![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
29 Cards in this Set
- Front
- Back
|
Chemical losses |
The primary chemical cleansing agent and the troposphere is the hydroxyl radical |
|
|
Dry and wet deposition |
Relative importance depends on, the phase of the substance gas or particle. The solubility at the substance in water. The mount a precipitation in the region. Terrain and type of surface cover |
|
|
Dry deposition depends on |
Turbulence at surface. For gasses, solubility and reactivity play a role. For particles, size density and shape. Properties of the surface |
|
|
Wet deposition requires |
Interception. Scavenging. Delivery |
|
|
Fundamental principle conversion of mass |

|
|
|
Chemical transformation, VOC |
VOC--> CO2 + H2O |
|
|
Burning of carbon based fuels |

|
|
|
National ambient air quality standards |
Control based on air quality criteria, 2 types primary which is health based and secondary which is welfare based And States must prepare implementation plans (sips) |
|
|
Hazardous air Pollutants (HAPS) |
Put in s'more hazardous thing criteria pollutants but are difficult to determine safe level the sources are often localized exposure |
|
|
Classification of sources |
Primary are emitted to the atmosphere and secondary are formed in the atmosphere |
|
|
Carbon monoxide source |
Incomplete combustion of carbon fuels. Sink : CO + OH --> CO2 + H |
|
|
National carbon monoxide a missions by source sector |
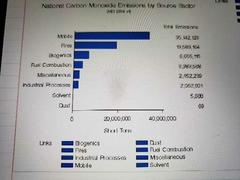
|
|
|
The global atmospheric processing of reactive nitrogen |
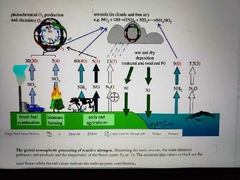
|
|
|
Global sources of nitrogen oxide |
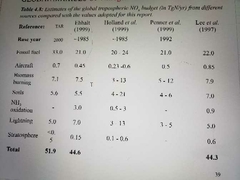
|
|
|
Trends and politicians over time |

|
|
|
Volitile organic compounds VOC |
Compounds primarily composed of hydrogen and carbon period of volitile meaning high propensity to be in a gaseous state Examples include methane benzene and isoprene |
|
|
Biogenics VOC emissions sensitive to |
Temperature, my, vegetation type and canopy structure |
|
|
National volatile organic compounds emissions by source |
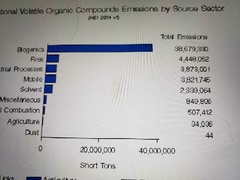
|
|
|
Well tile organic compound emissions by sources In multnomah county |
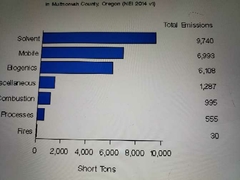
|
|
|
Build-up of ozone |
Is due to the chemical reactions of hydrocarbons and nitrogen oxides in the presence of sunlight |
|
|
Ozone formation and the troposphere |

|
|
|
Formation of ozone with NO |

|
|
|
Tropospheric oxidation of general Hydrocarbon |

So more and 02 is produced that makes more ozone |
|
|
Formation of ozone simplified |

|
|
|
Diurnal variations in precursors and photochemical oxidants |
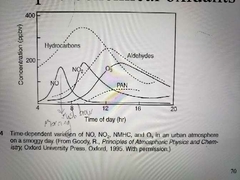
|
|
|
Modeling ozone formation |
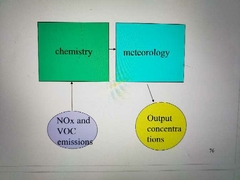
|
|
|
Urban air shed modeling |
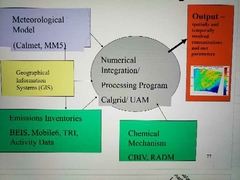
|
|
|
Anthropogenic nox emissions in the US by source category |
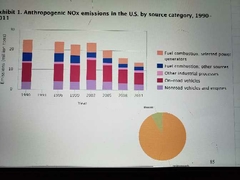
|
|
|
Ozone concentrations versus nox and VOC emissions |

Ozone reduction and control is complicated reducing one or both could do nothing or even increase ozone. Nox creates and destroys ozone so relationship None linear |

