen? |
amino |
|
|
Which of the following occurs when a covalent bond forms? |
Electrons in valence shells are shared between nuclei |

