![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
208 Cards in this Set
- Front
- Back
|
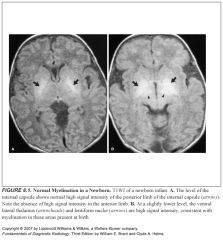
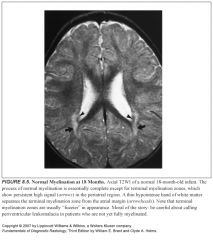
Best MR sequence to look at myelination patterns?
|
-T1 for the first 8 months
-T2 after 8 months |
|
|
What do myelinated structures look like on T1?
|
High signal against a background of low signal unmyelinated white matter
|
|
|
Why are myelinated structures high signal on T1?
|
Because the myelin sheath (a lipid) is hydrophobic, so myelinated structures have less water (the source of mobile hydrogen protons)
|
|
|
What do unmyelinated structures look like on T1?
|
Low signal
|
|
|
What sequence is recommended specifically for the infant brain?
|
Heavily T2 weighted images (long time of repetition/time of echo=3,000/120)
|
|
|
Why are heavily T2 weighted images used for pediatric neuroimaging?
|
-The water content of the infant brain is high
-Heavily T2 weighted images are needed to discriminate between many brain structures that have similarly long T2 relaxation times |
|
|
Assessment of every pediatric brain MR should begin with what?
|
Assessment of myelin development
|
|
|
In general, how does myelination proceed in the brain?
|
-From dorsal to ventral
-From caudal to cephalad -From central to peripheral |
|
|
What structures are actively myelinating at birth?
|
-Dorsal lentiform nucleus
-Lateral geniculate nucleus -Dorsal brainstem -Cerebellar peduncles -Ventrolateral thalamus -Posterior limb of the internal capsule -Corticospinal tract extending into the perirolandic white matter (precentral and postcentral) |
|
|
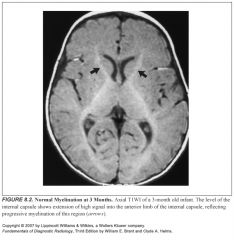
By when should the anterior limb of the internal capsule start myelinating (bright on T1)?
|
By 3 months
|
|
|
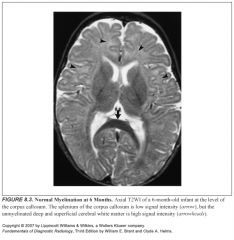
How does the corpus callosum myelinate?
|
The splenium becomes bright on T1 by 4 mos while the genu myelinates later, at 6 mos
|
|
|
Describe how myelination parallels developmental milestones:
|
-At birth, newborns can breathe and they have function of the motor components of the cranial nerves
-They slowly gain motor function (rolling over, crawling, standing), paralleling myelination of the internal capsule and corpus callosum -Higher cortical functions, such as speech, appear last, as the subcortical white matter starts to myelinate |
|
|
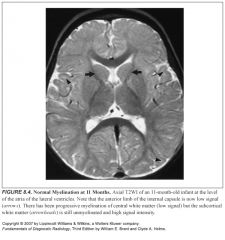
Where are the last places to myelinate?
|
The white matter around the atria and frontal horns of the lateral ventricles (funny, those are the first places to get demyelinated in leukoariosis)
|
|
|
Causes of delayed myelination?
|
-It’s a broad differential, the cause is unknown in many
-In utero insults (hypoxia, infections, toxins, coagulopathies) -Metabolic/nutritional disorders -Leukodystrophy (Pelizaeus-Merzbacker) |
|
|
Why would nutritional deficiencies or inborn errors of metabolism cause myelination delay?
|
Inadequate supply of myelin precursors
|
|
|
What is Pelizaeus-Merzbacker syndrome?
|
-Rare, X-linked leukodystrophy
-Causes lack of myelin formation because pts can’t metabolize long-chain fatty acids -Mimics other etiologies of delayed myelination |
|
|
What are the TORCH infections?
|
-Toxoplasma
-Other (includes syphilis) -Rubella -Cytomegalovirus -Herpes |
|
|
What is HIE?
|
-Hypoxic ischemic encephalopathy
-Occurring in utero or around the time of delivery |
|
|
Predisposing factors for HIE?
|
-Infection
-Intrauterine growth retardation -Metabolic derangements (e.g., hypoglycemia) -It is not indicative of any wrongdoing by the obstetrician |
|
|
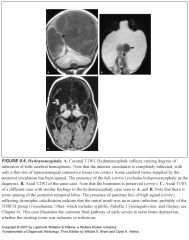
Describe hydranencephaly:
|
-Ischemic infarction of both cerebral hemispheres
-Thought to be caused by early compromise of both carotid arteries with preservation of posterior circulation |
|
|
What does hydranencephaly look like?
|
Little or no supratentorial brain tissue
|
|
|
How can you tell hydranencephaly from severe hydrocephalus?
|
Severe hydrocephalus usually has a thin rind of cortical gray matter
|
|
|
How do the white matter areas respond to in utero ischemia?
|
-They undergo neuronal loss and cystic cavitary atrophy
-Over time these cysts are incorporated into the ventricular wall, and the ventricles expand because of ex vacuo enlargement -The atrial margin is often crenulated (wavy) where the cysts are incoporated |
|
|
Define colpocephaly:
|
-Enlargement of the ventricular atria
-Caused by neuronal loss and cystic cavitary atrophy |
|
|
Define periventricular leukomalacia:
|
The entire process of damage and loss of the deep periventricular white matter
|
|
|
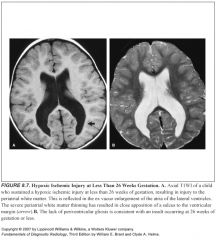
How does the brain respond to hypoxia/ischemia at different times of gestation?
|
Under 26 weeks:
-The brain does not mount a significant glial or scarring response -No T2 hyperintensity in areas of white matter damage After 26 weeks: -Able to mount a glial response (PVL) -High T2 signal in areas of damage |
|
|
What should you expect to see if an insult occurred in the first trimester?
|
Deep gray matter: spared
Cortex: irregularity, hydranencephaly if severe |
|
|
What should you expect to see if an insult occurred between 12-26 weeks?
|
-Deep gray matter: spared
-Cortex: spared -Periatrial injury -No gliosis -Ex vacuo enlargement |
|
|
What should you expect to see if an insult occurred after 26 weeks?
|
-Deep gray matter: spared
-Cortex: Watershed infarcts, Ulegyria -Variable gliosis and atrophy -Myelination delay -Injury to the hippocampi, pons |
|
|
Define gliosis:
|
Reaction of the brain to injury, much like fibrosis and scarring occurs elsewhere
|
|
|
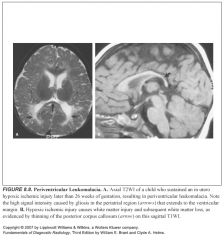
Define periventricular leukomalacia (PVL):
|
-“softening” of the white matter
-Seen in HIE after 26 weeks -End stage of periventricular white matter damage |
|
|
What sequence is PVL best seen on?
|
Proton density
|
|
|
What does PVL look like?
|
-Extension of high T2 signal to the ventricular margin
-Loss of the normal thin myelinated white matter roof of the atrium of the lateral ventricles -Periatrial white matter thinning and atrial enlargement are also evident |
|
|
Where are the important watershed areas?
|
-Parasagittal cerebrum, between the anterior and middle cerebral arterial distributions
-Posterior convexity: between the anterior, middle, and posterior cerebral arterial distributions -Medial surface between the anterior and posterior cerebral arteries |
|
|
Define Ulegyria:
|
-Mushroom shaped gyri
-Caused by subcortical infarcts that spare the gyri -Characteristic pattern |
|
|
How do you account for prematurity when imaging?
|
You should adjust according to how premature they are
|
|
|
Why are premature infants at risk for HIE?
|
-Extrauterine life places severe stresses on the developmentally immature brain, with oxygenation and nutrition being particular challenges
-Premature infants have periventricular border zones that are sensitive to HIE -Damage to the periventricular regions leading to varying degrees of PVL is common in premature infants |
|
|
What is the germinal matrix?
|
-A zone of proliferating, richly vascularized neuroectodermal cells
-Exquisitely sensitive to injury -Fragile blood vessels can easily rupture and cause hemorrhage when the infant is exposed to apnea, hypoxia, acidosis, unstable BP, etc. |
|
|
How do you monitor for germinal matrix hemorrhage?
|
Serial neonatal neurosonography
|
|
|
Why don’t term infants suffer from germinal matrix hemorrhages?
|
Developmentally, their germinal matrix has involuted and their arterial border zones have moved peripherally
|
|
|
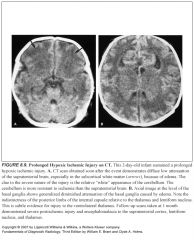
CT findings in acute “profound” perinatal HIE:
|
-Tends to damage central brain areas with relative sparing of the cerebral cortex
-On CT, this may lead to a peculiar appearance of deep gray matter structures becoming isodense to surrounding white matter |
|
|
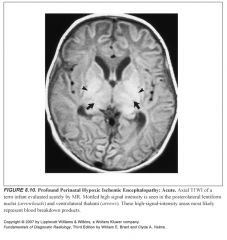
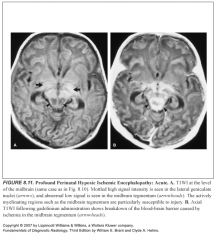
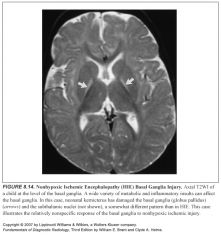
MR findings in acute (up to 3 weeks) “profound” perinatal HIE:
|
-Mottled, globular high signal on T1 in the basal ganglia (posterolateral lentiform nuclei) and ventral/lateral thalami
-These same areas show mottled low signal intensity on T2 -Additionally, similar signal abnormalities may be present in the tegmentum of the midbrain, lateral geniculate nuclei, and hippocampi |
|
|
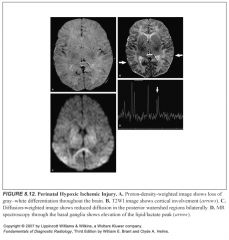
How do you NOT miss “profound” perinatal HIE?
|
-Scrutinize the proton density sequences, look for loss of gray-white interface
-Look at your DWI’s carefully -Spectroscopy is probably the earliest and most sensitive indicator of ischemic injury -This can be missed hyperacutely (first few days) |
|
|
What does “profound” perinatal HIE look like on spectroscopy?
|
Elevated lactate peak
|
|
|
What areas of cortex are involved in profound perinatal HIE?
|
The perirolandic cortex
|
|
|
MR findings in subacute profound perinatal HIE?
|
-Deep gray matter: variable signal on both T1 and T2
-Cortex: (perirolandic) high signal on T1, low signal on T2 -Atrophy of the hippocampi, lateral geniculate, midbrain tegmentum |
|
|
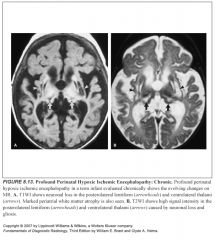
What are some MR findings in chronic profound perinatal HIE?
|
-High signal on T2 in deep gray matter and perirolandic cortex (gliosis)
-Thinned gyri -Atrophy of the hippocampi, lateral geniculate, midbrain tegmentum -Variable myelination delay |
|
|
What area of the brain is especially sensitive to toxic/metabolic insults?
|
-The basal ganglia
-Exhibit a relatively nonspecific response to nonhypoxic insults |
|
|
What does partial or mild perinatal HIE look like?
|
-There will be sparing of the central areas with damage to the peripheral gray matter
-This evolves into petechial gyral hemorrhage and eventually to areas of cortical thinning |
|
|
Why?
|
Diving reflex preserves bloodflow to the basal ganglia, brainstem, and cerebellum
|
|
|
Why is it so darn hard to see acute ischemia in a newborn?
|
Because the unmyelinated structures are bright on T2’s
It’s like looking for watery areas in an ocean |
|
|
What are your best tools?
|
-DWI
-Spectroscopy -After 24-72 hours after the insult, T2s are helpful, since you will see loss of the normal gray matter hypointensity amidst a sea of hyperintense (unmyelinated) white matter |
|
|
Why are T1’s confusing in a newborn with possible HIE?
|
-Because you will confuse the normal areas of active myelination (basal ganglia, thalami, cerebral peduncles, perirolandic white matter) with petechial hemorrhages
-If you see punctate high signal on T1 in areas NOT actively undergoing myelination, then it’s petechial hemorrhage |
|
|
Characteristic CT signs of diffuse cerebral edema?
|
-White cerebellum sign (correlates to dark cerebellum sign on T2)
-Pseudosubarachnoid hemorrhage |
|
|
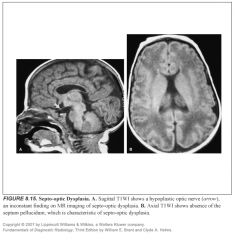
Define septo-optic dysplasia:
|
-Hypoplasia of the optic nerves with compete or partial absence of the septum pellucidum
-Also variable hypoplasia of the optic nerves, which may be limited to the optic disks, sparing the optic nerves |
|
|
Septo-optic dysplasia is associted with:
|
-Endocrine abnormalities (hypothalamic-pituitary axis abnormalities)
-Migration anomalies -Periventricular cysts |
|
|
How does septo-optic dysplasia relate to holoprosencephaly?
|
It can be considered the mildest form of the continuum
|
|
|
What does the face look like in kids with holoprosencephaly?
|
-Orbital hypotelorism (eyes too close)
-Varying degrees of facial dysmorphism |
|
|
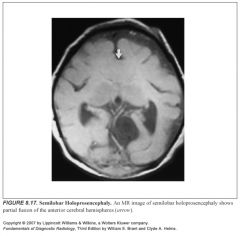
Describe the spectrum of holoprosencephaly?
|
From mildest to most severe:
Septo-optic dysplasia--lobar--semilobar--alobar |
|
|
What’s your mnemonic for alobar holoprosencephaly?
|
AH=Ah Ha! An Aunt Minnie!
|
|
|
What feature is common to all of the holoprosencephalies?
|
-Absent septum pellucidum
-Any portion of a visible septum excludes HP and should prompt consideration of severe hydrocephalus or agenesis of the corpus callosum |
|
|
What part of the brain doesn’t develop correctly in holoprosencephaly?
|
The prosencephalon
|
|
|
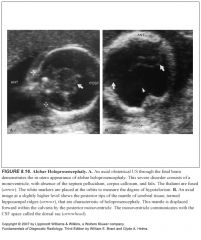
Describe alobar holoprosencephaly?
|
-Severe malformation
-Anterior rind of brain tissue -Monoventricle which communicates with a dorsal cyst -Thalami are fused -Septum pellucidum, corpus callosum, and falx are absent |
|
|
What two conditions can mimic alobar holoprosencephaly?
|
-Hydranencephaly
-Severe hydrocephalus with secondary pressure atrophy of the septum pellucidum |
|
|
How do you distinguish alobar holoprosencephaly from hydranencephaly and severe hydrocephalus?
|
-AH has an upside-down U shaped mantle of brain anteriorly
-Known as hippocampal ridges |
|
|
Describe semilobar holoprosencephaly:
|
-Partial fusion of the hemispheres
-The corpus callosum and the septum pellucidum are absent or dysgenic -The posterior portion of the interhemispheric fissure and flax are usually formed -Jason calls this “fused brain in the middle there” |
|
|
Semilobar holoprosencephaly is highly associated with:
|
Migration anomalies
|
|
|
Describe lobar holoprosencephaly:
|
-Relatively normal appearing brain
-Partial absence of the frontal interhemispheric fissure -The body and splenium of the corpus callosum are usually present, with the genu and rostrum absent |
|
|
What other entities are associated with an absent septum pellucidum?
|
-Facial anomalies (proboscis, cyclopia, cleft palate/lip)
-Solitary median maxillary central incisor -Anomalies of the cerebral cortex like schizencephaly, polymicrogyria, pachygyria |
|
|
What’s the best way to evaluate for fusion of the hemispheres?
|
Coronal T1
|
|
|
How does the corpus callosum form and how does it myelinate?
|
It forms from front to back
It myelinates from back to front |
|
|
If the corpus is malformed, which part will be affected?
|
The back—the body and the splenium (since the corpus forms from front to back, there will be sequential malformation extending from the front)
|
|
|
Which conditions defy the typical sequential front-to-back rule of corpus callosum malformations?
|
-Secondary destruction of the corpus callosum
-Holoprosencephaly Both can result in nonsequential segments of absent callosum |
|
|
If the corpus callosum is absent, what else will be absent too?
|
The cingulate gyrus and sulcus
|
|
|
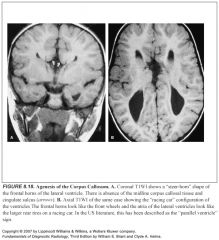
Describe imaging findings in absent corpus callosum?
|
-The medial hemisphere sulci will extend to the 3rd ventricle, giving sagittal images a radial, spoke-wheel appearance
-The ventricles will look different because there’s no corpus holding them in place -This makes the frontal horns look like steer horns on the coronal place and like racing cars on the axial plane |
|
|
Define colpocephaly:
|
-Dilatation of the occipital horns of the lateral ventricles
-Seen with absent corpus callosum (they’re the big tires on the racing car) |
|
|
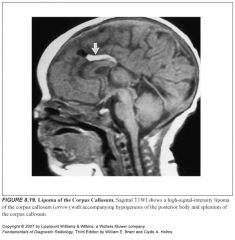
What are lipomas of the corpus callosum associated with?
|
Callosal anomalies
|
|
|
What do lipomas of the corpus look like?
|
-High T1 signal
-Suppress with fat sat -They do no cause mass effect -Vessels course through them unperturbed. |
|
|
How can you tell a lipoma in the corpus from interhemispheric hemorrhage or fat within the falx (and you don’t have fat sats)?
|
Look for chemical shift artifact along the frequency encoding direction of the scan
|
|
|
What do you think about when you see fat globules floating in the CSF spaces?
|
Ruptured dermoid!
|
|
|
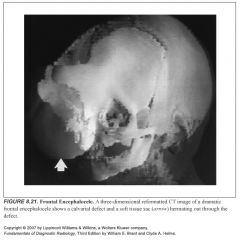
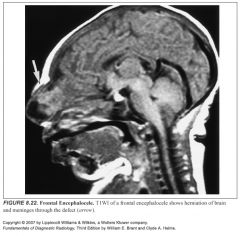
Define cephalocele:
|
Failure of the skull and dura to close over the brain, leading to a herniation of intracranial contents (brain and leptomeninges) through the defect
|
|
|
Which are more common, occipital or frontal encephaloceles?
|
Occipital
|
|
|
What is the mildest form of frontal encephalocele?
|
Nasal dermoid and sphenoid encephaloceles
|
|
|
What do clinicians want to know about nasal dermoids?
|
They require evaluation with sagittal T2s to ensure there is no potential for CSF leak developing with resection.
|
|
|
How would a sphenoid encephalocelel present?
|
-They are often occult
-They present as a nasopharyngeal mass that contains variable amounts of herniated 3rd ventricle, hypothalamus, and optic chiasm |
|
|
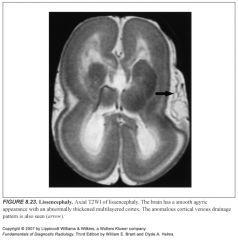
Define Lissencephaly:
|
Absence of gyri with abnormally thick cortex
(“listen”-cephaly because the brain looks like two ears!) |
|
|
What can mimic lissencephaly?
|
Premature infants’ brains
|
|
|
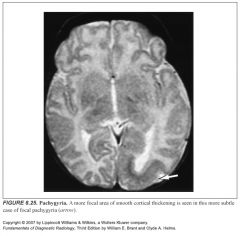
Define pachygyria:
|
Broad thick gyri with shallow sulci
|
|
|
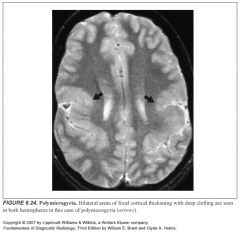
Define polymicrogyria:
|
Thick mantle of gray matter with multiple small gyri
|
|
|
How can you differentiate pachygyria from polymicrogyria?
|
Underlying white matter gliosis in polymircogyria can sometimes help differentiate it from pachygyria
|
|
|
What’s the difference between pachygyria/polymicrogyria and lissencephaly?
|
Polymicrogyria and pachygyria are more focal, lissencephaly involves the whole brain
|
|
|
What vascular anomalies are associated with cotical dysplasias?
|
-Anomalous draining cortical veins
-Don’t confuse these for AVMs |
|
|
What do you call it if an entire hemisphere is enlarged and composed of polymicrogyric and pachygyric areas?
|
Hemimegalencephaly
|
|
|
What is hemimegalencephaly associated with?
|
Neurofibromatosis and other various syndromes
|
|
|
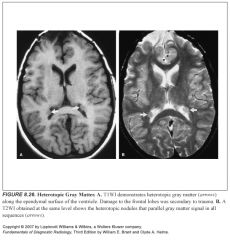
How do nests of heterotopic gray matter form?
|
As neurons migrate from the germinal matrix to the overlying cerebral cortex, their journey may be disrupted
|
|
|
Where is heterotopic gray matter seen?
|
Anywhere between the ependymal surface and the subcortical white matter
|
|
|
Imaging characteristics of heterotopic gray matter?
|
-Isotense to normal gray matter on all imaging sequences
-Does not enhance -Does not calcify |
|
|
What can mimic hetertopic gray matter?
|
Subependymal nodules seen in tuberous sclerosis
|
|
|
How can you distinguish heterotopic gray matter from subependymal nodules of tuberous sclerosis?
|
The tuberous sclerosis lesions calcify
|
|
|
Types of heterotopic gray matter:
|
-Most are nodular
-Band or laminar heterotopias can be seen |
|
|
What does a band heterotopia look like?
|
-A smooth layer of gray matter within the subcortical white matter, resulting in a double cortex appearance
-This is typically associated with severe seizure disorders and significant developmental delay |
|
|
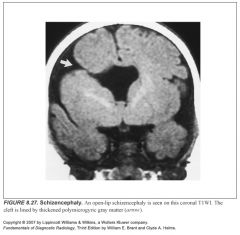
Define schizencephaly:
|
-Abnormality of neuronal migration resulting in gray matter lined clefts that deeply invaginate into the brain
-These clefts often extend from the ventricular ependymal surface to the pial cortical surface, giving rise to a pial-ependymal seam which communicates with the ventricle |
|
|
What causes schizencephaly?
|
Thought to be an in utero insult or expression of a genetic factor that damages the germinal matrix and impedes neural migration
|
|
|
What’s the difference between open and closed lip schizencephaly?
|
-Open means the lips are wide open
-Closed means the lips are apposed |
|
|
What do you call a cleft that does not extend to the ventricle?
|
Focal cortical dysplasia or polymicrogyral clefts
|
|
|
What is schizencephaly associated with?
|
Other migrational anomalies, such as cortical dysplasias and heterotopic gray matter
|
|
|
What could you confuse schizencephaly with?
|
Porencephaly
|
|
|
What is porencephaly?
|
Zone of encephalomalacia that communicates with the ventricle
|
|
|
How do you distinguish porencephaly from schizencephaly?
|
-Schizencephalic clefts are lined by gray matter
-Porencehpalic cysts are lined by a thin layer of white matter |
|
|
How children with migrational anomalies present?
|
Seizures
|
|
|
What are Chiari malformations?
|
Hindbrain anomalies
|
|
|
How many types of Chiaris are there?
|
Four
|
|
|
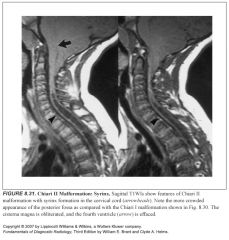
What is a Chiari II malformation?
|
Serious neural tube disorders that are screened for in maternal prenatal US and alpha-fetoprotein programs
|
|
|
Supratentorial components of Chiari II malformation:
|
-Most will have hydrocephalus
-Most have partial or complete agenesis of the corpus -Falx cerebri is often fenestrated, resulting in herniation of individual gyri across midline -Mass intermedia (connects thalami in midline) is enlarged -The posterior cingulate gyrus is often dysplastic |
|
|
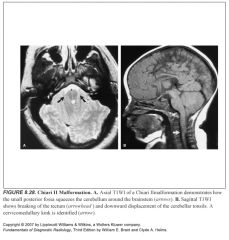
Posterior fossa components of Chiari II malformation:
|
-Diminuitive posterior fossa
-The cerebellum is squeezed against the tentorium, down through the foramen magnum and forward around the brainstem -The 4th ventricle is squeeed into a small vertical slit -The pons and medulla are squeezed inferiorly, with the fixed attachment to the upper cervical cord causing a cervicomedullary kink |
|
|
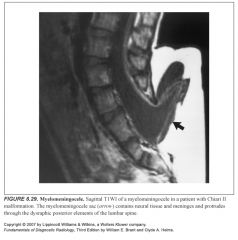
Spine components of Chiari II malformation:
|
Most have a myelomeningocele which often fails to induce a dural or bony covering
|
|
|
Define cerebellar tonsillar ectopia:
|
Tonsils extend >5mm below the foramen magnum
|
|
|
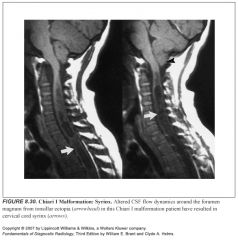
What is a Chiari I malformation?
|
-Cerebellar tonsillar ectopia
-Pts may be asymptomatic, but alterations in CSF dynamics at the level of the foramen magnum may give rise to a cervical spinal cord syrinx in some pts |
|
|
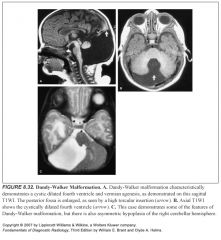
Define Dandy-Walker malformation:
|
-Large posterior fossa filled by a cystically dilated fourth ventricle
-High tentorial insertion (high torcula) -The posterior fossa is filled by a cystically dilated fourth ventricle that exerts mass effect -Hypoplasia or absence of the cerebellar vermis and cerebellar hemispheres -Hydrocephalus is also common, as is callosal hypogenesis |
|
|
What are the less severe Dandy-Walker variants?
|
Dandy-Walker variant
Mega cisterna magna |
|
|
Describe Dandy-Walker variant:
|
-Normal-sized posterior fossa
-Hypoplasia or absence of the vermis and cerebellar hemispheres but no siginificant mass effect |
|
|
Describe mega cisterna magna:
|
-Normal-sized posterior fossa
-Relatively normal cerebellar hemispheres and vermis -Prominent cisterna magna CSF space without mass effect |
|
|
How can you tell a Dandy-Walker from a varient?
|
-The variants will have a normal position of the torcula
-Regular Dandy-Walker has a high torcula |
|
|
What could mega cisterna magna be confused with?
|
-Retrocerebellar arachnoid cysts
-Epidermoid neoplasms |
|
|
How can you tell the difference between a mega cisterna magna and a retrocerebellar arachnoid cyst or epidermoid neoplasm?
|
-The retrocerebellar arachnoid cyst or epidermoid neoplasm are mass lesions causing an inward convex bowing of brain tissue at their interface.
-Long-standing masses will cause smooth erosion of the inner table of the skull -Mega cisterna magna doesn't exert mass effect |
|
|
What is a phakomatosis?
|
A hereditary syndrome of the neuroectodermal system
|
|
|
Name the phakomatoses:
|
-NF 1 & 2
-Tuberous sclerosis -Sturge Weber -Von Hippel Lindau |
|
|
What chromosome is NF-1 linked to?
|
17
|
|
|
Bony and spine findings in NF-1:
|
-Scoliosis (most common skeletal manifestation)
-Bones, especially the ribs, can develop chronic erosions (pits) from the constant pressure of adjacent neurofibromas and schwannomas -Sphenoid dysplasia -Lambdoid suture dysplasia -Thinning and bowing of long bones (e.g., ribbon ribs) with a tendency to fracture and not heal, yielding a pseudarthrosis (tibia is the most common site) -Spinal neurofibromas with associated widening of the neurofomaina -Meningoceles -Dural ectasia can lead to scalloping of the posterior vertebral bodies -Unilateral overgrowth of a limb |
|
|
What is the most common site for a pseudoarthrosis in NF-1?
|
Tibia
|
|
|
Why do some pts with NF-1 get unilateral overgrowth of a limb?
|
When a plexiform neurofibroma manifests on a leg or arm, it will cause extra blood circulation, and may thus accelerate the growth of the limb. This may cause considerable difference in length between left and right limbs.
|
|
|
Peripheral nerve findings in NF-1?
|
-Neurofibromas
-Plexiform neurofibromas -Schwannomas -Malignant peripheral nerve-sheath tumors (MPNST) |
|
|
What's the lifetime risk of a plexiform neurofibroma degenerating into a malignant peripheral nerve sheath tumor?
|
A plexiform neurofibromas has a lifetime risk of 8-12% of transformation
|
|
|
What's the main difference clinically between a schwannoma and a neurofibroma?
|
A schwannoma can be resected without sacrificing the underlying nerve, unlike the neurofibroma
|
|
|
Central nervous system findings in NF-1?
|
-Optic and other gliomas
-"unidentified bright object" or UBO's -Astrocytomas -Dural ectasia (focal thickening) |
|
|
Vascular lesions of NF-1?
|
Aneurysms, vascular ectasias, stenosis, moyamoya are all associated with NF-1
|
|
|
NF1 is also known as:
|
Von Recklinghausen disease
|
|
|
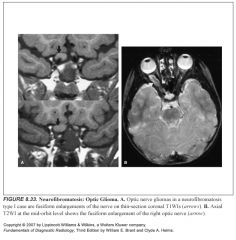
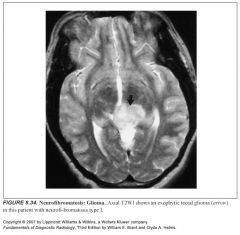
What does an optic glioma look like?
|
-Fusiform enlargement of the optic nerve
-Chiasm, optic tracts, and optic radiations can also become involved |
|
|
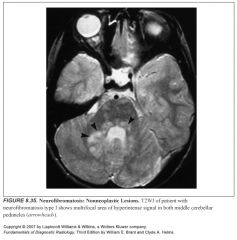
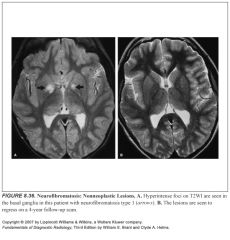
What kind of white matter changes are seen in NF1?
|
-High T2 signal lesions in the deep cerebral and cerebellar white matter
-Lesions wax and wane, do not cause mass effect, do not enhance -Tend to regress as the pts age (if they don’t you need to worry about neoplasm) |
|
|
Signs of neoplastic transformation of white matter lesions in NF-1:
|
-Significant enlargement
-New mass effect -Gadolinium enhancement |
|
|
What basal ganglia structure tends to have high T2 signal in NF1?
|
Globus palladi
|
|
|
What chromosome is NF2 associated with?
|
22
|
|
|
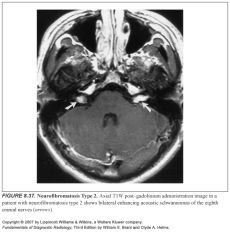
Features of NF2:
|
-Bilateral vestibular schwannomas (aka acoustic neuromas)
-Other cranial nerves meningiomas and schwannomas (CNV is 2nd most commonly affected) -Meningiomas -Ependymomas -Spinal glial tumors -Optic and other gliomas |
|
|
Skin lesions of tuberous sclerosis?
|
-Ash leaf spots
-Adenoma sebaceum -Shagreen patches -Subungual fibromas |
|
|
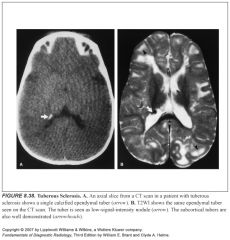
Brain lesions of tuberous sclerosis?
|
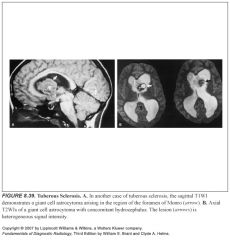
-Subependymal hamartomas
-Subependymal giant cell astrocytomas (basically a subependymal hamartoma that has degenerated--they enlarge, cause mass effect and invade brain tissue) -Cortical tubers Both cortical and subependymal lesions can undergo age-dependent calcification |
|
|
Where are subependymal giant cell tumors found in tuberous sclerosis?
|
At the foramen of Monro
|
|
|
What do subependymal tumors look like?
|
-They are hamartomas, so they tend to parallel white matter signal on MR (before they calcify)
-Calcific nodules may be isointense or hyperintense on T1 |
|
|
How do you tell if a subependymal tumor has undergone malignant transformation?
|
-Look for brain invasion
-Look for increase in size -If they enhance, it does not mean there’s malignant transformation |
|
|
What do cortical tubers look like?
|
-They are usually hypointense on T1 and hyperintense on T2
-Can calcify |
|
|
Another name for Sturge-Weber disease?
|
Encephalotrigeminal angiomatosis
|
|
|
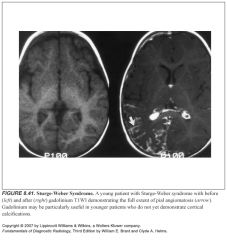
Describe Sturge-Weber disease:
|
It is an embryonal developmental anomaly resulting from errors in mesodermal and ectodermal development.
Unlike other neurocutaneous disorders (phakomatoses), Sturge-Weber does not have a hereditary tendency but occurs sporadically. It is caused by an arteriovenous malformation that occurs in the cerebrum of the brain on the same side as the physical signs described above. Normally, only one side of the head is affected. |
|
|
What is the facial lesion of Sturge-Weber disease?
|
-Port wine stain
-Appears in the ophthalmic division of CNV |
|
|
What is the neuro lesion of Sturge-Weber disease?
|
Pial angiomatosis
|
|
|
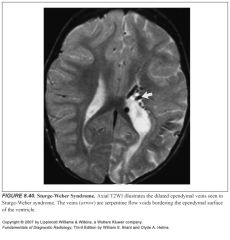
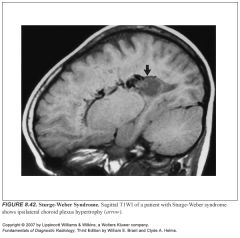
Three sequelae of pial angiomatosis:
|
-Gyral atrophy and underlying gliosis
-Enlargement of deep and subependymal veins -Ipsilateral hypertrophy of the choroid plexus |
|
|
Why is gyral atrophy seen with pial angiomatosis of Sturge Weber disease?
|
Because of chronic ischemia of the gray matter
|
|
|
What can dilated subependymal veins of Sturge Weber mimic?
|
AVMs
|
|
|
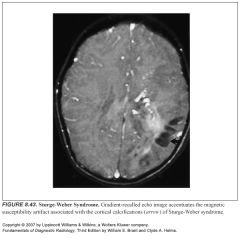
What do pial angiomas of Sturge Weber look like?
|
-They undergo age-dependent calcification
-Gyral cortical calcifications -Young children may show subtle hypointensity of the underlying white matter on T2 before calcification occurs |
|
|
What MR sequence helps demonstrate calcium?
|
Gradient-recalled echo
|
|
|
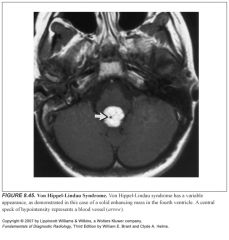
Neuro features of Von Hippel-Lindau Syndrome?
|
-Retinal angiomas
-Cerebellar and spinal hemangioblastomas |
|
|
Complications of hemangioblastomas?
|
-Although they’re benign, they’re prone to recurrence (up to 25%)
-Sudden spontaneous hemorrhage can occur |
|
|
What are the non-neuro lesions of von Hippel-Lindau?
|
-Renal cell carcinoma
-Angiomas of the liver and kidney -Pancreatic cysts -Pheochromocytoma |
|
|
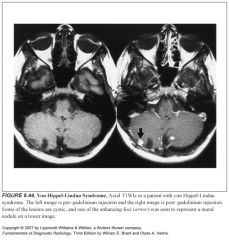
What do cerebellar hemangioblastomas look like?
|
-Well-circumscribed cystic lesion with an enhancing mural nodule
-Can look like a solid tumor, a solid tumor with a central cyst, or a cyst alone -A helpful finding is a large blood vessel leading to the nodule -Small multifocal hemangioblastoma nodules are seen near the pial surgace of the cerebellum or spinal cord |
|
|
What do spinal cord hemangioblastomas look like?
|
Multifocal spinal cord nodules near the pia-arachnoid surface
|
|
|
1
|
|
|
2
|
|
|
3
|
|
|
4
|
|
|
5
|
|
|
6
|
|
|
7
|
|
|
8
|
|
|
9
|
|
|
10
|
|
|
11
|
|
|
12
|
|
|
13
|
|
|
14
|
|
|
15
|
|
|
16
|
|
|
17
|
|
|
18
|
|
|
19
|
|
|
20
|
|
|
21
|
|
|
22
|
|
|
23
|
|
|
24
|
|
|
25
|
|
|
26
|
|
|
27
|
|
|
28
|
|
|
29
|
|
|
30
|
|
|
31
|
|
|
32
|
|
|
33
|
|
|
34
|
|
|
35
|
|
|
36
|
|
|
37
|
|
|
38
|
|
|
39
|
|
|
40
|
|
|
41
|
|
|
42
|
|
|
43
|
|
|
44
|
|
|
45
|

