![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
58 Cards in this Set
- Front
- Back
|
Chemical Bonds
|
Covalent, Ionic, Hydrogen
|
|
|
Atomic particles
|
Protons(+) and Neutrons (no charge) found in nucleus, electrons(-)
|
|
|
Covalent bonds
|
share a pair of electrons between 2 atoms
more stable bonds |
|
|
nonpolar covalent
|
electrons shared equally
|
|
|
polar covalent
|
one atom is more electronegativily charged which causes the electron to move closer to one atom - so one side of molecule is more positive and one more negative
|
|
|
Ionic bonds
|
2 or more ions bonded together due to their opposite charge
NaCl sodium(+) cloride(-) |
|
|
Ions
|
an atom whose # of electrons differs from its # of protons.
when an atom gains or loses electrons. |
|
|
Hydrogen bonding
|
linkage of an already covalently bonded H atom w an electronegative atom.
Normally a linkage that nearly always pairs H w/ O or N. |
|
|
Carbon
|
Life based on Carbon
4 valance electrons links w/ 4 more electrons to be stable Covalent bonds straight chains Ring structures |
|
|
4 atoms that make up living things
|
Carbon
Hydrogen Oxygen Nitrogen |
|
|
4 Molecules of life
|
Carbs - CH2O - proportion
Lipids - fats Proteins - amino acids, peptide bonds Nucleic acids |
|
|
Building blocks *
|
monomers - smaller building blocks that link together to create large molecules
polymers- large chains of monomers polysaccharide - many monosaccharides units together |
|
|
Carbs *
|
monosaccharides - glucose
disaccharides - succrose, lactose polysaccharides - starch glycogen, cellulose, chitin |
|
|
Components of carbs
|
Carbon
Hydrogen 2 Oxygen |
|
|
carbs are stored as ________
used as ___________ |
glycogen
glucose |
|
|
Lipids made of
|
Carbon. hydrogen, oxygen
|
|
|
lipids - long definition
|
pure lipids are not readily broken down.
very high energy source stored in triglyceride form Used in fatty acid and glycerol form |
|
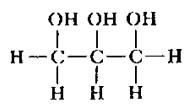
|
glycerol
|
|
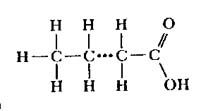
|
fatty acid
|
|
|
types of lipids
|
glycerides
steroids phospholipids |
|
|
Glycerides made of 2 parts *
|
glycerol-specific kind of alcohol, makes head of molecule, contain OH group
fatty acid (long chain of carbon and Hydrogen atoms) w/ carboxyl group (COOH) |
|
|
Triglycerides
|
3 fatty acids linked w a glycerol
most important of the glycerides 90% of lipid weight in foods normally known as "fats" |
|
|
Types of fat
|
saturated fatty acid - no double bonds
mono-unsaturated fatty acid - 1 double bond poly-unsaturated - multiple double bonds |
|
|
saturated fatty acid & health
|
Linked to heart disease
great source of energy valuable source of insulators in cold weather need to be taken in limited amounts increases blood cholesterol |
|
|
saturated fatty acid (alkanes)
|
saturated w/ hydrogen atoms
no double bonds normally solid at room temps found in animal products and tropical oils |
|
|
Unsaturated fats (alkenes)
|
one or more double bonds between the carbon atoms of its hydrogen chain
|
|
|
Steroids
|
linked w 4 carbon rings (cyclic hydrocarbons)
well-known ones are cholesterol, testosterone and estrogen |
|
|
cholesterol
|
part of outer membrane of many cells
component of bile which emulsifies fat in small intestine precursor for testosterone & estrogen |
|
|
Phospholipids
|
glycerol head
2 fatty attached - hydrophobic linked to 3rd OH group is a phosphate group instead of the 3rd fatty acid as in triglycerides; outer membrane of cells largely made of phospholipids - hydrophilic |
|
|
proteins
|
enzymes
hormones transport contractile protective structural storage toxins communication |
|
|
enzymes
|
biological catalyst
speed up or enables chemical reactions in living things Lactase Sucrase Lipase |
|
|
Hormones
|
chemical messengers which prompts physiological activity w/in and organism, both plants and animals.
Thyroxin Estrogen Testosterone |
|
|
Amino acids - short answer
|
building block of proteins
polypeptides are made up of chains of amino acids 20 amino acids in nature |
|
|
structure of an amino acid
|
amino group
central carbon carboxyl group (COOH) -R side chain which differentiates one amino acid from another |
|
|
amino acid
|
each amino acid has at least 1 amine.
1 acid functional group as the name implies. The different properties result from variations in the structures of different R groups. The R group is often refered to as the amino acid side chain |
|
|
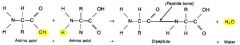
Chaining of amino acids
|
carboxyl group one amino acid joins to the amino group of the next w/ the loss of the water molecule
|
|
|
Protein Structure *
|
"form follows function"
Primary structure Secondary structure Tertiary structure Quaternary structure |
|
|
Denature
|
Breakdown, unfolding or altering of shape of proteins:
-PH acid, alcohol -Heat |
|
|
Metabolism
|
sum of all the chemical reactions that a cell or larger organism carries out
|
|
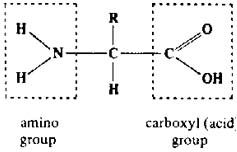
|
amino acid
|
|
|
dhscarbo
|
|

|
dhs lipid
|
|
|
dhs protien 2
|
|
|
metabolic pathway
|
a+b then c+d then e+f ....
sequentioal set of enzymatically controlled reactions where product of one reaction serves and the substrate for the next reaction. |
|
|
Activation energy
|
energy required to initiate chemical reaction
enzymes lower activation energy needed active site - place in enzyme where substrate binds and is transformed coenzyme helps catalyze substrates |
|
|
Enzymes
|
facilitate chemical process in living things.
accelerate chemical reactions that would happen anyway. Enable reactions because living things can wait for reactions to occur naturally Catalysts-retain their original chemical composition |
|
|
Coenzymes
|
accessory molecule part of the activation site of an enzyme and allows it to function.
vitamins are one type |
|
|
regulation of enzymes
|
Amount of substance
Negative feed back - product of a process that reduces the activity. Allosteric regulation - product of a reaction binds to the enzyme |
|
|
Oxidation
|
loss of electrons
electrons carriers molecules that shuttle electrons down the energy hill - NAD |
|
|
Reduction
|
Gain of electrons
|
|
|
3 cycles of respiration
|
Glycolysis
Krebs cycle Electron transport chain |
|
|
Adenosine Triphosphate
|
nitrogen containing molecule w/ 3 phosphate groups attached.
produced in cell by breakdown of glucose. ATP used in cell by splitting and one phosphate group removed. |
|
|
Glycolysis
|
conversion of glucose into pyruvate w/ production of ATP
|
|
|
Lactic acid production
|
o not avail
Hyd dropped back to pyruvic acid to form lactic a some diffuses out of cell to LV O is present LA converted back into pyruvic acid |
|
|
ETS electron transport system
|
accepts energy from carrier in the matrix and stores it to a form that can be used to phosphorylate ADP.
two energy carriers are known to donate to ETS - NAD & FAD |
|
|
ETS - NADH binds to a
|
prosthetic group called FMN and is reoxidized to NAD.
NAD is recycled acting as an energy shuttle |
|
|
ETS - What happens to hydrogen that comes off the NADH?
|
FMN gets H + 2 electrons
also picks up a proton from the matrix - passes electrons to iron-sulfer clusters and forces two protons into intermembraneous space. |
|
|
Proton Gradient
|
built up as a result of NADH feeding electrons into ETS
|

