![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
39 Cards in this Set
- Front
- Back
|
What is the definition of a Polymer and Monomer? |
Polymer: Molecules made up of a large number of monomers joined together Monomer: The smaller units from which larger molecules are made |
|
|
How are disaccharides and polysaccharides formed? |
Disaccharides are formed by the condensation of two monosaccharides.
Polysaccharides are formed by the condensation of many glucose of monosaccharide units e.g Starch from Alpha Glucose. Cellulose from Beta. |
|
|
How are Maltose, Sucrose and Lactose formed? |
Maltose from Condensation of two glucose Lactose from Condensation of glucose and galactose Sucrose from Condensation of Glucose and Fructose |
|
|
How can disaccharides be be broken down into monosaccharides? |
Hydrolysis reaction by boiling with acid.
Or by warming with an Enzyme at optimum temperature. |
|
|
State the purpose of Sodium Ions In living things |
Co-Transport of glucose and amino acids across cell membranes
|
|
|
State the purpose of Iron ions in living things |
A component of Haemoglobin which transports oxygen |
|
|
State the purpose of Hydrogen ions in living things |
They are important in determining pH, thus affecting enzyme activity and protein structure |
|
|
State the purpose of Phosphate ions in living things |
They are important as a structural component in DNA, RNA and ATP |
|
|
Explain ways in which water has an important role in Metabolic Reactions |
Water is a solvent in which metabolic reactions can occur
Water is a metabolite in Metabolic Reactions such as Hydrolysis and Condensation
Water is required for photosynthesis
Water is a metabolic product of respiration. Particularly important for creatures in dry habitats |
|
|
Explain ways in which water is an important solvent |
Water can transport dissolved nutrients such as glucose in the blood and sucrose in the phloem of plants Water can remove excretory products such as urea and ammonia by dissolving them Water is an important solvent for metabolic reactions to occur in |
|
|
Explain ways in which water is important for Temperature Regulation |
Water has a Relatively High Specific Heat Capacity. Takes a lot of energy to heat up 1 gram of water by 1 degree. This minimises temperature changes in cells due to heat given off by biochemical reactions , keeping them at optimum temp. Prevents dangerous fluctuations in temperature of aquatic habitats. Water has a large latent heat of vaporisation. Provides a cooling effect through sweating as heat energy is absorbed by the water in order to break its bonds and make it evaporate. Allows animals to maintain a constant body temperature . Allows plants to cool themselves via transpiration, prevent overheating |
|
|
Offer an explanation for the properties of water in Temperature Control and Support |
Hydrogen bonds form between water molecules with cohesive forces between molecules which form a cohesive structure. Water has a High Specific Heat Capacity because a large amount of heat energy can be absorbed by the bonds between molecules before the molecules themselves begin to heat. Buffers temperature change. Similarly, water has a Large Latent Heat of Vaporisation because it takes energy to break the hydrogen bonds between molecules and allow the molecules to become gaseous. |
|
|
Describe / Draw the general structure of an Amino Acid |
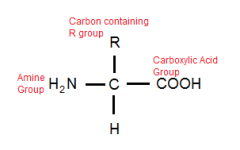
Carbon, with an attached Carboxylic Acid (-COOH-), an Amine Group (-NH2), one Hydrogen and a Carbon Containing R group which differs between different amino acids.
|
|
|
State the enzymes used in the hydrolysis and condensation of Adenosine Triphosphate (ATP)
|
ATP Hydrolase hydrolises ATP into Adenosine DiPhosphate and a Phosphate, releasing energy. ATP Synthase resynthesizes ATP from ADP and a Phosphate using energy from photosynthesis and respiration |
|
|
What is the structure of ATP?
|
The nitrogen containing base Adenine bound to a molecule of the pentose suger Ribose and Three Phosphates
|
|
|
Besides energy, give another use of ATP
|
The inorganic phosphate released can be used to phosphorylate other compounds, making them more reactive. e.g Glucose can be phosphorylated to form a more reactive Glucose Phosphate |
|
|
What is DNA made of?
|
The pentose sugar Deoxyribose
A phosphate group One of the four different nitrogen containing bases (Cytosine, Thymine, Adenine, Guanine) |
|
|
What are the properties of Triglycerides?
How does this relate to their function? |
|
|
|
What are the properties of Phospholipids?
How does this relate to their function? |
Hydrophobic tails. Hydrophilic head. Lets them be used in Phospholipid Layers for cell membranes. |
|
|
What can cause the denaturation of proteins?
|
The breaking of hydrogen and ionic bonds is caused by High Temperatures, Extreme pH Changes and Heavy Metals
|
|
|
What would be the control for an investigation into the effect of temperature on enzyme activity?
|
Use a thermostatically controlled water bath. Check regularly to make sure no temperature fluctuations. |
|
|
What would be the control for an investigation into the effect of pH on enzyme activity? |
Use a buffer. This will resist change in pH |
|
|
What would be the control for an investigation into the effect of Concentrations (of enzymes, substrates or inhibitors) on enzyme activity? |
Use the same volumes of each solution, |
|
|
Explain the stages of semi-conservative replication of DNA |
1. The enzyme DNA Helicase unwinds the double helix and breaks the bonds between the two strands. 2. Nucleotides with complimentary bases align next to the exposed bases on the template strand, according to specific complimentary base pairing 3. The enzyme DNA Polymerase joins the new nucleotides with phosphodiester bonds. 4. The two new strands coil up into double helixes after the two strands of DNA join together by hydrogen bonds |
|
|
How is DNA adapted for its function? |
Sugar Phosphate backbone for Strength Coiling of DNA makes it compact Double Helix means 2 strands can be templates, protects base sequence, makes molecule more stable Large molecule to store all genetic information Many hydrogen bonds give stability and allow it to be unzipped for replication Sequence of bases allow genes to code for specific proteins Complementary base pairing enables accurate and easy replication |
|
|
Outline the Biuret test |
Add an alkali to the sample such as Sodium Hydroxide Add Biuret reagent. If it goes from blue to purple protein is present |
|
|
Outline Benedicts Test |
A sample is mixed with a small amount of Benedict's solution.
This is heated in a water bath. If a brick red or orange precipitate is seen, it is positive for reducing sugar. If it stays blue, no reducing sugar. |
|
|
Outline Benedicts Test for a non-reducing sugar (sucrose) |
Carry out Benedicts Test to confirm a negative result. Heat another sample with acid to hydolyse it. Neutralise this sample using an alkali e.g NaOH. Add benedicts to the hydrolysed sample and heat in water bath. Look for brick red. |
|
|
What is the difference between Semi Quantitative Benadicts Test and Quantitative Benadicts Test? |
Semi Quantative looks at the colour or amount of precipitate but is subjective as different people see different things.
Quantitative uses a colorimeter to measure absorbency of known concentrations and then compare these to the unknown using a calibration curve. Is more accurate as uses numerical data and is objective. |
|
|
When would a Quantitative Benadicts test be done?
|
To compare the concentration of reducing sugar in multiple different solutions. |
|
|
Why is starch suitable as a Storage Molecule? |
|
|
|
Why is Glycogen suitable as a Storage Molecule? |
|
|
|
How does Cellulose differ from Glycogen and Starch and how is it suitable for its purpose? |
It is formed from monomers of Beta Glucose instead of Alpha. This means it forms straight unbranched chains. Suitable for its Purpose Because:
|
|
|
How can proteins be broken down? |
Hydrolysis by heating with acid By Enzymes at Optimum Temperature (proteases / peptidases) |
|
|
How does Denaturation of Proteins occur? |
The Hydrogen and Ionic bonds which cause the protein to be folded into the correct shape break. This causes the shape of the protein to shape and can cause it to become non-functional. This can happen due to High Temperature, Extreme pH changes or Heavy Metals (which disrupt disulfide bridges. |
|
|
What are Enzymes and what do they do?
|
They are Biological Catalysts which lower the activation energy of chemical reactions and so increase the rate of reaction. They do this by forming Enzyme-Substrate complexes and then weakening bonds to allow them to break or react with other compounds. |
|
|
What is the Induced Fit model of Enzyme Action? |
Enzyme has a unique active site based on tertiary structure which are specific and will only bind to certain substrates to form enzyme substrate complexes BUT the enzyme active site is not a complementary shape when it is not bound, it is flexible and changes its shape once they bind to become complementary. |
|
|
What is the difference between Competitive Inhibitors and Non-Competitive inhibitors? |
Competitive have a similar structure to the substrate and compete for attachment to the active site. Non-Competitive do not have a similar structure but attach to a site on the Enzyme which causes the tertiary structure and shape of the active site to change. This prevents substrate from binding to the active site and forming enzyme-substrate complexes. |
|
|
|
|

