![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
9 Cards in this Set
- Front
- Back
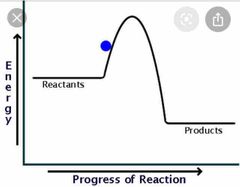
Label the Graph for Exothermic Reaction |
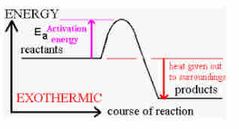
Energy is conserved
|
|
|
Exothermic Reaction |
1) Energy is transferred to the surroundings. 2) They are used to heat stuff I.e hand warmers, self heating cans. 3) Bonds are made 4) Get hotter in practicals
|
|
|
Examples of Exothermic Reactions |
Combustion - burning fuels Oxidation - respiration Neutralisation - acids and bases |
|
|
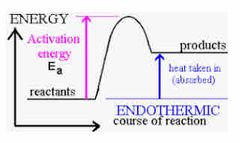
Endothermic Reaction |
1) Energy is absorbed from the surroundings so the temp of the surrounding decreases. 2) Can be used to cool things down like cold packs. 3) Bonds are broken 4) Get colder in reactions |
|
|
Examples of Endothermic reactions |
Thermal Decomposition Photosynthesis Some types of Electrolysis |
|
|
Required Practical: Investigating temp changes. |
1) Use polystyrene cup and thermometer 2) Record initial temperature 3) Add substance 4) Record max or min temp change |
|
|
Label the Graph for Endothermic Reaction |
Back (Definition) |
|
|
Activation Energy |
The amount of energy required for the reaction to happen. Greater in Endothermic than in Exothermic Reactions |
|
|
Bond energy |
1) Energy needed to break the bond between two atoms 2) Each bond has a different bond energy |

