![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
53 Cards in this Set
- Front
- Back
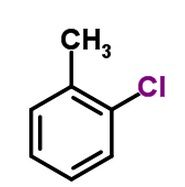
What is the functional group( black) attached to the ring? b. ketone d. methyl f. amine h. phosphate |
Methyl |
|
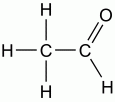
What is the functional group? b. ketone d. methyl f. amine h. phosphate
|
Aldehyde |
|
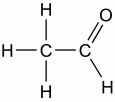
This functional group is |
polar
|
|
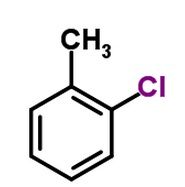
This functional group (black) is |
non-polar |
|
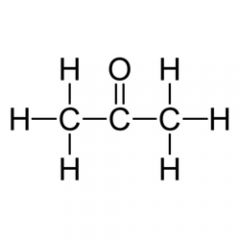
What is the functional group? a. aldehyde b. ketone d. methyl f. amine h. phosphate |
Ketone
|
|
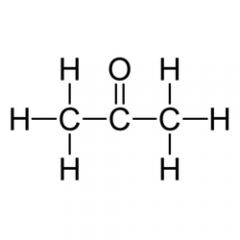
This functional group is |
polar |
|
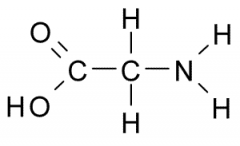
The functional group on the right is |
basic
|
|
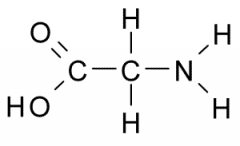
The functional group on the right is _______ charged when it ionizes. |
positively
|
|
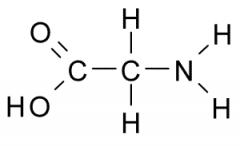
The functional group on the left is _______ charged when it ionizes.
a. negatively b. positively |
negatively
|
|
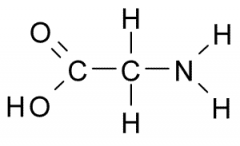
The functional group on the left is |
acidic
|
|
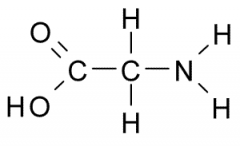
The functional group on the left is a(an) a. aldehyde b. ketone d. methyl f. amine h. phosphate |
carboxyl
|
|
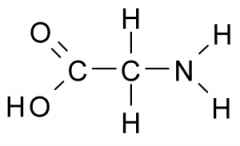
The functional group on the right is a(an) a. aldehyde b. ketone d. methyl f. amine h. phosphate |
amine |
|
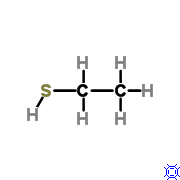
What is the functional group? a. aldehyde b. ketone d. methyl f. amine h. phosphate |
Sulhydryl |
|
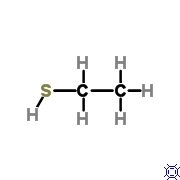
Because this molecule has a sulfhydryl group it is a __________.
|
thiol |
|
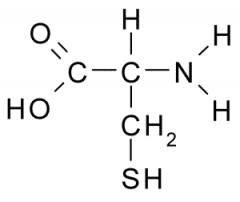
The sulfhydryl groups between two cysteines (amino acids) ) forms connecting bridges called _________.
|
disulfide bridges |
|
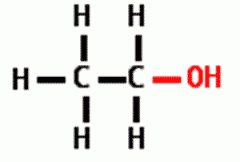
What is the functional group? a. aldehyde b. ketone d. methyl f. amine h. phosphate |
Hydroxyl |
|
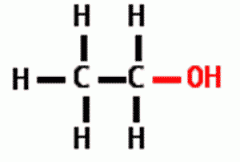
This functional group is |
polar |
|
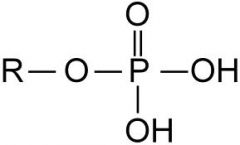
This functional group is |
acidic |
|
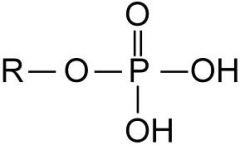
What is the functional group? a. aldehyde b. ketone d. methyl f. amine h. phosphate |
phosphate
|
|
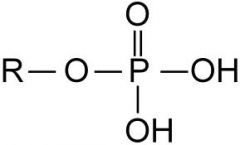
The functional group is _______ charged when it ionizes.
a. negatively b. positively |
negatively
|
|
|
The branch of chemistry that specializes in the study of carbon compounds is ______ |
Organic chemistry
|
|
|
Molecules containing carbon are ____ |
Organic molecules |
|
|
Carbon can form ____ covalent bonds with a variety of atoms |
four
|
|
|
Each carbon bonded to four other atoms has a ____ shape |
tetrahedral
|
|
|
Molecules containing only hydrogen and carbon. are ________. |
Hydrocarbons
|
|
|
Compounds with the same molecular formula but with different structures and hence different properties are ______ |
Isomers
|
|
|
Isomers that have the same molecular formula but differ in the covalent arrangement of their atoms are _____ |
Structural isomers |
|
|
Isomers which share the same covalent partnership, but differ in their spacial arrangement around a carbon-carbon double bond are _____. |
Geometric (cis-trans) isomers
|
|
|
Isomers that are mirror images of each other are _____. |
Stereoisomers (Enantiomers)
|
|
|
_________ are the components of organic molecules that are most commonly involved in chemical reactions. |
Functional groups
|
|
|
_____ is a functional group that consists of a hydrogen atom bonded to an oxygen atom, which in turn forms a polar covalent bond to carbon. Found in alcohols. |
Hydroxyl group
|
|
|
____ is a functional group that consists of a carbon atom double-bonded to oxygen. |
Carbonyl group
|
|
|
____ is a functional group that is a carbonyl within a carbon chain |
Ketone |
|
|
____ is a functional group that is a carbonyl on a terminal carbon |
Aldehyde |
|
|
___ is a functional group that consists of a carbon atom that is both double-bonded to an oxygen and single bonded to the oxygen of a hydroxyl group. Acts as an acid. |
Carboxyl group
|
|
|
____ is a functional group that consists of a nitrogen atom bonded to two hydrogens and to the carbon skeleton. Acts as a base. |
Amino group |
|
|
___ is a functional group that consists of an atom of sulfur bonded to an atom of hydrogen. Forms disulfide bridges between two cysteine amino acids. |
Sulfhydryl group (Thiol)
|
|
|
____ is a functional group which is the dissociated form of phosphoric acid which consists of phosphorus bound to four oxygen atoms (three with single bonds and one with a double bond). Acts as an acid. |
Phosphate group
|
|
|
_____ is a non-polar functional group that is a carbon and surrounding hydrogens. |
Methyl group
|
|
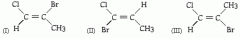
Which 2 molecules are geometric isomers? |
I and III |
|
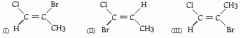
What type of isomers are molecules I and II? A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Structural (not cis/trans) |
|
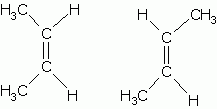
These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Geometric |
|

These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Geometric |
|
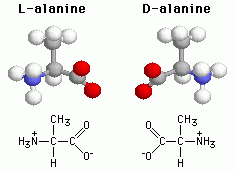
These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Enantiomers |
|
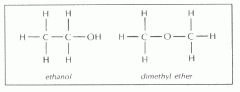
These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Structural |
|
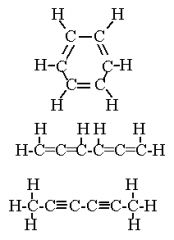
These three isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Structural |
|
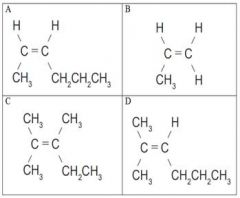
Which two isomers are are structural isomers? |
C and D |
|
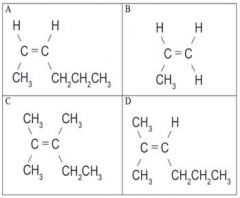
Which two isomers are are geometric isomers? |
None are geometric isomers |
|
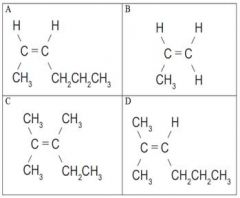
Molecule A and C are A. Structural
B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
not isomers |
|
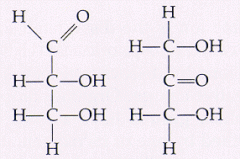
These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Structural |
|
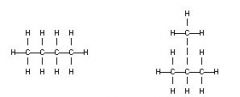
These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Structural |
|
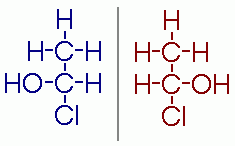
These two isomers are A. Structural B. Geometric (cis-trans) C. Enantiomers D. The same molecule E. Not isomers |
Enantiomers |
|
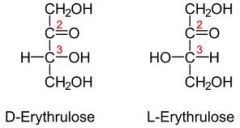
Which of the four carbons are asymmetric (chiral)?
|
Carbon #3 |

