![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
234 Cards in this Set
- Front
- Back
|
heterocyclic cmpds
|
cmpds w rings that contain >1 element
|
|
|
heteroatoms
|
atoms other than C
|
|
|
altho chem of many sat heterocyclic cmpds is analogous to that of noncyclic counterparts
|
significant number unsat heterocyclic cmpds exhibit aromatic behavior
|
|
|
nomenclature of aromatic heterocycles
|
heteroatom given number 1 unless isoquinoline, O & S given lower # than N
|
|
|
substituent groups are given
|
the lowest # consistent w the ring numbering
|
|
|
resonance hybrids of furan
|
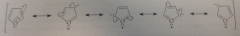
bc separation of charge is present in all but the first structure, the first is more important, but others evident in comparison of dipole moments of furan & tetrahydrofuran a sat heterocyclic ether
|
|
|
dipole moment of tetrahydrofuran attributable mostly to
|
bond dipoles of its polar C-O sb (e in the sigma bonds are pulled toward the O bc of its electroneg)
|
|
|
Same effect present in furan plus
|
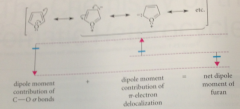
resonance delocalization of O unshared e into ring tends to push e away from the O into the pi-e system of the ring - two effects cancel & furan has small dipole moment
|
|
|
pyridine structures of less importance reflect relative electroneg of N & C
|
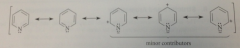
|
|
|
vinylic heteroatoms such as N of pyridine contribute
|
1 pi e to the 6 pi e aromatic system just like each of C atoms in pi system
|
|
|
orbital containing unshared e pair of the pyridine N
|
perpendicular to the 2p orbitals of the ring and is not involved in pi bonding
|
|
|
allylic heteroatoms such as N of pyrrole contribute
|
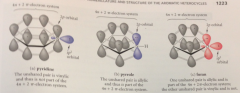
2 e unshared pair to aromatic pi e system
|
|
|
N adopts sp2 hybridization & trigonal planar geometry
|
its unshared e pair can occupy a 2p orbital which has optimum shape & orientation to overlap w the C 2p orbitals & thus to be part of the aormatic pi-e system so H of pyrrole lies in the plane of the ring
|
|
|
O of furan contributes
|
1 unshared e pair to the aromatic pi e system, & other unshared e pair occupies position analogous to C-H bond of pyrrole in the ring plane perpendicular to the 2p orbitals of the ring
|
|
|
empirical resonance E can be used to
|
estimate the additional stability of a heterocyclic cmpd due to its aromaticity
|
|
|
to the extent that resonance E is a measure of aromatic character
|
furan has the least aromatic character of the heterocyclic cmpds
|
|
|
pyridine & quinoline act as
|
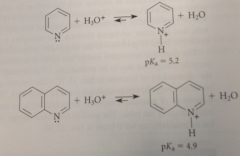
ordinary amine bases
|
|
|
pyridine & quinoline are much ___ basic than aliphatic tertiary amines bc
|
less, bc of sp2 hybridization of the N unshared e pairs
|
|
|
pyrrole and indole are protonated
|
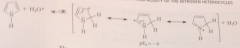
only in strong acid & protonation occurs on C, not N
|
|
|
marked contrast btwn basicities of pyridine & pyrrole can be understood by
|
considering role of N unshared e pair in the aromaticity of each cmpd
|
|
|
protonation of the pyrrole N would disrupt the aromatic system of 6 pi e
|
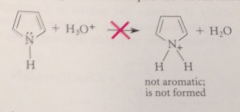
by taking the N unshared pair out of circulation
|
|
|
altho protonation of the C of pyrrole also disrupts the aromatic pi-e system
|
the resulting cation is resonance stabilized
|
|
|
protonation of the pyridine unshared e pair occurs easily bc
|
this e pair is not part of the pi e system, so protonation does not disrupt pyridine's aromaticity
|
|
|
how acidic are pyrrole & indol?
|
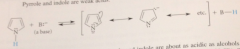
weakly
|
|
|
greater acidity of pyrroles and indoles over primary, secondary amines is a consequence of
|
resonance stabilization of their conj base anions
|
|
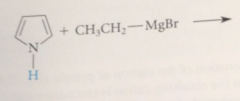
|
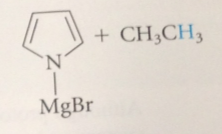
|
|
|
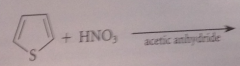
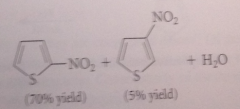
do furan, thiophene & pyrrole undergo EAS?
|
Yes
|
|

|
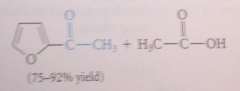
|
|
|
|
|
|
pyrrole, furan & thiophene are all much
|
more reactive than benzene in EAS
|
|
|
relative rates of bromination
|
pyrrole > furan > thiophene > benzene
|
|
|
milder rxn conditions must be used w
|
more reactive cmpds - too vigorous bring about side rxns so polymerization & tar formation occur
|
|
|
Less reactive acylating reagent
|
used in acylation of furan than in acylation of benzene
|
|
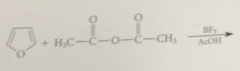
|
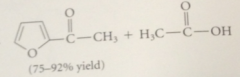
|
|
|
reactivity order of heterocycles
|
is a consequence of the relative abilities of heteroatoms to stabilize pos charge in intermediate carbocations
|
|
|
pyrrole & furan have heteroatoms
|
from the 2nd period of the periodic table
|
|
|
Bc N is better than O @ delocalizing pos charge (N is less electroneg)
|
pyrrole is more reactive than furan
|
|
|
S of thiphene is 3rd period element, and tho less electroneg than O
|
3p orbitals overlap less efficiently w 2p orbitals of aromatic pi e system
|
|
|
Reactivity order of heterocycles in aromatic sub
|
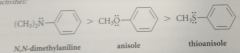
parallels reactivity order of correspondingly sub benzene derivatives
|
|
|
superimposed on normal activating & directing effects of substituents in aromatic sub
|
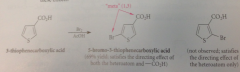
is normal effect of heterocyclic atom in directing substitution to the 2-position
|
|
|
-CO2H group directs the 2nd substituent into a
|
meta relationship, thiophene ring tends to sub @ 2-position
|
|

..
|

count around the C framework of the heterocyclic cmpd, not thru the heteroatom
|
|

when directing effects of substituents & ring compete
|

not unusual to observe mixtures of prod
|
|

if both 2-positions are occupied
|

3-substitution takes place
|
|
|
implication of furan's small resonance E
|
least aromatic character - greatest tendency to behave like a conj diene
|
|
|
diff to prep monosub pyridines by EAS bc
|
pyridine has low reactivity (less reactive than benzene)
|
|
|
reason for pyridine low reactivity
|
protonated under very acidic conditions of most EAS rxns - resulting pos charge on N makes it diff to form carbocation intermediate, which would place 2nd pos charge within same ring
|
|
|
number of monosub pyridines are available from
|
natural sources
|
|
|
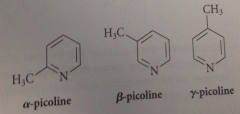
methylpyridines or picolines
|
|
|
|
picoles & other methylated pyridines are obtained from
|
coal tar
|
|
|
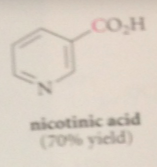
useful monosub derivative of pyridine
|
nicotinic acid (pyridine-3-CA)
|
|
|
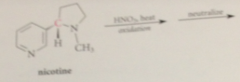
how is nicotinic acid prep
|
side-chain oxidation of nicotine, an alkaloid present in tobacco
|
|
|
|
|
|
altho EAS not very useful for intro sub into pyridine itself
|
pyridine rings sub w activating groups such as methyl groups do undergo such rxns
|
|
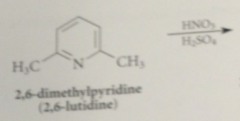
|
|
|
|
rxns which occur in acidic soln
|
take place on very small amt of unprotonated pyridine that is in rapid equil w larger amt of conj acid
|
|
|
bc reactive unprotonated heterocycle is present in very small conc
|
methyl-sub pyridines are not very reactive, despite presence of activating methyl sub
|
|
|
sub in pyridine generally takes place in the
|
3-position
|
|
|
3-sub reason
|
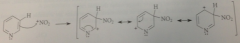
carbocation intermed formed in sub has 3 diff resonance structures
|
|
|
sub at 4-position also involves carbocation intermed w 3 resonance structures
|
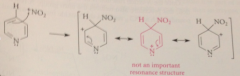
but one shown in red is particularly unstable & unimportant bc the N, an electroneg atom, is e-deficient
|
|
|
pyrolle N is pos charged but not e-deficient bc
|
it has a complete octet
|
|
|
e-deficient electroneg atom is very
|
unfavorable energetically
|
|
|
how to obtain pyridine derivatives sub @ other positions
|
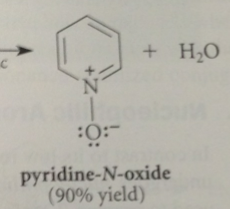
for 4-sub, use pyridine-N-oxide, formed by oxidation of pyridine w 30% hydrogen peroxide
|
|
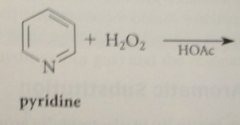
|
|
|
|
bc N of pyridine-N-oxide has a pos charge
|
cmpd is much less reactive than phenol or phenoxide but pyridine-N-oxide undergoes useful aromatic sub @ 4
|
|
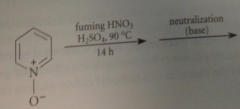
|
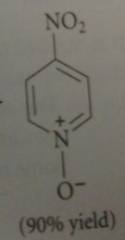
|
|
|
once N-oxide func no longer needed
|
can be removed by cat hydrogenation (reduces nitro group)
|
|
|
rxn w trivalent phosphorus cmpds such as PCl3
|
removes N-oxide func w/o reducing nitro group
|
|
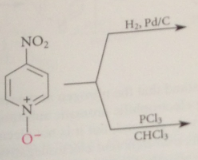
|
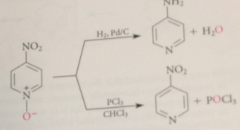
|
|
|
pyridine ring readily undergoes
|
NAS
|
|
|
Chichibabin rxn
|
treatment of pyridine derivative w strong base sodium amide brings about direct sub of amino group for ring hydrogen
|
|
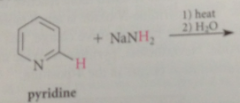
|
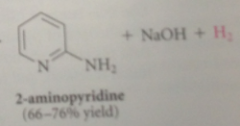
|
|
|
first step of mech
|
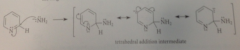
amide ion reacts as nuc @ 2-position of ring to form tetrahedral addition intermed
|
|
|
C=N linkage of the pyridine ring
|
somewhat analogous to a carbonyl group (C @ 2 has some character of a carbonyl C, can react w nuc)
|
|
|
C=N of pyridine is much ___ reactive than a carbonyl group
|
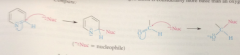
less, bc part of aromatic system, N anion more basic than an O anion
|
|
|
2nd step of mech
|
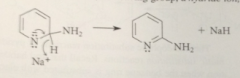
LG, hydride ion, lost
|
|
|
Hydride ion
|
poor so unusual LG, very basic
|
|
|
Rxn occurs bc
|
aromatic pyridine ring reformed, aromaticity lost in formation of tetrahedral addition intermediate regained when LG departs + basic hydride prod in rxn reacts w NH2 group irreversibly to form dihydrogen & resonance stabilized conj base anion of 2-aminopyridine
|
|
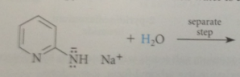
|
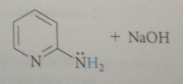
|
|
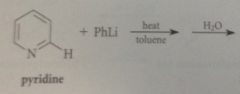
|
|
|
|
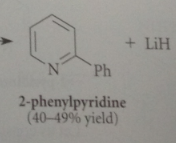
when pyridine is sub w better LG than hydride @ 2
|
reacts more rapidly w nuc
|
|
|
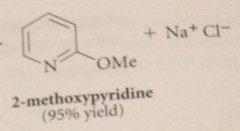
|
|
|
nuc sub can be related to analogous rxn of carbonyl cmpd
|
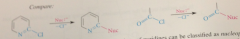
acid chlorides much more reactive than 2-halopyridines
|
|
|
EWG in rxns of pyridines
|
pyridine N itself
|
|
|
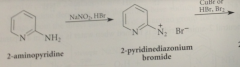
tetrahedral addition intermed
|
analogous to Meisenheimer complex of NAS
|
|
|
|
|
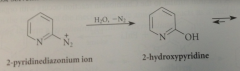
|
|
|
|
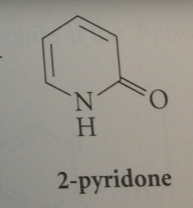
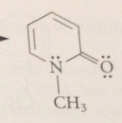
equil btwn 2-hydroxypyridine & 2-pyridone
|
analogous to keto-enol equil bt keto form is amide
|
|
|
ratio of hydroxy form to carbonyl
|
1:910 in water, but varies w conc & solvent
|
|
|
enough of each form present so that either can be involved in chem rxns, much more
|
carbonyl isomer is present than in phenol
|
|
|
what determines if aromatic hydroxy cmpd exists as carbonyl or hydroxy form
|
whether E advantage of aromaticity (resonance stabilization of aromatic hydroxy isomer) outweights large carbonyl C=O bond E
|
|
|
phenol
|
resonance stabilization of benzene ring large enough that phenol isomer strongly preferred
|
|
|
resonance E thus stabilization of pyridine
|
considerably smaller than benzene
|
|
|
resonance interaction of amide N w carbonyl group
|
further stabilizes 2-pyridone
|
|
|
resulting resonance structure
|
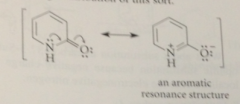
aromatic
|
|
|
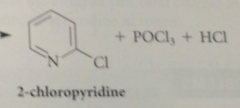
|
|
|

|
|
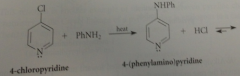
|
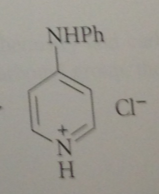
|
|
|
nuc sub rxns @ 2/4 positions of pyridine ring
|
particularly common - neg charge in addition intermediate delocalized onto electroneg pyridine N
|
|
|
sub at C 2
|
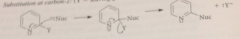
|
|
|
sub at C 4
|
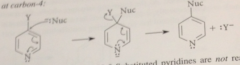
|
|
|
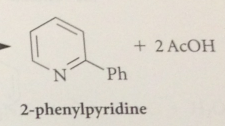
3 sub pyridines not reactive in nuc sub bc
|
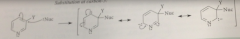
neg charge in addition intermed cannot be delocalized onto electroneg N
|
|
|
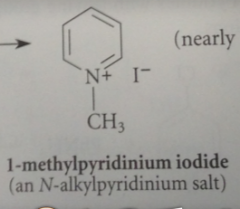
|
|
|
N-alkylpyridinium salts are activated toward nuc rxns @ 2, 4 positions of the ring more than pyridines bc
|
pos charged N is more electroneg, so better e acceptor, than neutral N of a pyridine
|
|
|
When nuc in displacement rxns are anions
|
charge is neutralized
|
|
|
|
|
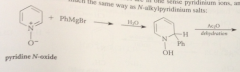
|
|
|
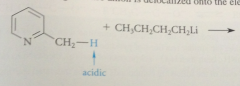
|
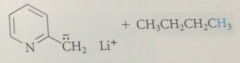
|
|
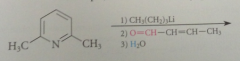
|
|
|
|
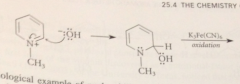
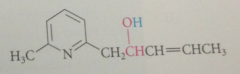
benzylic H of 2 / 4 alkylpyridinium salts are much more acidic than those of analogous pyridines
|
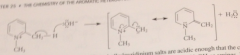
bc conj base anion is actually a neutral cmpd
|
|
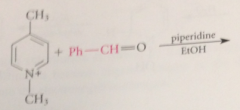
|
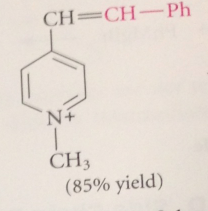
conj base of a pyridinium salt is used as the enolate component in a variation of the Claisen-Schmidt condensation
|
|
|
benzylic H of 2/4 alkylpyridinium salts acidic enough that conj base anions
|
can be formed in useful conc by aq. NaOH or amines
|
|
|
many side-chain rxns of pyridines
|
analogous to those of corresponding benzene derivatives
|
|
|
N of pyridine ring can serve as
|
acceptor of e, particularly enhanced in pyridinium ions
|
|
|
pyridinium ion strongly activated toward
|
rxns w nuc (e flow onto pos charged N)
|
|
|
pos charged N of the pyridinium ion serves to
|
stabilize the attached carbanion by resonance
|
|
|
Pyridinium ion of NAD+ serves as
|
an e acceptor in biochem rxns
|
|
|
important pyridine derivative
|
pyridoxal phosphate - one of several forms of vitamin B6
|
|
|
pyridoxal phosphate
|
essential coenzyme in several important biochem transformations
|
|
|
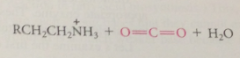
utilized for prod of bio important amines i.e. neurotransmitters serotonin & dopamine in human brain & vasoconstrictor histamine
|
|
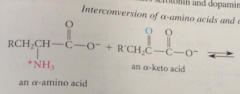
|
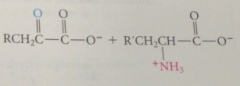
|
|
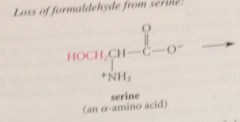
|
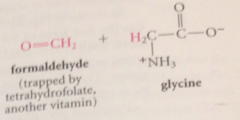
important source of single C units for bio processes that involve single C transfer
|
|
|
In bio systems
|
each rxn catalyzed by pyridoxal phosphate & appropriate enzyme
|
|
|
pyridoxal phosphate exists in the bio world as
|
imine derivatives in which it is covalently attached to various enzymes
|
|
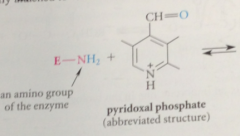
|
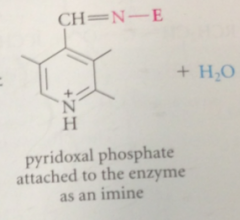
|
|
|
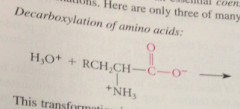
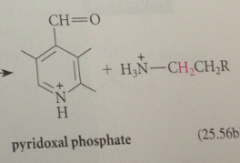
in the first step of all pyridoxal-cat rxns of a-amino acids
|
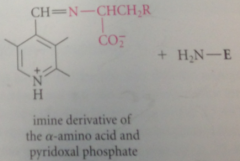
amino group acting as nuc in its unprotonated form, forms an imine w pyridoxal phosphate (exactly like imine formation from an amine & aldehyde except rxn of the amino group is w C=N bond of imine rather than w C=O bond of an aldehyde
|
|
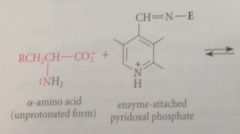
|
|
|
|
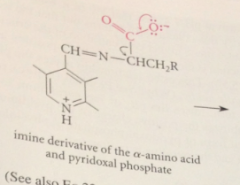
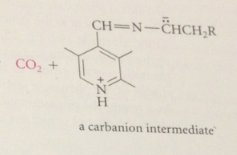
decarboxylation yields
|
carbanion intermediate
|
|
|
|
|
|
most carbanions- such strong bases that they cannot exist under phys conditions but this carbanion much weaker B bc
|
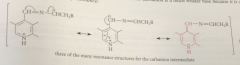
stabilized by resonance
|
|
|
pyridinium ion stabilizes negative charge by
|
accepting e
|
|
|
carbanion not really carbanion at all
|
neutral mlc
|
|
|
same type of carbanion involved in
|
all pyridoxal-cat transformations
|
|
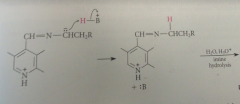
protonation of anion & hydrolysis of resulting imine gives
|
pyridoxal phosphate
|
|
|
typical pKa of pyridinium ions
|
5 yet rxns promoted by pyridoxal phosphate take place at phys pH values ~ 7.4
|
|
|
If pyridinium ion in pyridoxal phosphate had pKa near 5
|
most would exist in conj B pyridine form at pH 7.4 - less than 1% would exist in conj A pyridinium ion form
|
|
|
mlclr architecture of pyridoxal phosphate
|
ensures much higher conc of crucial pyridinium ion form
|
|
|
key element in structure
|
OH group in the 3 position & ortho relationship to aldehyde
|
|
|
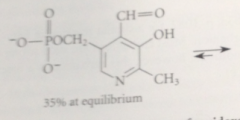
Ortho relationship makes
|
phenolic OH group of pyridoxal phosphate unusually acidic
|
|
|
Ionization of the phenolic OH group
|
raises the pKa of the pyridinium ion bc neg charge of the phenolate stabilizes the pos charge of the pyridinium ion & vice versa => predominant form of pyridoxal phosphate at phs pH is form in which phneol is ionized & pyridine is protonated
|
|
|
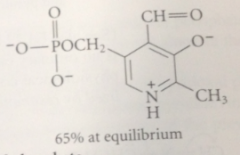
|
|
|
when pyridoxal phosphate is bound to the enzymes that cat its rxns
|
the pyridinium form is further stabilized
|
|
|
this stabilization is the result of
|
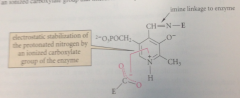
an ionized carboxylate group that interacts directly w pos charged N
|
|
|
ribonucleoside
|
cmpd formed btwn furanose form of D-ribose & heterocyclic cmpd
|
|
|
heterocyclic group
|
base
|
|
|
ribose
|
sugar
|
|
|
stereochem of bond btwn base & ribose most commonly
|
B
|
|
|
deoxyribonucleoside
|
similar derivative of D-2-deoxyribose & a heterocyclic base
|
|
|
prefix deoxy
|
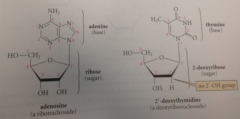
w/o oxygen
|
|
|
sugar ring & heterocyclic ring
|
numbered separately
|
|
|
how to differentiate the 2 sets of #s
|
primes used in referring to atoms of the sugar
|
|
|
2' C of adenosine
|
C-2 of the sugar ring
|
|
|
bases that occur most frequently in nucleosides
|
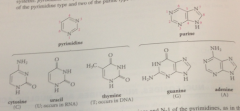
derived from 2 heterocyclic ring systems: pyrimidine & purine
|
|
|
base attached to the sugar @
|
N-9 of purines & N-1 of pyrimidines
|
|
|
in living systems, 5' OH group of ribose in a nucleoside
|
usually found esterified to a phosphate group
|
|
|
nucleotide
|
5' phosphorylated nucleoside
|
|
|
ribonucleotide
|
derived from the monosaccharide ribose
|
|
|
deoxyribonucleotide
|
derived from 2-deoxyribose
|
|
|
nucleotides either contain
|
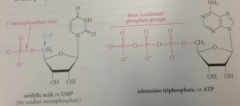
single phosphate group or 2/3 phosphate groups condensed in phosphoric anhydride linkages
|
|
|
altho ionization state of phosphate groups depends on pH
|
groups written conventionally in ionized form
|
|
|
to name corresponding 2' deoxy derivatives from ribonucleosides & ribonucleotides
|
prefix 2'-deoxy appended to names of corresponding ribose derivatives
|
|
|
AMP
|
adenosine monophosphate
|
|
|
dTMP
|
2'-deoxythymidylic acid
|
|
|
NAD+ & coenzyme A
|
ribonucleotides
|
|
|
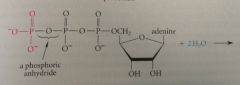
ATP
|
anhydride of phosphoric acid
|
|
|
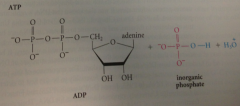
hydrolysis of anhydrides
|
very favorable rxn
|
|
|
|
|
|
overall process for muscle contraction
|
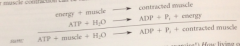
|
|
|
nucleic acids
|
polymers of nucleotides
|
|
|
deoxyribonucleic acid
|
polymer of deoxyribonucleotides - storehouse of genetic info thu all nature
|
|
|
residues
|
monomeric units of DNA polymer
|
|
|
nucleotide residues in DNA
|
interconnected by phosphate groups that are esterified both to the 3' OH group of one ribose & 5' OH of another
|
|
|
DNA polymer incorporates
|
adenine, thymine, guanine, cytoside as nucleotide bases
|
|
|
typical strand of DNA
|
thousands of mill nucleotides long
|
|
|
Each residue in a polynucleotide differentiated by
|
identity of its base & sequence of bases encodes genetic info in DNA
|
|
|
DNA polymer
|
backbone of alternating phosphates & 2'-deoxyribose groups to which are connected bases that differ from residue to residue
|
|
|
Ends of DNA polymer
|
labeled 3' or 5' corresponding to deoxyribose C to which terminal hydroxy group attached
|
|
|
ribonucleic acid
|
polymers conceptually much like DNA polymers except ribose rather than 2'-deoxyribose is sugar
|
|
|
fourth base in RNA
|
uracil instead of thymine - some rare bases found in certain types of RNA
|
|
|
Chargaff's first parity rule
|
ratios of adenine to thymine & guanosine to cytosine are 1.0
|
|
|
Watson & Crick
|
proposed DNA structure based on x-ray diffraction patterns of DNA fibers
|
|
|
structure of DNA
|
2 right handed helical polynuc chains run opp directions coiled around common axis - double helix
|
|
|
the helix makes a complete turn every
|
10 nucleotide residues
|
|
|
sugars & phosphates (rich in OH groups & charges)
|
outside of helix, interact w solvent water or other hydrophilic cmpds
|
|
|
Bases
|
hydrophobic, buried in interior of the double helix away from h2o
|
|
|
Chains held together by
|
H bonds btwn bases
|
|
|
Each A bonds to
|
T
|
|
|
Each G bonds to
|
C
|
|
|
Every purine in one chain H bonded to
|
a pyrimidine in the other
|
|
|
A is
|
complementary to T
|
|
|
Watson-Crick base pairs
|
H bonded AT & GC pairs
|
|
|
Planes of successive complementary base pairs
|
stacked one on top of the other & perpendicular to axis of helix
|
|
|
Distance btwn each successive base pair plane
|
3.4 A
|
|
|
Distance along helix per complete turn
|
10 x 3.4 = 34 A
|
|
|
Double helical structure of DNA results in
|
two grooves, wrap around double helix along periphery
|
|
|
Larger groove
|
Major groove
|
|
|
Smaller groove
|
Minor groove
|
|
|
Grooves, particularly major
|
Sites at which other macromlcs such as proteins are found to interact w DNA
|
|
|
Is there an intrinsic restriction on the sequence of bases in a polynucleotide?
|
No, but bc of base pairing, sequence of one polynucleotide strand (Watson) in double helix complementary to that in the other (Crick) so everywhere there is A, T in other, where G, C in other
|
|
|
Chargaff parity rule accounted for by
|
H bonding complementarity in DNA
|
|
|
How does structure suggest a reasonable mech for the duplication of DNA during cell division
|
two strands come apart & new strand grown as a complement of each original strand
|
|
|
Proper sequence of each new DNA strand during cell reprod ensured by
|
H bonding complementarity
|
|
|
DNA sequencing
|
development of methods for rapidly determining the sequential arrangement of individual bases in DNA
|
|
|
DNA sequences of human genes provide
|
codes that living orgs use for biosynth of messenger RNAs whose sequences serve as codes for biosynth of proteins
|
|
|
Sequence data beginning to
|
unlock genetic bases of diseases & genetic variations that occur among individuals
|
|
|
If H bonding btwn pyrimidine & purine base disrupted
|
Watson-Crick base pairing complementarity can also be disrupted & bio processes that rely on it
|
|
|
Alkylating agents
|
type of chem damage to DNA - react w DNA by alkylating 1+ nucleotide bases
|
|
|
Carcinogens
|
cancer causing cmpds (include alkylating agents: methyl methanesulfonate, dimethyl sulfate)
|
|
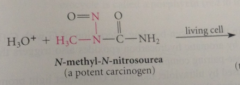
|
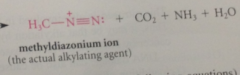
|
|
|
When alkylating agents react w DNA
|
alkylated guanosines are among products
|
|
|
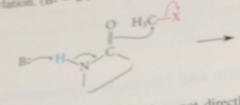
Major product
|
alkylated on N-7 of guanine base, but important minor prod: alkylated on oxygen @ C-6 (O-6 position)
|
|
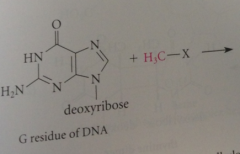
|
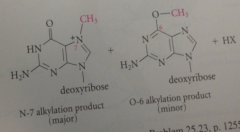
|
|
|
alkylation @ O-6 prevents
|
N-1 nitrogen from acting as a H-bond donor in Watson-Crick base pair bc H lost from N as a result of alkylation
|
|
|
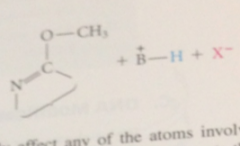
|
|
|
N-7 alkylation
|
does not affect any atoms involved in H-bonding complementarity
|
|
|
Alkylating agents that are most potent carcinogens also yield
|
greatest amt guanines alkylated @ O-6 & thymes alkylated @ O-4
|
|
|
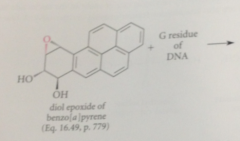
Epoxides have been shown to react w
|
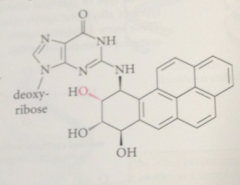
DNA, prod include guanosine residue alkylated on N @ C-2 of the guanine base
|
|
|
|
|
|
N also involved in
|
H-bonding interaction of G w C
|
|
|
May be that alkylation by aromatic hydrocarbon epoxides
|
triggers onset of cancer by interfering w base-pairing complementarity
|
|
|
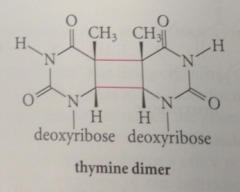
Ultraviolet light promotes
|
cycloaddition of 2 pyrimidines when occur in adjacent positions on a strand of DNA
|
|
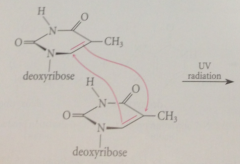
|
|
|
|
Most ppl have bio repair system that
|
effects removal of modified pyrimidines & repairs DNA
|
|
|
Xeroderma pigmentosum
|
genetic deficiency in enzyme that initiates repair - most contract skin cancer, die @ early age
|
|
|
Do N heterocycles occur widely in nature?
|
Yes - alkaloids, many contain heterocyclic ring systems
|
|
|
Naturally occurring amino acids proline, histidine, tryptophan contain
|
pyrrolidine, imidazole & indole ring
|
|
|
Number of vitamins are
|
heterocyclic cmpds w/o with many important metabolic processes could not take place
|
|
|
Porphyrin
|
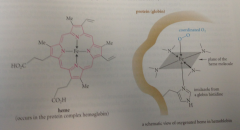
Iron complex of heme, a heterocycle composed of pyrrole units
|
|
|
Heme
|
Fe(II) complex of an aromatic heterocycle found in red blood cells tightly bound to a protein called globin
|
|
|
Iron, held in position by coordination w N of heme & imidazole of globin
|
complexes reversibly w O
|
|
|
Hemoglobin
|
O carrier of blood - oxygenated hemoglobin responsible for red color of blood
|
|
|
Carbon monoxide & cyanide
|
well-known respiratory poisons complex w iron in hemoglobin & w iron in heme groups of other respiratory proteins
|
|
|
chlorophyll
|
class of cmpds closely related to porphyrins, causes green color of plants
|
|
|
Absorption of sunlight by chlorophylls
|
first step in conversion of sunlight into usable energy by plants
|

