![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
6 Cards in this Set
- Front
- Back
|
State the products of complete combustion of a hydrocarbon and give an example of hydrocarbons with complete combustion |
Carbon dioxide + water Alkanes |
|
|
State 2 things that happen during combustion of hydrocarbon fuels |
- Releases energy - Carbon and hydrogen in fuels are oxidised |
|
|
What does it mean when hydrogen and carbon molecules are oxidised |
They gain oxygen or react with oxygen |
|
|
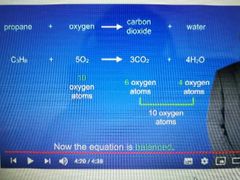
When balancing equations in hydrocarbons what is the order of elements which you start and finish with for the most efficient way of balancing |
- Carbon atoms balance on ecah side first - Hydrogen atoms second - Oxygen atoms 3rd |
|
|
Write the general equation and symbol equation for combustion of hydrocarbons |
Alkane/alkene + oxygen - - - - - > carbon dioxide + water C2H2n/+2 + O2 - - - > CO2 + H2O |
|

Balance this equation |
|

